Saturated and Unsaturated Hydrocarbons Saturated hydrocarbons have no

Saturated and Unsaturated Hydrocarbons Saturated hydrocarbons have no double bonds. Unsaturated hydrocarbons have one or more double bonds. אלקנים 6© 2014 Pearson Education, Inc. 1
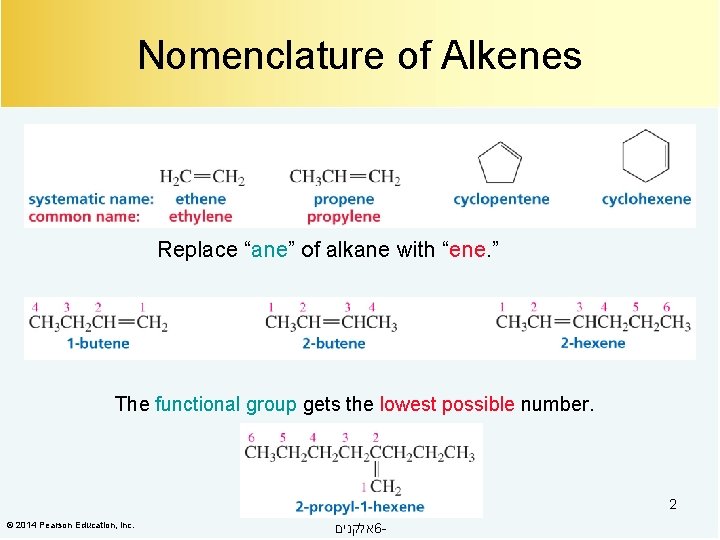
Nomenclature of Alkenes Replace “ane” of alkane with “ene. ” The functional group gets the lowest possible number. 2 © 2014 Pearson Education, Inc. אלקנים 6 -
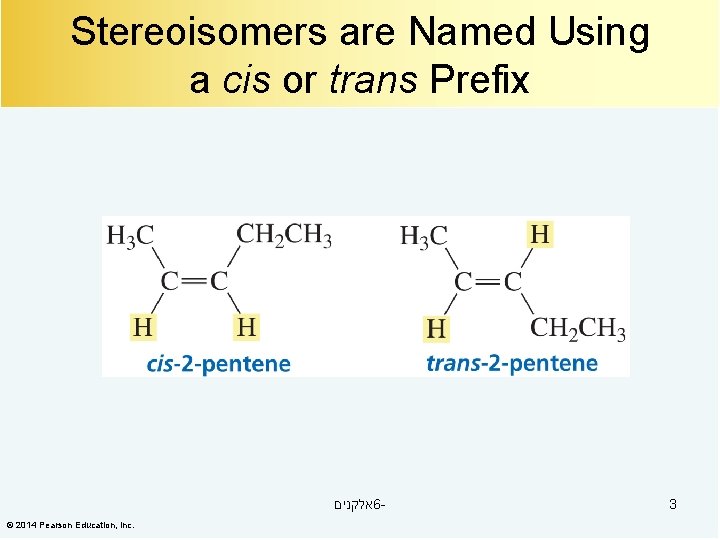
Stereoisomers are Named Using a cis or trans Prefix אלקנים 6© 2014 Pearson Education, Inc. 3
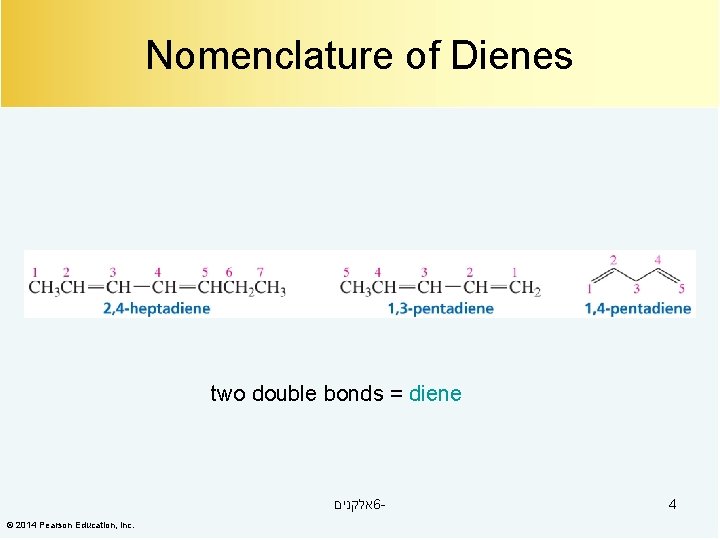
Nomenclature of Dienes two double bonds = diene אלקנים 6© 2014 Pearson Education, Inc. 4
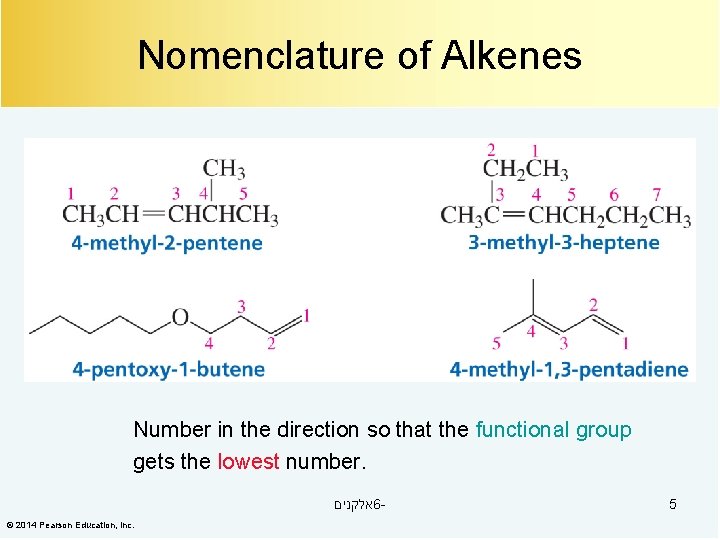
Nomenclature of Alkenes Number in the direction so that the functional group gets the lowest number. אלקנים 6© 2014 Pearson Education, Inc. 5
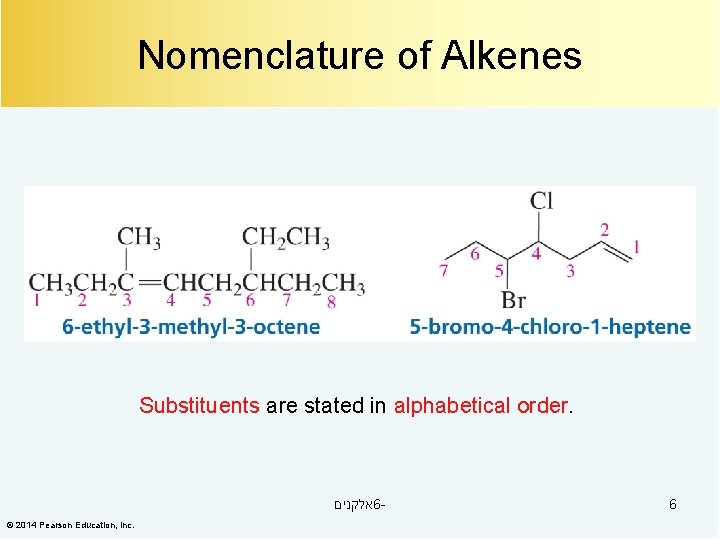
Nomenclature of Alkenes Substituents are stated in alphabetical order. אלקנים 6© 2014 Pearson Education, Inc. 6

Nomenclature of Alkenes אלקנים 6© 2014 Pearson Education, Inc. 7
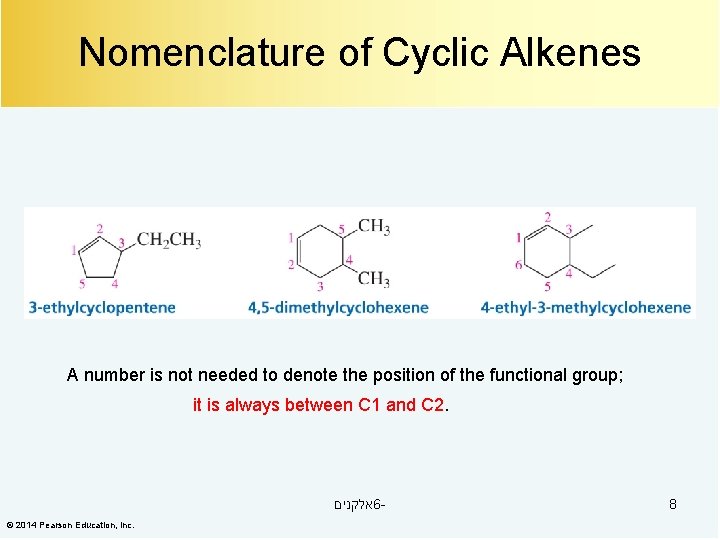
Nomenclature of Cyclic Alkenes A number is not needed to denote the position of the functional group; it is always between C 1 and C 2. אלקנים 6© 2014 Pearson Education, Inc. 8
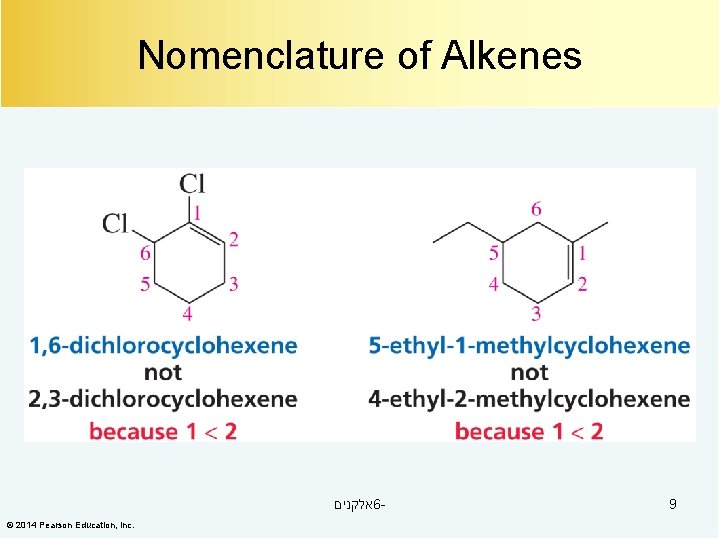
Nomenclature of Alkenes אלקנים 6© 2014 Pearson Education, Inc. 9
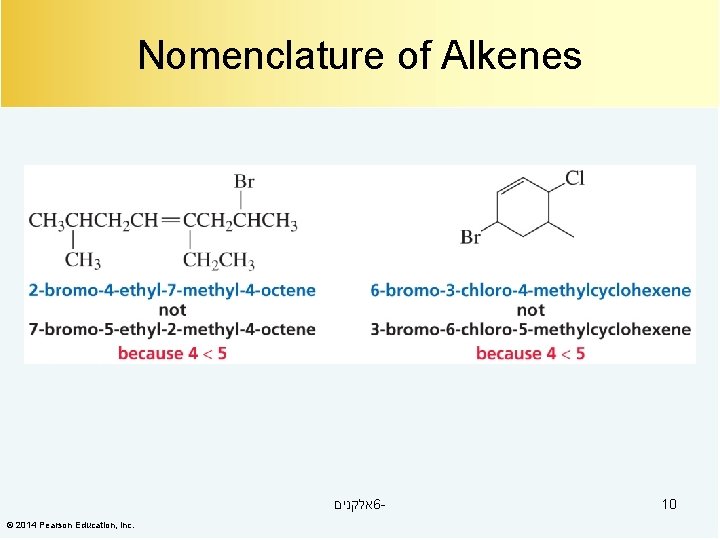
Nomenclature of Alkenes אלקנים 6© 2014 Pearson Education, Inc. 10

Vinylic and Allylic Carbons vinylic carbon: the sp 2 carbon of an alkene allylic carbon: a carbon adjacent to a vinylic carbon © 2014 Pearson Education, Inc. אלקנים 6 - 11
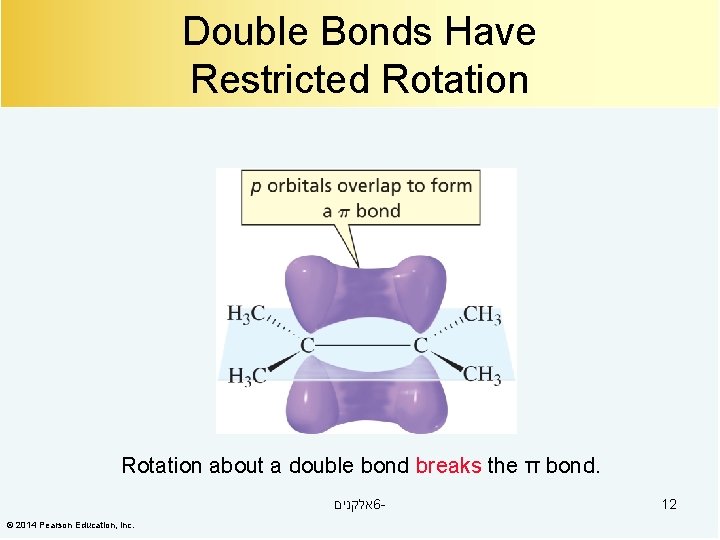
Double Bonds Have Restricted Rotation about a double bond breaks the π bond. אלקנים 6© 2014 Pearson Education, Inc. 12
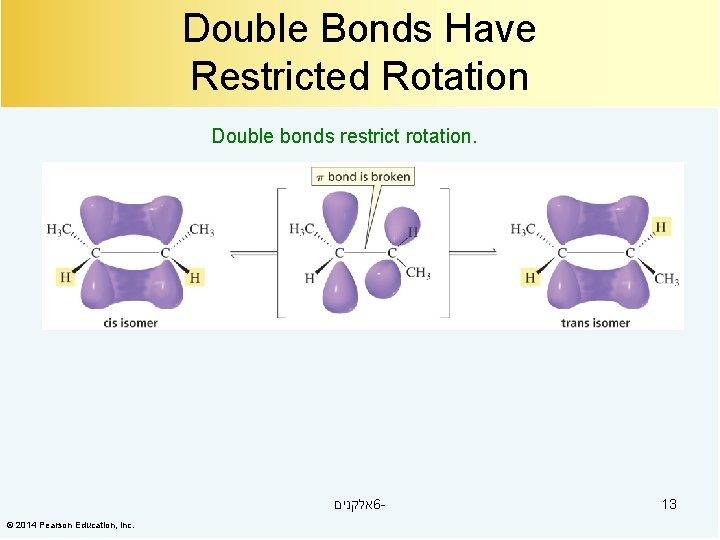
Double Bonds Have Restricted Rotation Double bonds restrict rotation. אלקנים 6© 2014 Pearson Education, Inc. 13

Six Atoms of an Alkene are in the Same Plane אלקנים 6© 2014 Pearson Education, Inc. 14
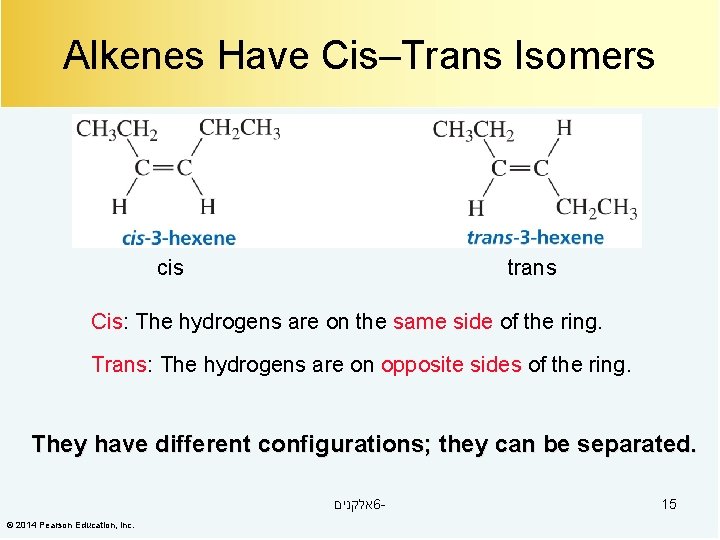
Alkenes Have Cis–Trans Isomers cis trans Cis: The hydrogens are on the same side of the ring. Trans: The hydrogens are on opposite sides of the ring. They have different configurations; they can be separated. אלקנים 6© 2014 Pearson Education, Inc. 15
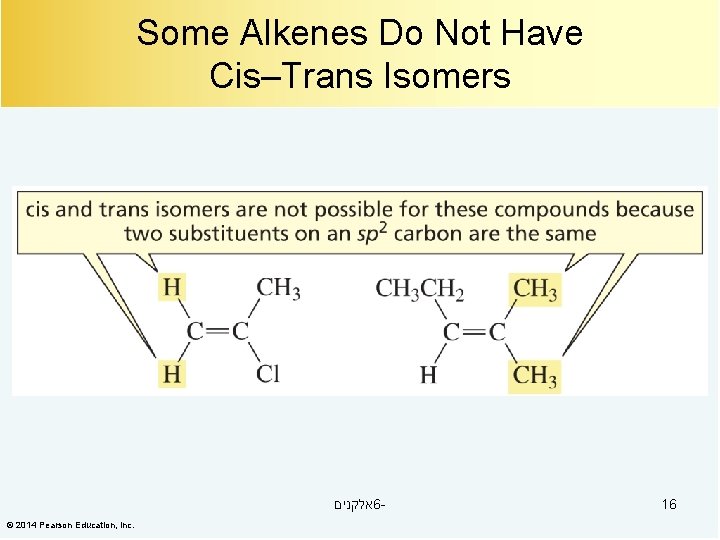
Some Alkenes Do Not Have Cis–Trans Isomers אלקנים 6© 2014 Pearson Education, Inc. 16
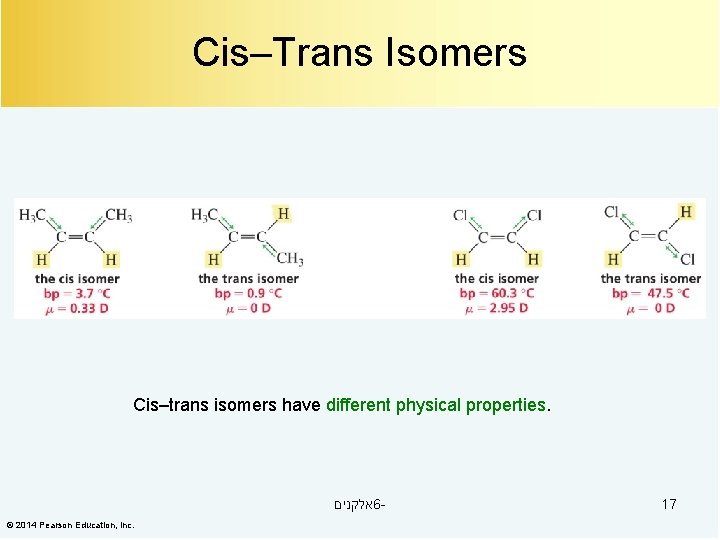
Cis–Trans Isomers Cis–trans isomers have different physical properties. אלקנים 6© 2014 Pearson Education, Inc. 17
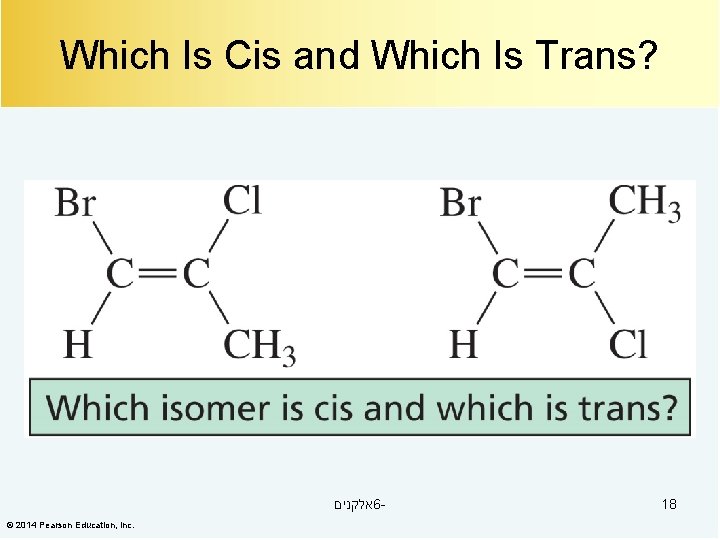
Which Is Cis and Which Is Trans? אלקנים 6© 2014 Pearson Education, Inc. 18
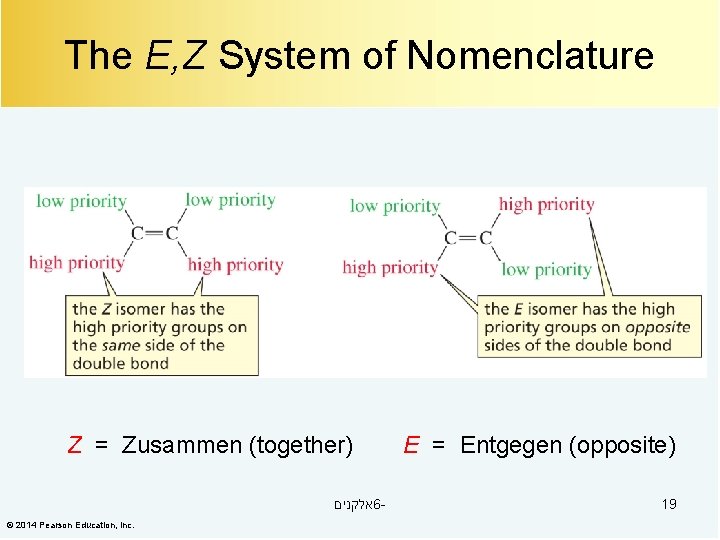
The E, Z System of Nomenclature Z = Zusammen (together) אלקנים 6© 2014 Pearson Education, Inc. E = Entgegen (opposite) 19
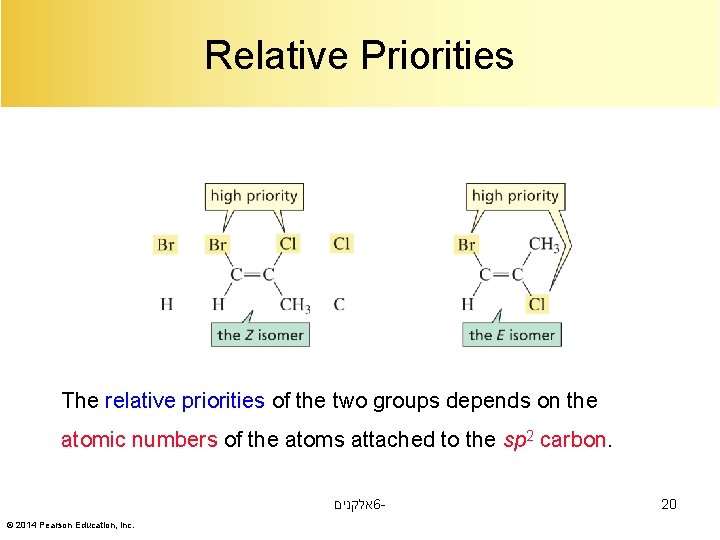
Relative Priorities The relative priorities of the two groups depends on the atomic numbers of the atoms attached to the sp 2 carbon. אלקנים 6© 2014 Pearson Education, Inc. 20
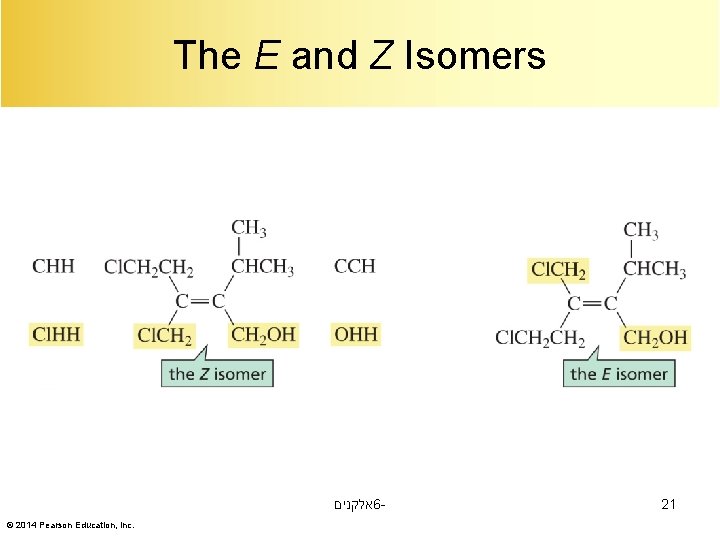
The E and Z Isomers אלקנים 6© 2014 Pearson Education, Inc. 21
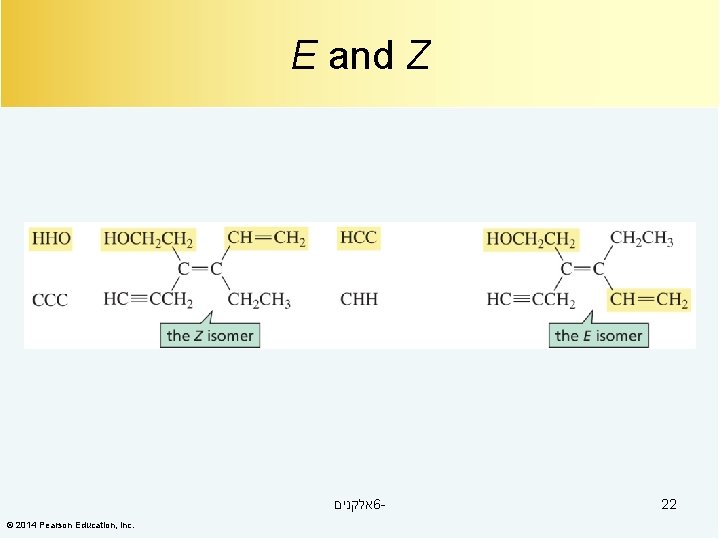
E and Z אלקנים 6© 2014 Pearson Education, Inc. 22
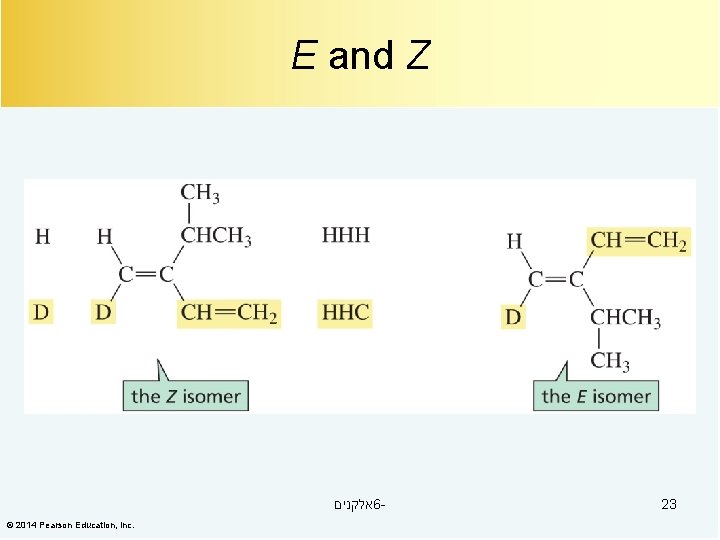
E and Z אלקנים 6© 2014 Pearson Education, Inc. 23
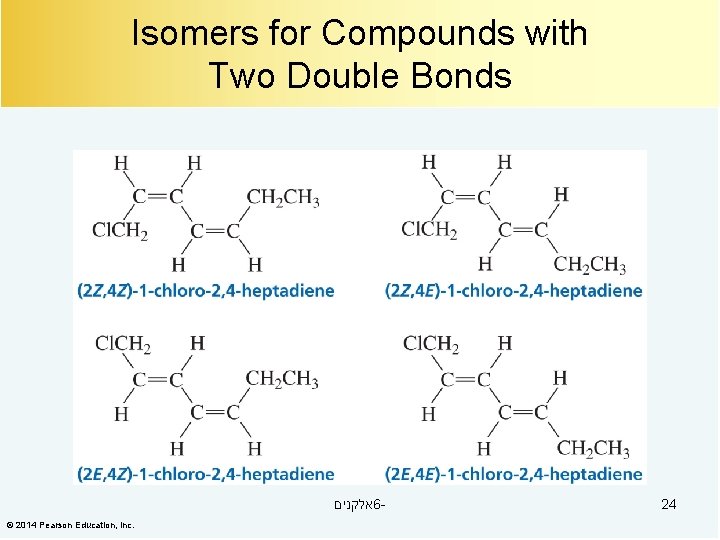
Isomers for Compounds with Two Double Bonds אלקנים 6© 2014 Pearson Education, Inc. 24
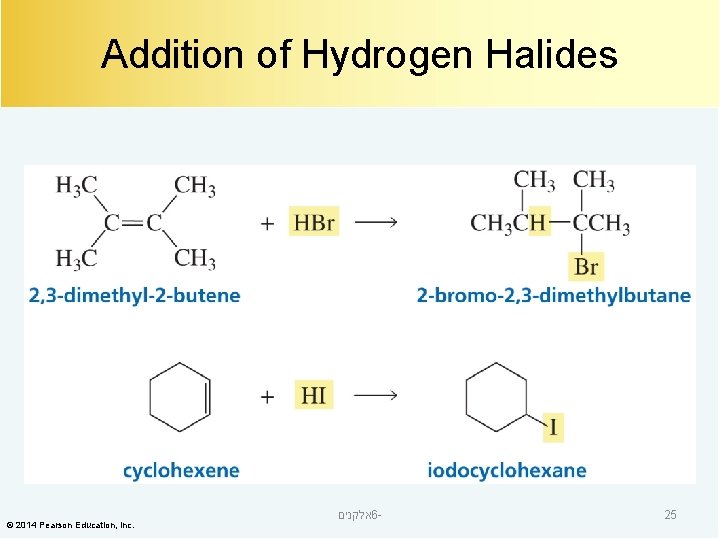
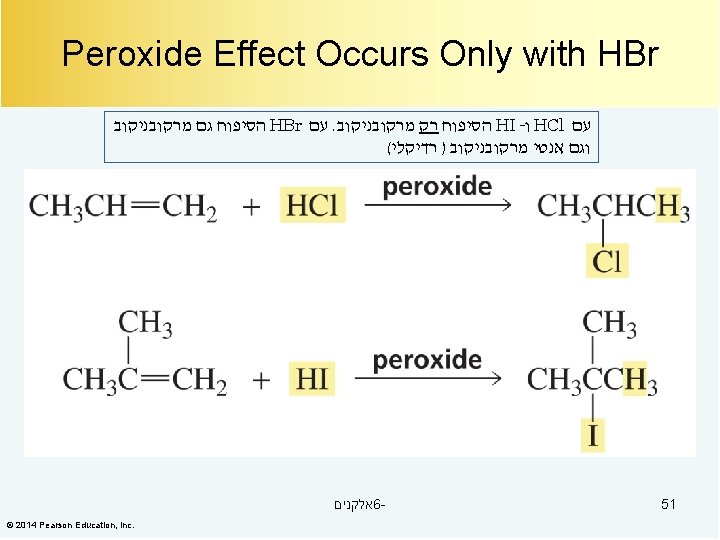
Addition of Hydrogen Halides © 2014 Pearson Education, Inc. אלקנים 6 - 25
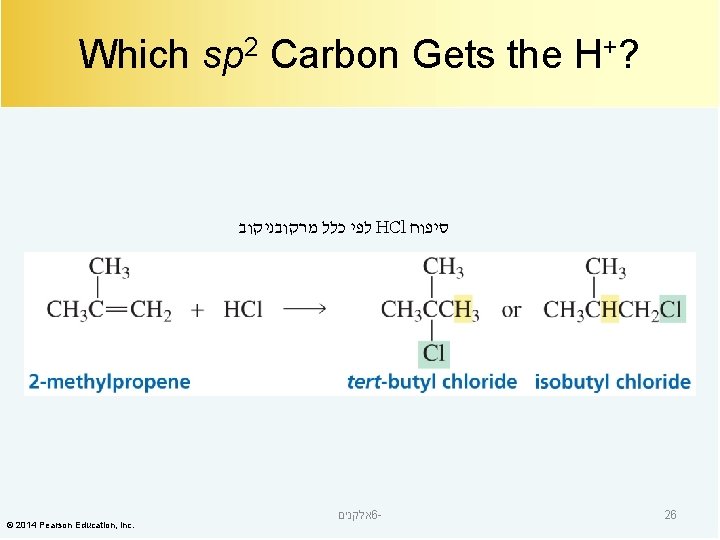
Which sp 2 Carbon Gets the H+? לפי כלל מרקובניקוב HCl סיפוח © 2014 Pearson Education, Inc. אלקנים 6 - 26
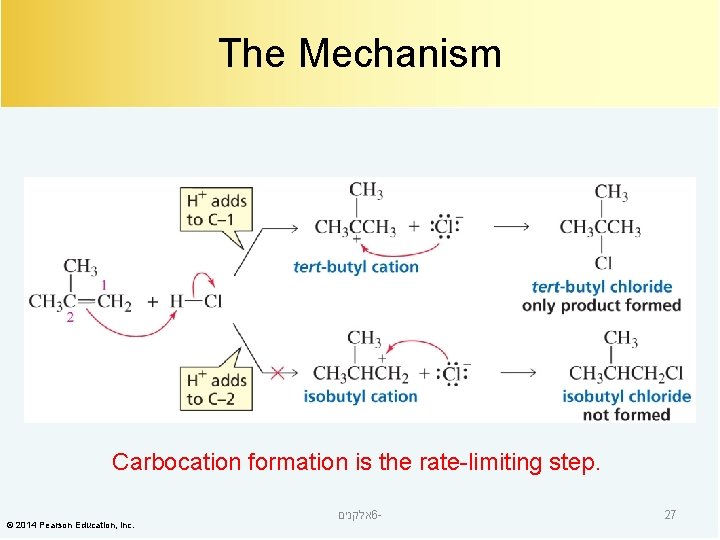
The Mechanism Carbocation formation is the rate-limiting step. © 2014 Pearson Education, Inc. אלקנים 6 - 27
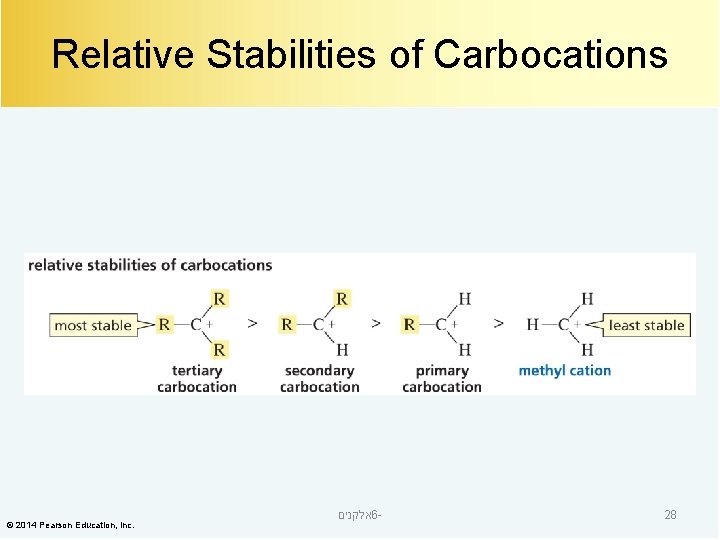
Relative Stabilities of Carbocations © 2014 Pearson Education, Inc. אלקנים 6 - 28
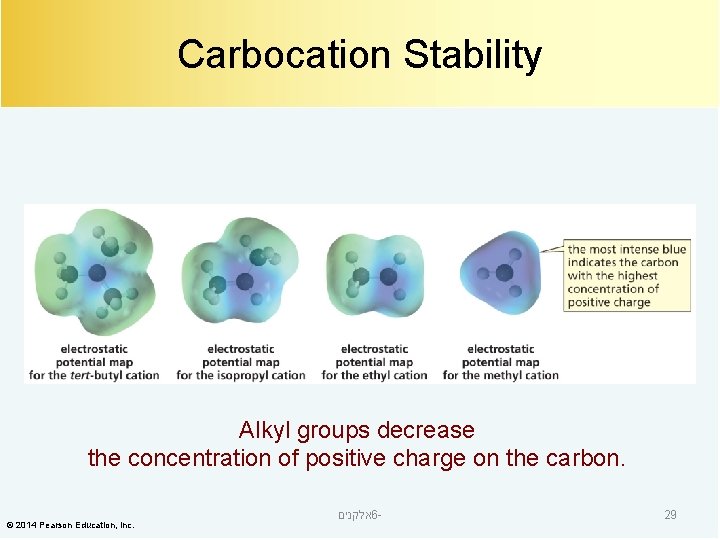
Carbocation Stability Alkyl groups decrease the concentration of positive charge on the carbon. © 2014 Pearson Education, Inc. אלקנים 6 - 29
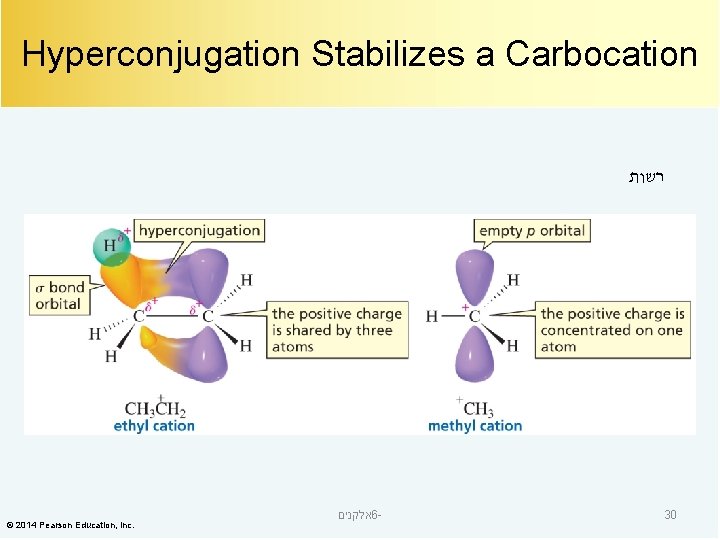
Hyperconjugation Stabilizes a Carbocation רשות © 2014 Pearson Education, Inc. אלקנים 6 - 30
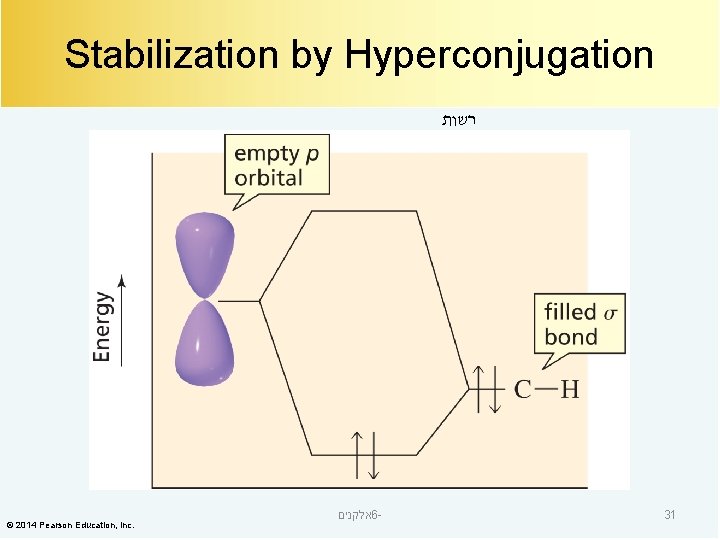
Stabilization by Hyperconjugation רשות © 2014 Pearson Education, Inc. אלקנים 6 - 31
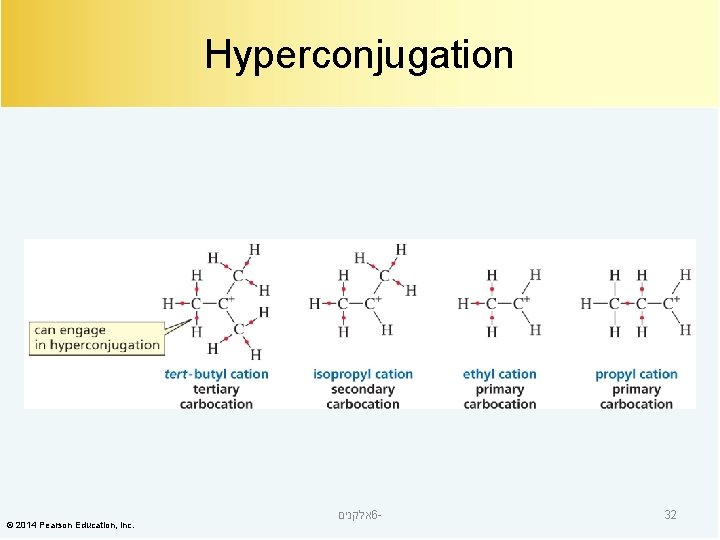
Hyperconjugation © 2014 Pearson Education, Inc. אלקנים 6 - 32
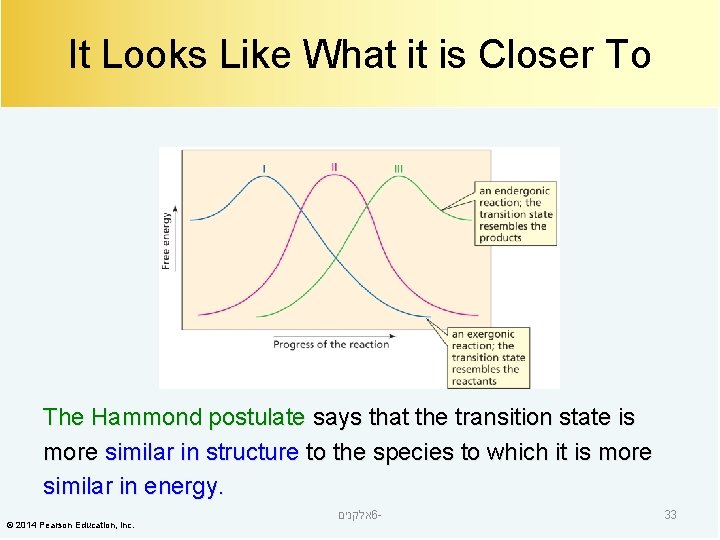
It Looks Like What it is Closer To The Hammond postulate says that the transition state is more similar in structure to the species to which it is more similar in energy. © 2014 Pearson Education, Inc. אלקנים 6 - 33
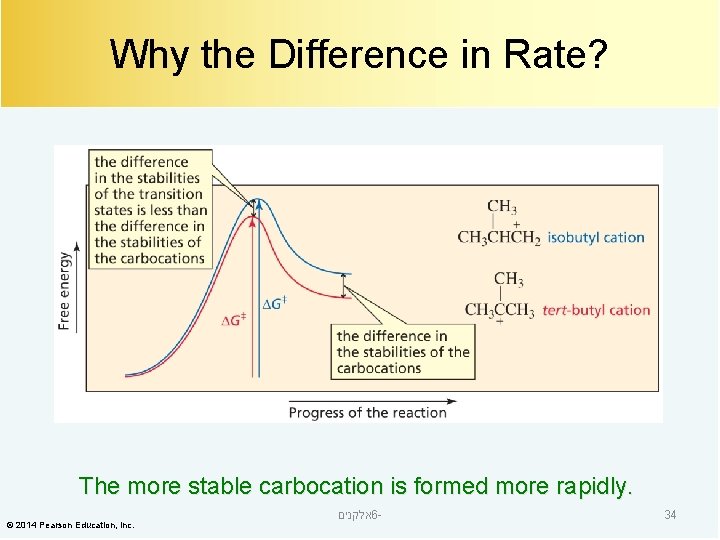
Why the Difference in Rate? The more stable carbocation is formed more rapidly. © 2014 Pearson Education, Inc. אלקנים 6 - 34
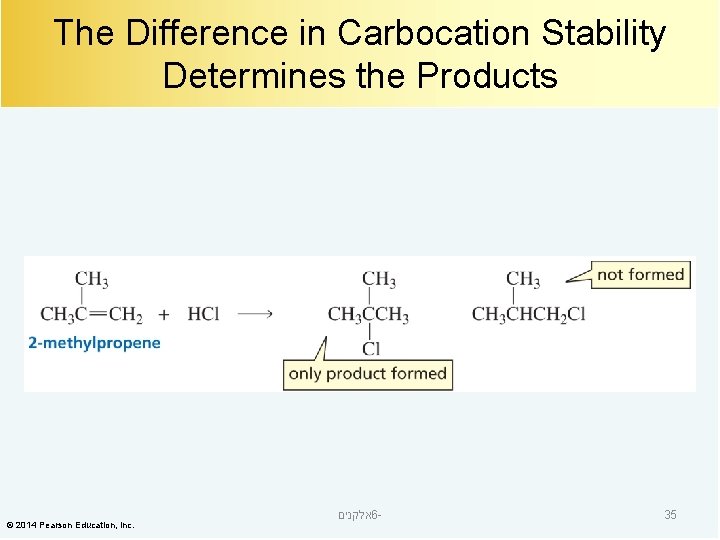
The Difference in Carbocation Stability Determines the Products © 2014 Pearson Education, Inc. אלקנים 6 - 35
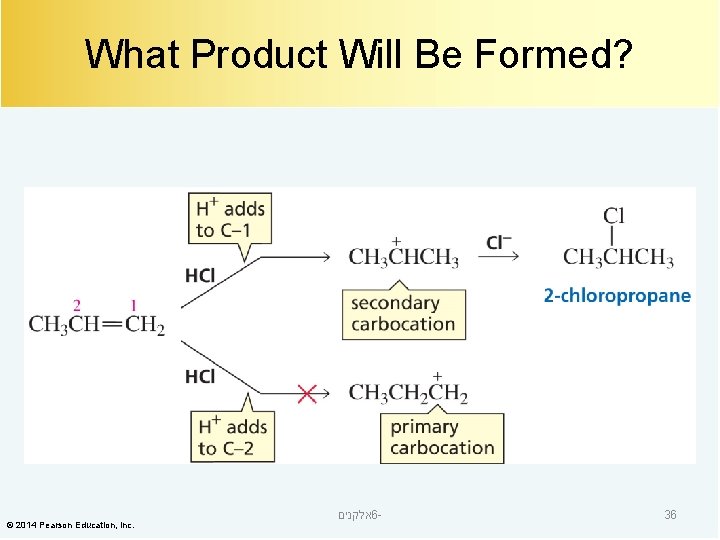
What Product Will Be Formed? © 2014 Pearson Education, Inc. אלקנים 6 - 36
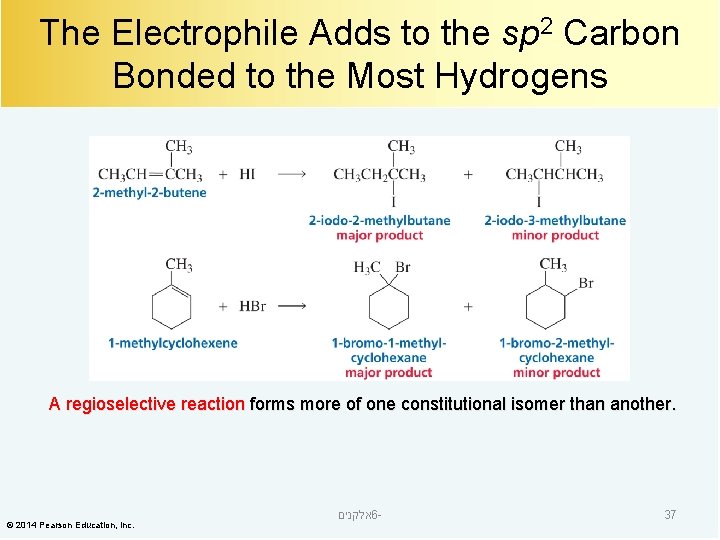
The Electrophile Adds to the sp 2 Carbon Bonded to the Most Hydrogens A regioselective reaction forms more of one constitutional isomer than another. © 2014 Pearson Education, Inc. אלקנים 6 - 37

Not Regioselective © 2014 Pearson Education, Inc. אלקנים 6 - 38
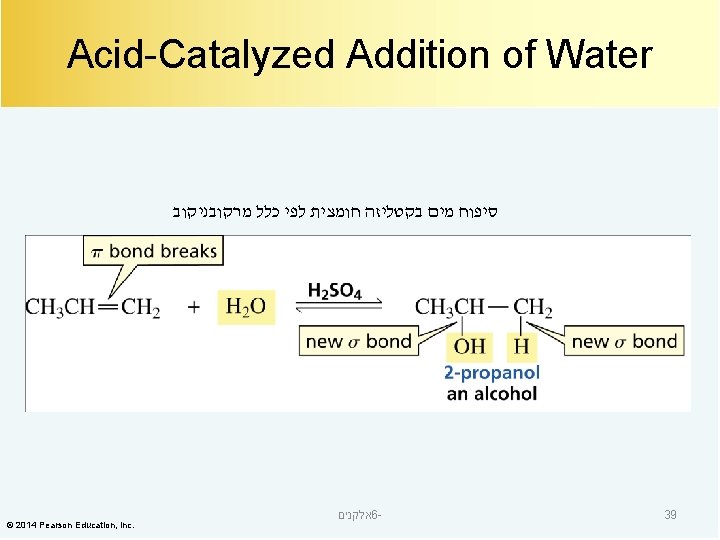
Acid-Catalyzed Addition of Water סיפוח מים בקטליזה חומצית לפי כלל מרקובניקוב © 2014 Pearson Education, Inc. אלקנים 6 - 39
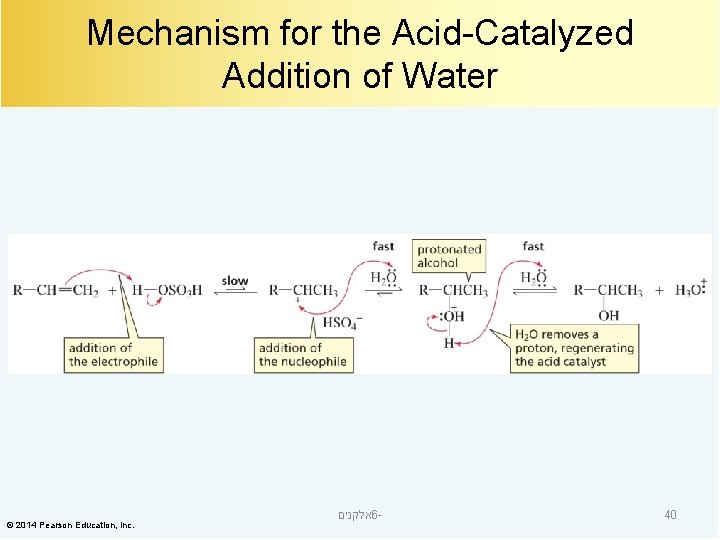
Mechanism for the Acid-Catalyzed Addition of Water © 2014 Pearson Education, Inc. אלקנים 6 - 40
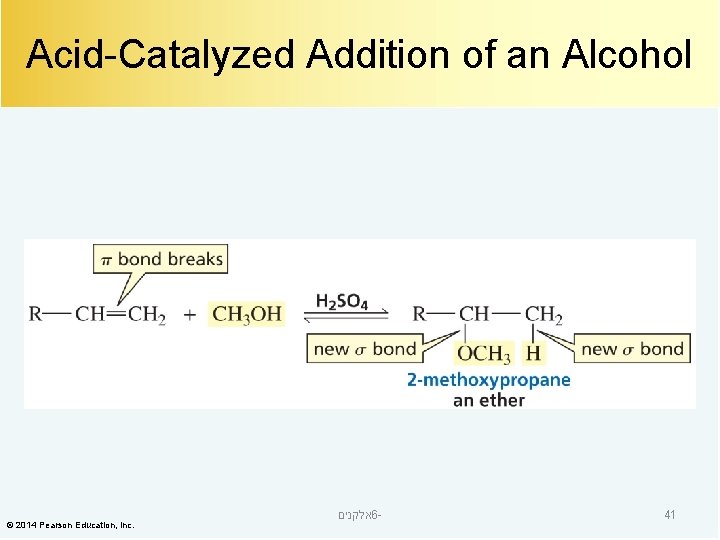
Acid-Catalyzed Addition of an Alcohol © 2014 Pearson Education, Inc. אלקנים 6 - 41

Mechanism for the Acid-Catalyzed Addition of an Alcohol © 2014 Pearson Education, Inc. אלקנים 6 - 42
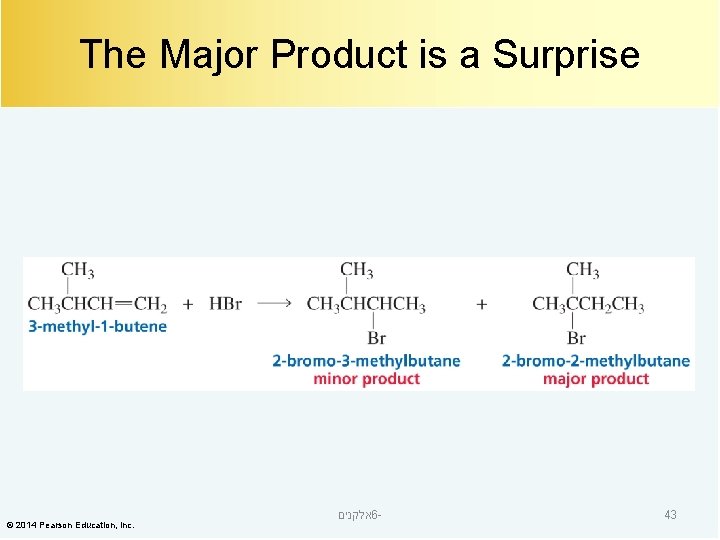
The Major Product is a Surprise © 2014 Pearson Education, Inc. אלקנים 6 - 43
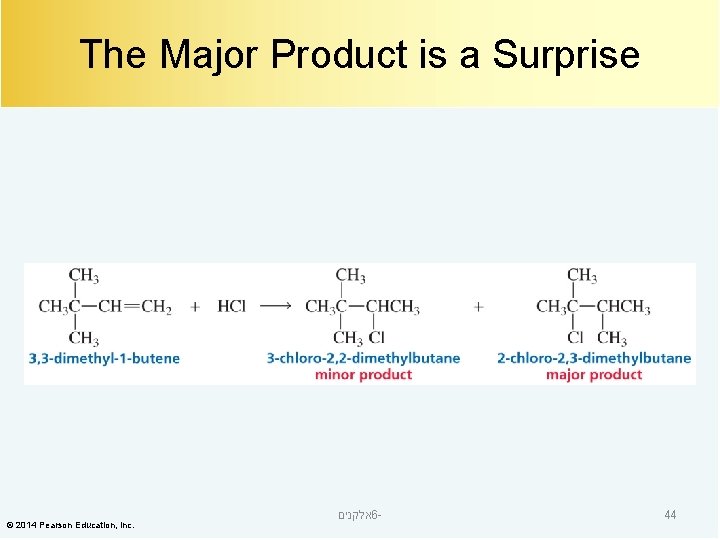
The Major Product is a Surprise © 2014 Pearson Education, Inc. אלקנים 6 - 44
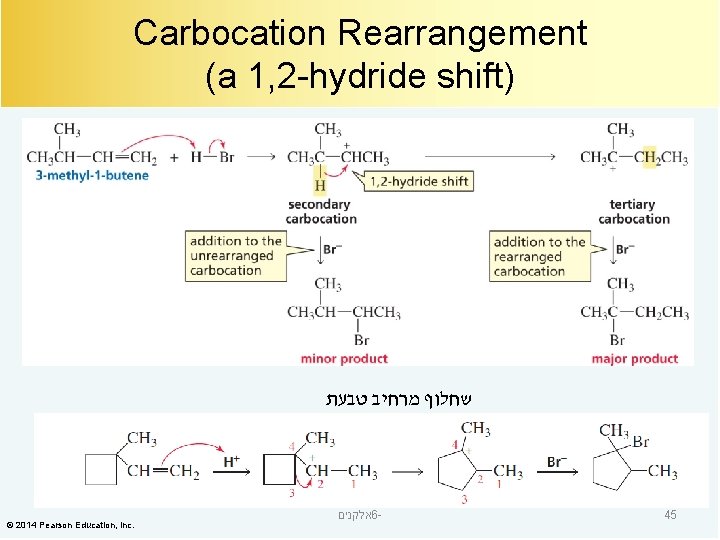
Carbocation Rearrangement (a 1, 2 -hydride shift) שחלוף מרחיב טבעת © 2014 Pearson Education, Inc. אלקנים 6 - 45
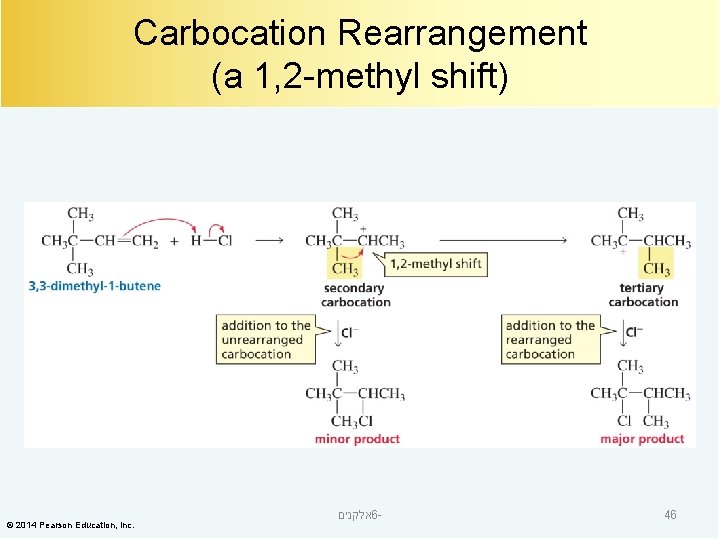
Carbocation Rearrangement (a 1, 2 -methyl shift) © 2014 Pearson Education, Inc. אלקנים 6 - 46

The Carbocation Does Not Rearrange (No Improvement in Carbocation Stability) © 2014 Pearson Education, Inc. אלקנים 6 - 47
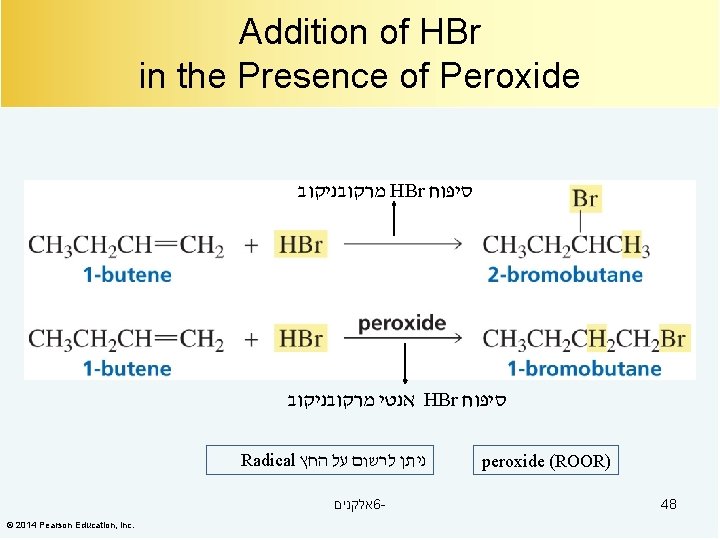
Addition of HBr in the Presence of Peroxide מרקובניקוב HBr סיפוח אנטי מרקובניקוב HBr סיפוח Radical ניתן לרשום על החץ אלקנים 6© 2014 Pearson Education, Inc. peroxide (ROOR) 48
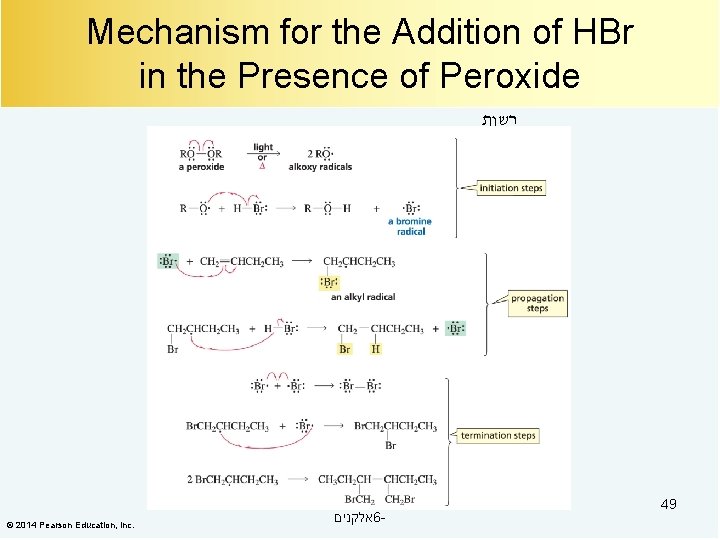
Mechanism for the Addition of HBr in the Presence of Peroxide רשות © 2014 Pearson Education, Inc. אלקנים 6 - 49

A Carbocation Intermediate is Not Formed, So a Carbocation Rearrangement Does Not Occur אלקנים 6© 2014 Pearson Education, Inc. 50
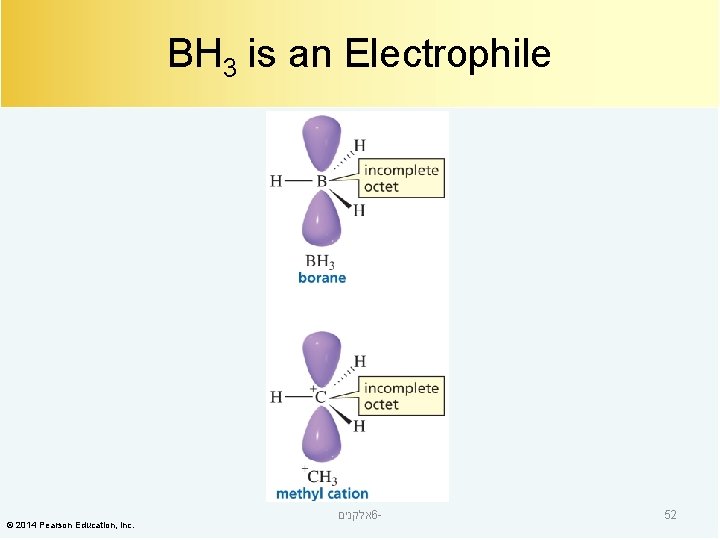
BH 3 is an Electrophile © 2014 Pearson Education, Inc. אלקנים 6 - 52
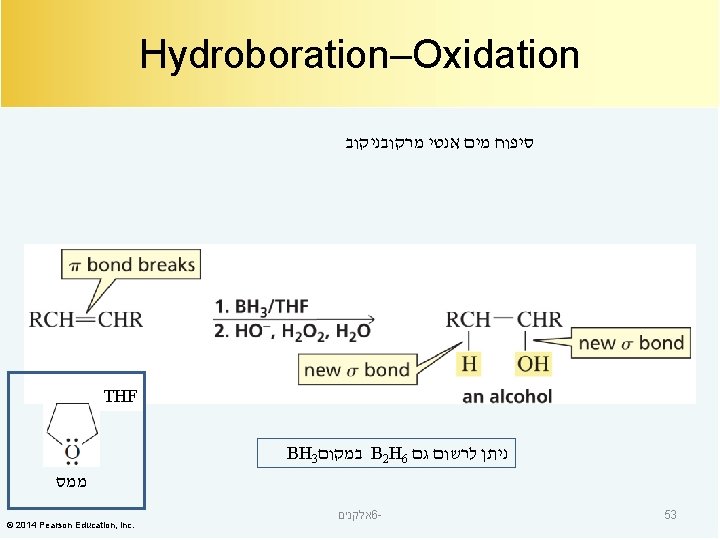

The Electrophile Adds to the sp 2 Carbon Bonded to the Most Hydrogens סיפוח מים מרקובניקוב סיפוח מים אנטי מרקובניקוב The reagents are numbered because the second set of reagents is not added until the first reaction is over. © 2014 Pearson Education, Inc. אלקנים 6 - 54
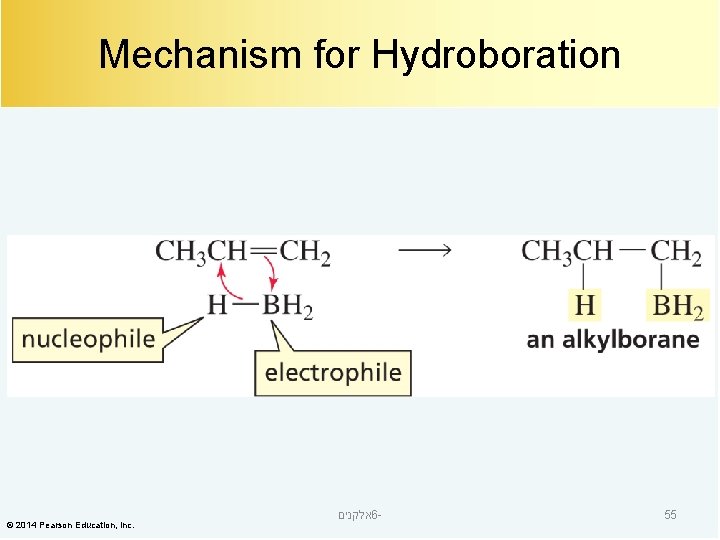
Mechanism for Hydroboration © 2014 Pearson Education, Inc. אלקנים 6 - 55
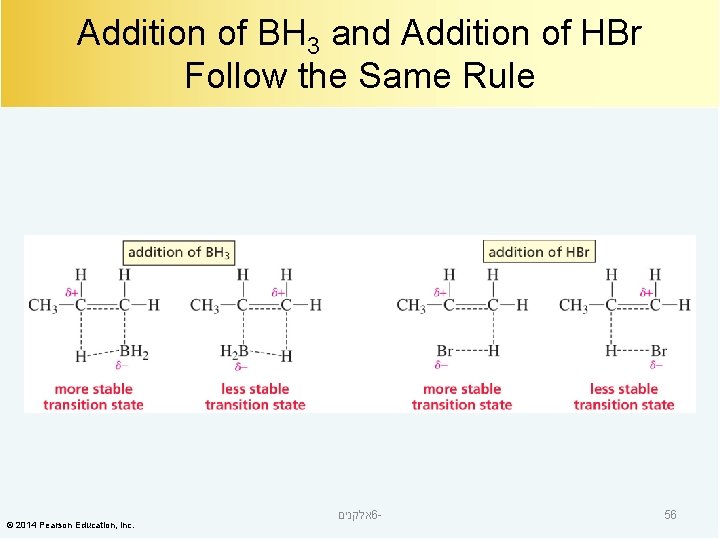
Addition of BH 3 and Addition of HBr Follow the Same Rule © 2014 Pearson Education, Inc. אלקנים 6 - 56
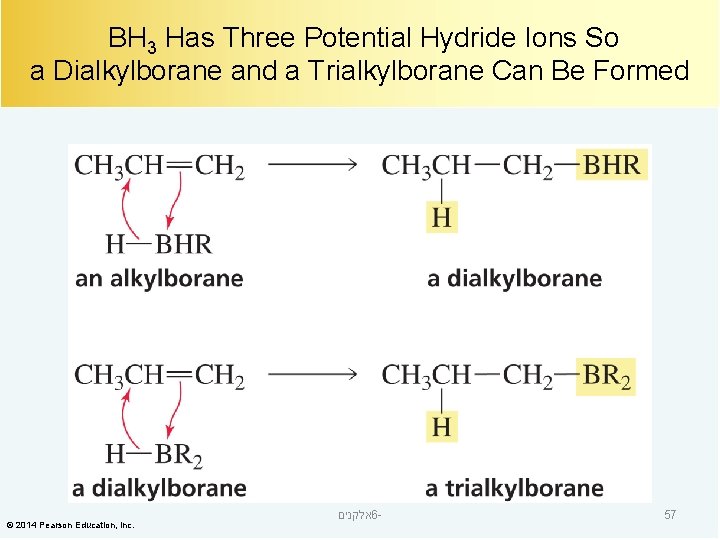
BH 3 Has Three Potential Hydride Ions So a Dialkylborane and a Trialkylborane Can Be Formed © 2014 Pearson Education, Inc. אלקנים 6 - 57
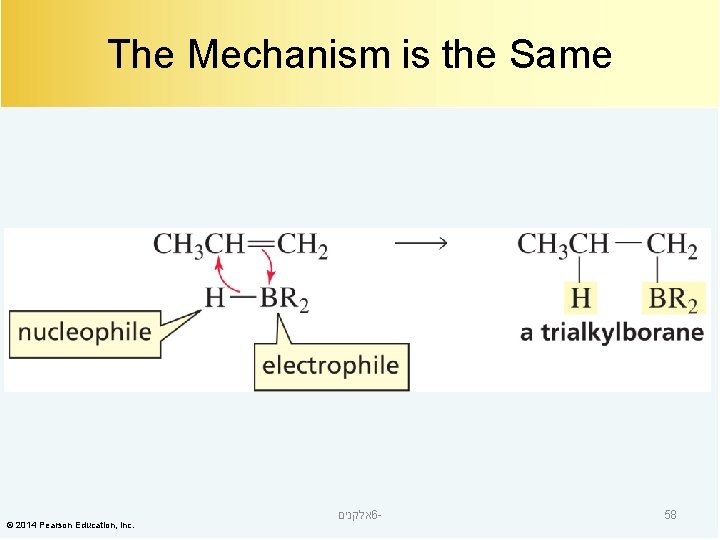
The Mechanism is the Same © 2014 Pearson Education, Inc. אלקנים 6 - 58

OH Replaces BR 2 © 2014 Pearson Education, Inc. אלקנים 6 - 59
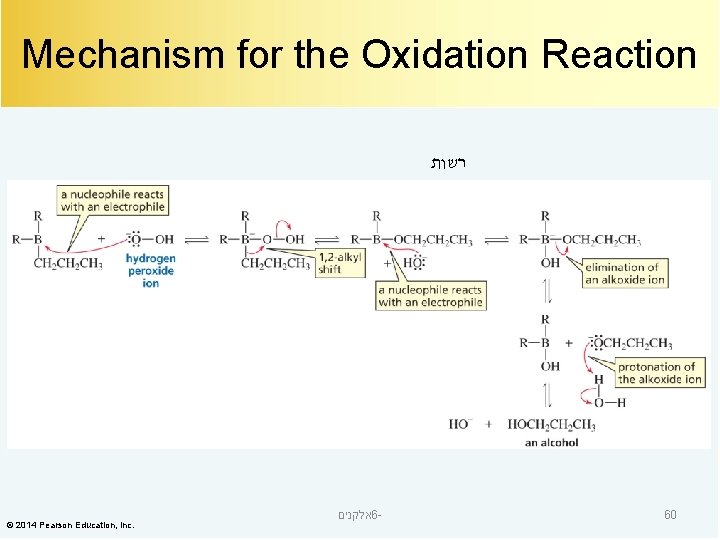
Mechanism for the Oxidation Reaction רשות © 2014 Pearson Education, Inc. אלקנים 6 - 60

No Carbocation Rearrangements © 2014 Pearson Education, Inc. אלקנים 6 - 61

Addition of Br 2 or Cl 2 © 2014 Pearson Education, Inc. אלקנים 6 - 62
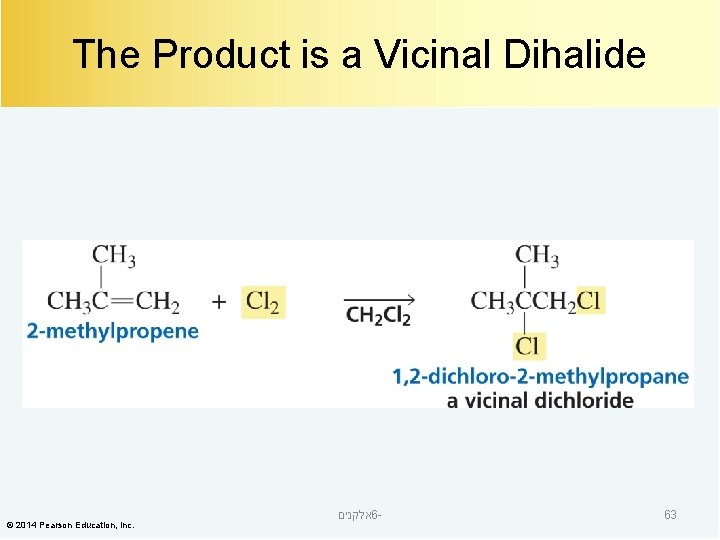
The Product is a Vicinal Dihalide © 2014 Pearson Education, Inc. אלקנים 6 - 63
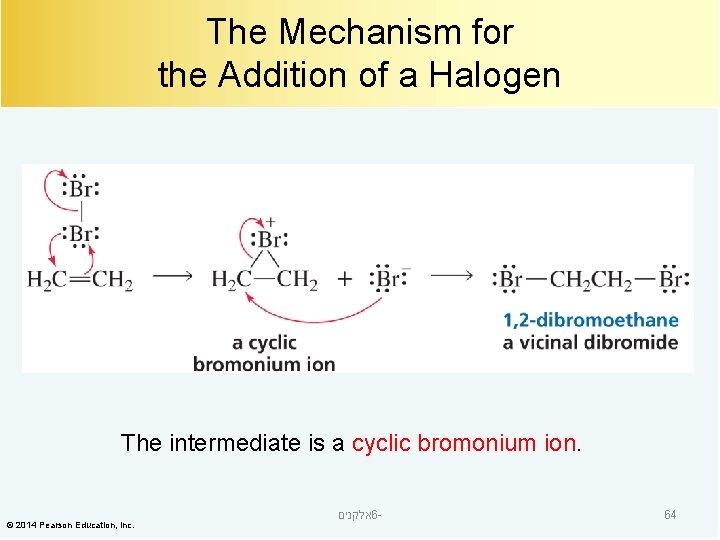
The Mechanism for the Addition of a Halogen The intermediate is a cyclic bromonium ion. © 2014 Pearson Education, Inc. אלקנים 6 - 64

A Cyclic Bromonium Ion The carbons are the most electrophilic atoms. © 2014 Pearson Education, Inc. אלקנים 6 - 65
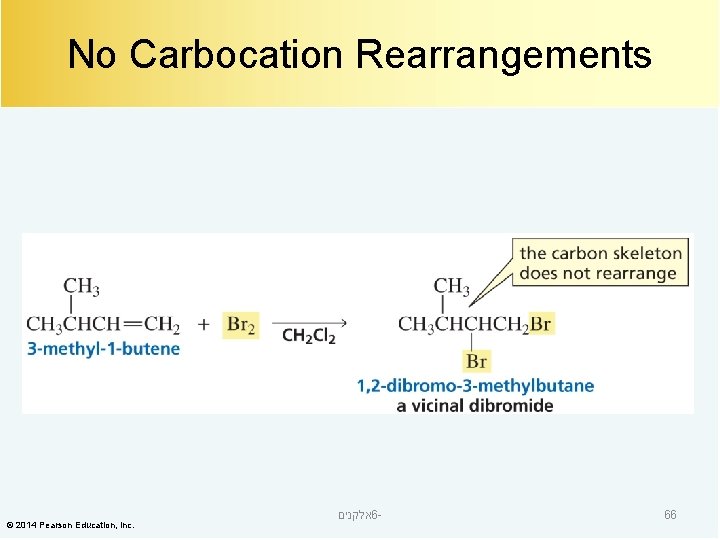
No Carbocation Rearrangements © 2014 Pearson Education, Inc. אלקנים 6 - 66

Alkenes Do Not Add I 2 © 2014 Pearson Education, Inc. אלקנים 6 - 67
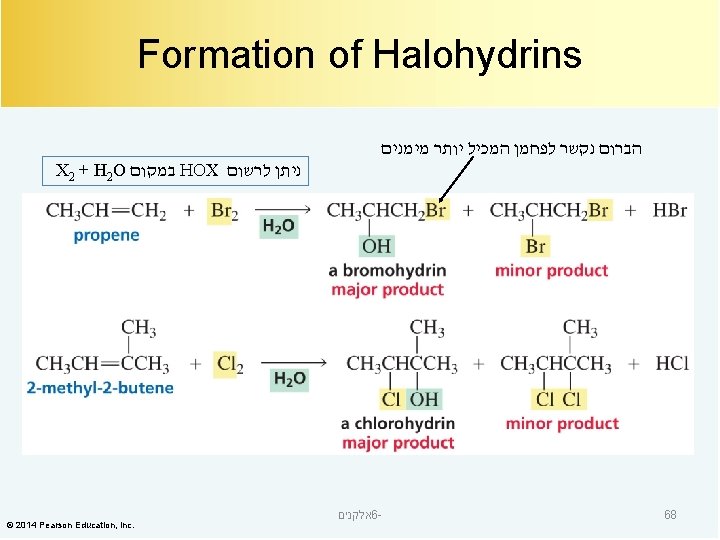
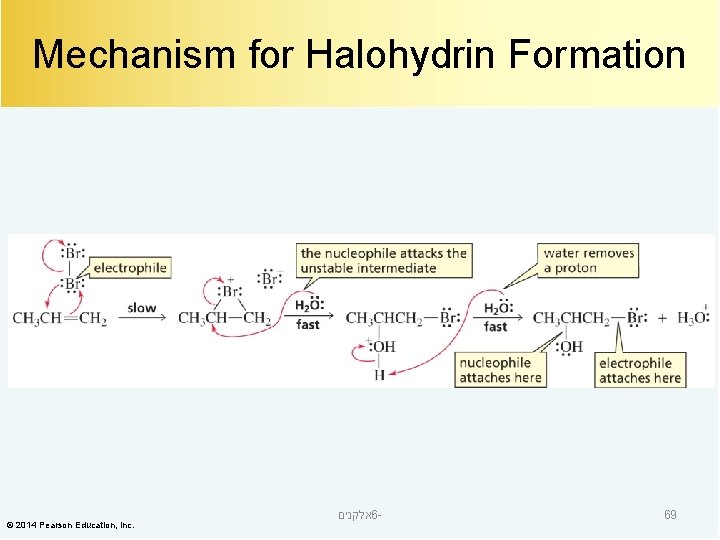
Mechanism for Halohydrin Formation © 2014 Pearson Education, Inc. אלקנים 6 - 69
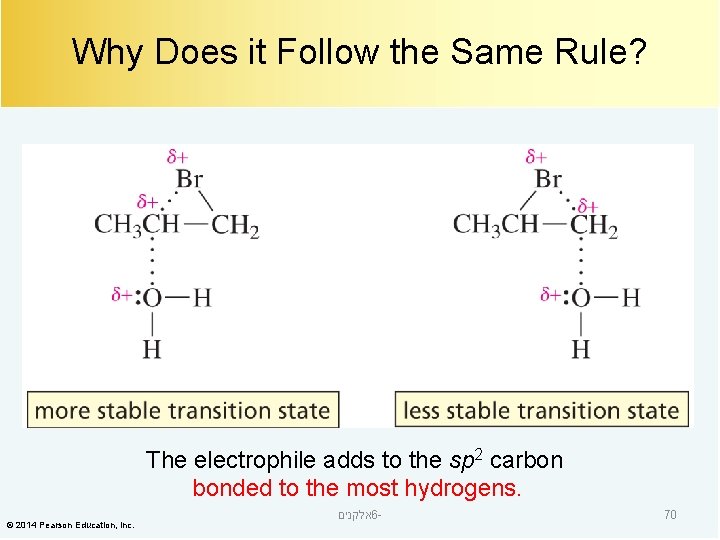
Why Does it Follow the Same Rule? The electrophile adds to the sp 2 carbon bonded to the most hydrogens. © 2014 Pearson Education, Inc. אלקנים 6 - 70
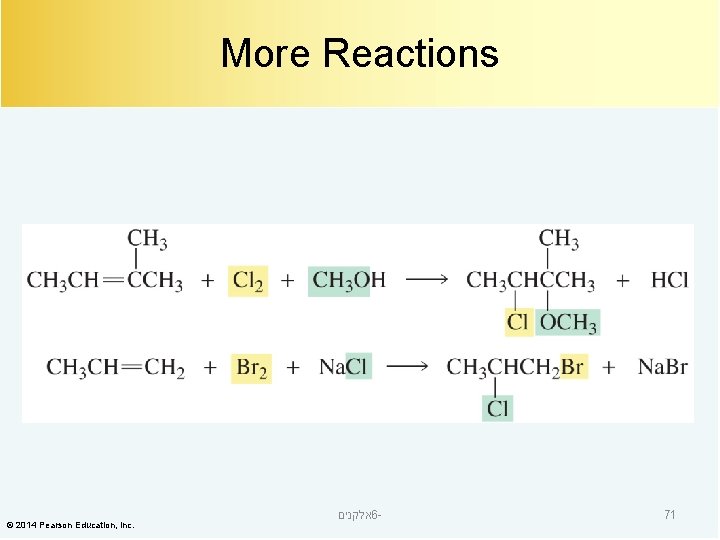
More Reactions © 2014 Pearson Education, Inc. אלקנים 6 - 71
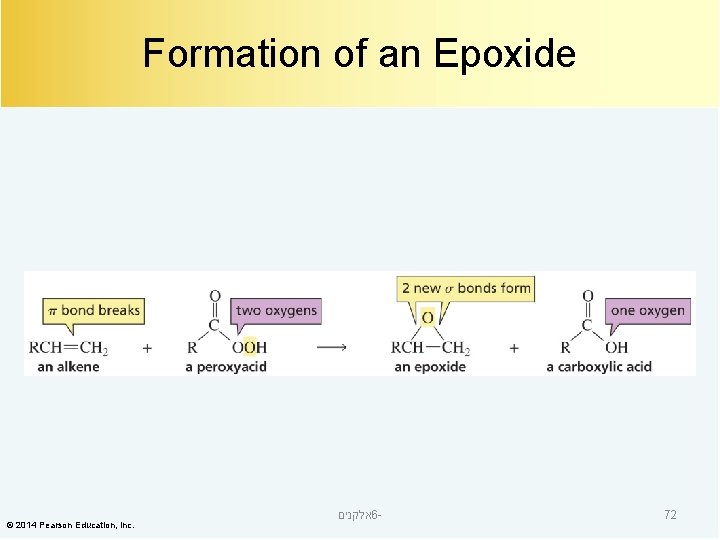
Formation of an Epoxide © 2014 Pearson Education, Inc. אלקנים 6 - 72
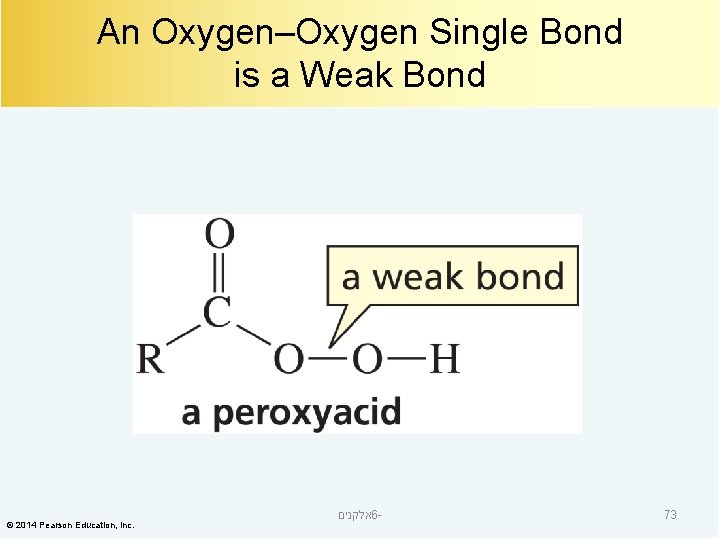
An Oxygen–Oxygen Single Bond is a Weak Bond © 2014 Pearson Education, Inc. אלקנים 6 - 73
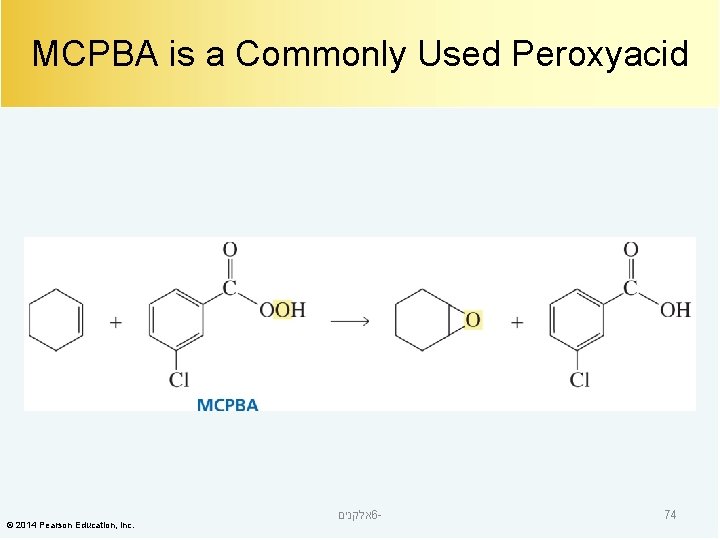
MCPBA is a Commonly Used Peroxyacid © 2014 Pearson Education, Inc. אלקנים 6 - 74
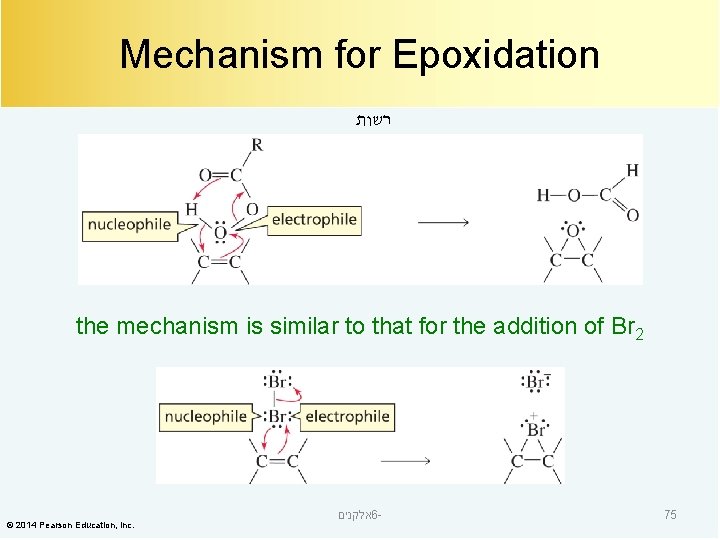
Mechanism for Epoxidation רשות the mechanism is similar to that for the addition of Br 2 © 2014 Pearson Education, Inc. אלקנים 6 - 75
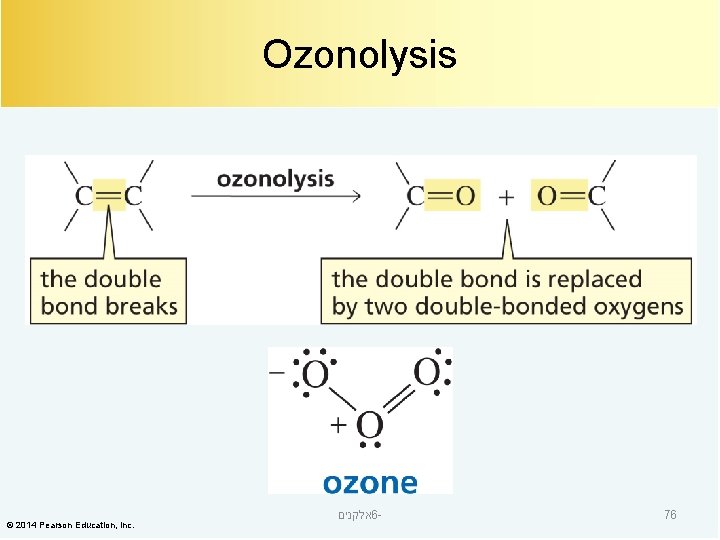
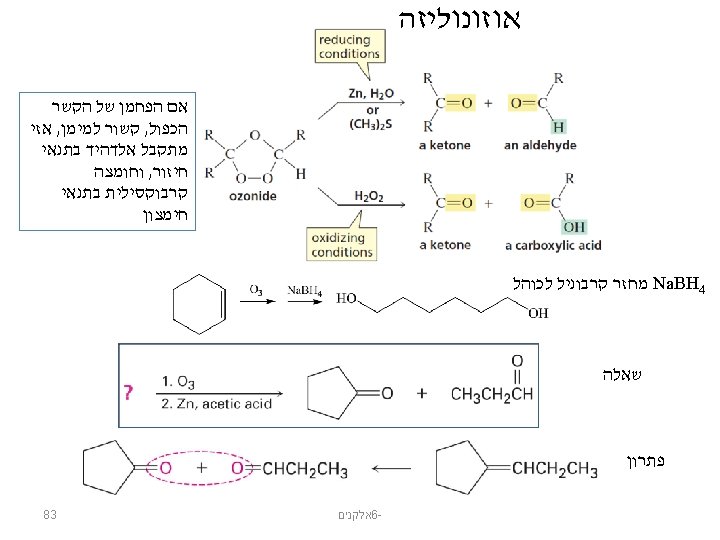
Ozonolysis © 2014 Pearson Education, Inc. אלקנים 6 - 76
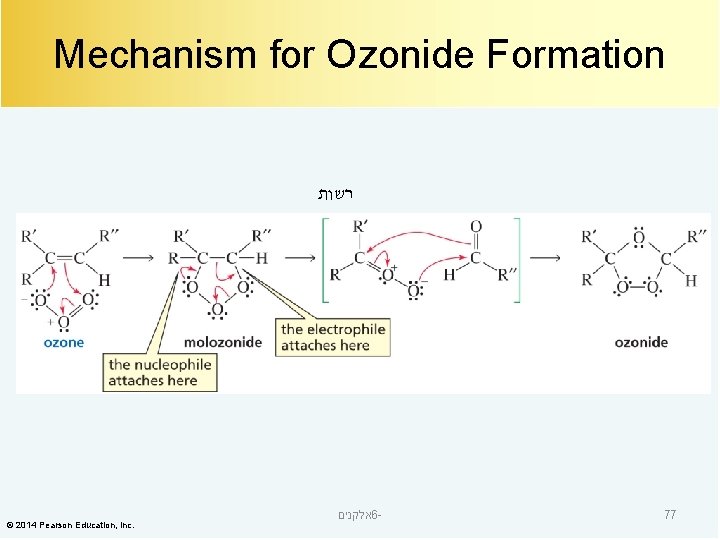
Mechanism for Ozonide Formation רשות © 2014 Pearson Education, Inc. אלקנים 6 - 77
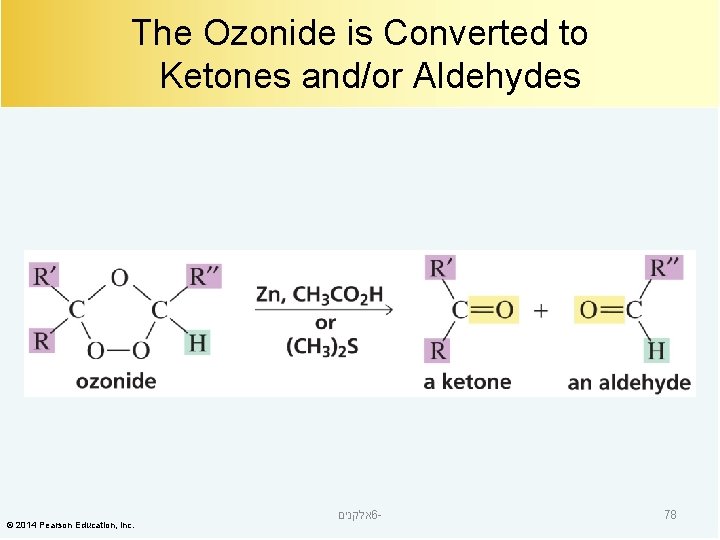
The Ozonide is Converted to Ketones and/or Aldehydes © 2014 Pearson Education, Inc. אלקנים 6 - 78
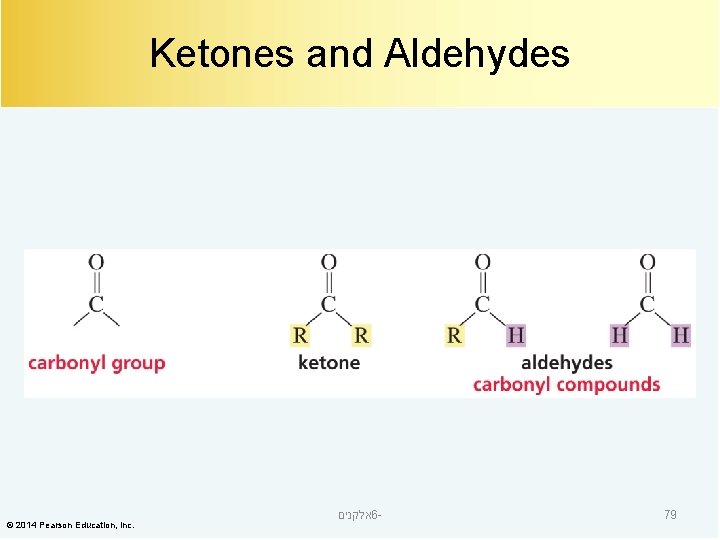
Ketones and Aldehydes © 2014 Pearson Education, Inc. אלקנים 6 - 79
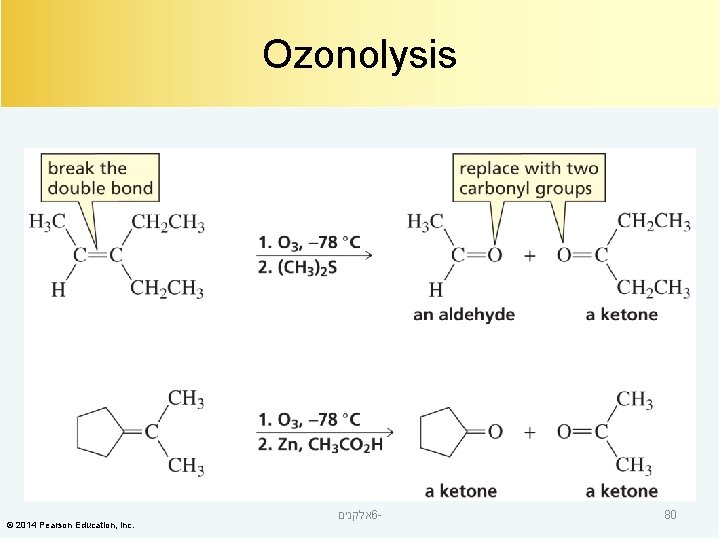
Ozonolysis © 2014 Pearson Education, Inc. אלקנים 6 - 80
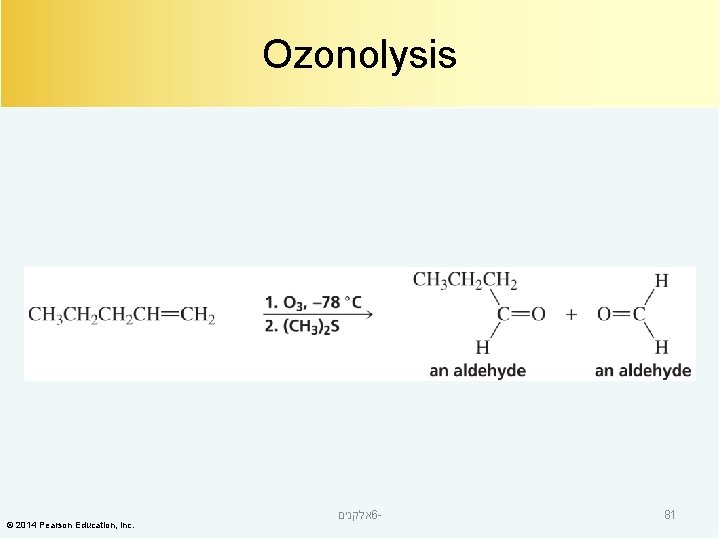
Ozonolysis © 2014 Pearson Education, Inc. אלקנים 6 - 81
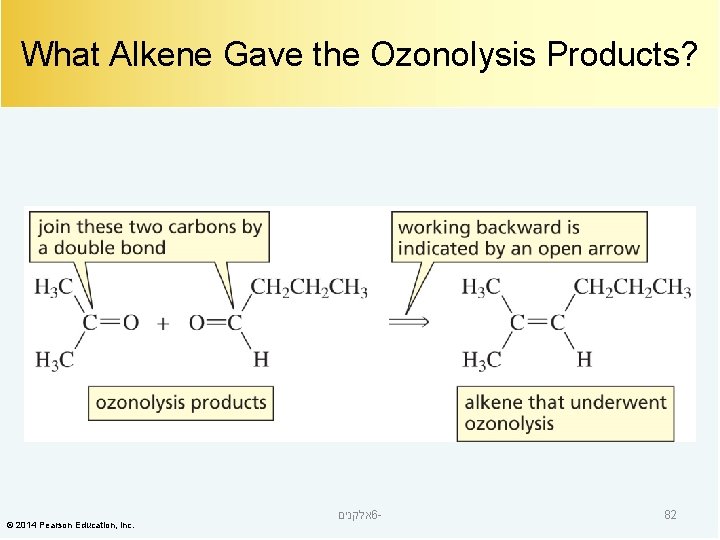
What Alkene Gave the Ozonolysis Products? © 2014 Pearson Education, Inc. אלקנים 6 - 82
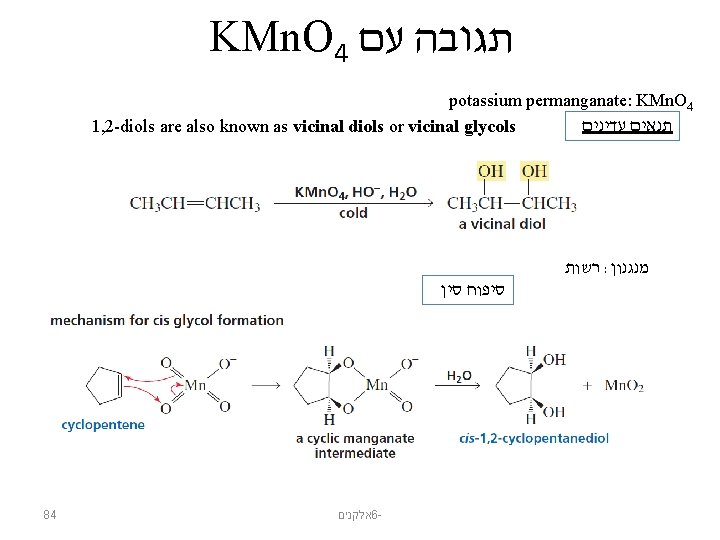
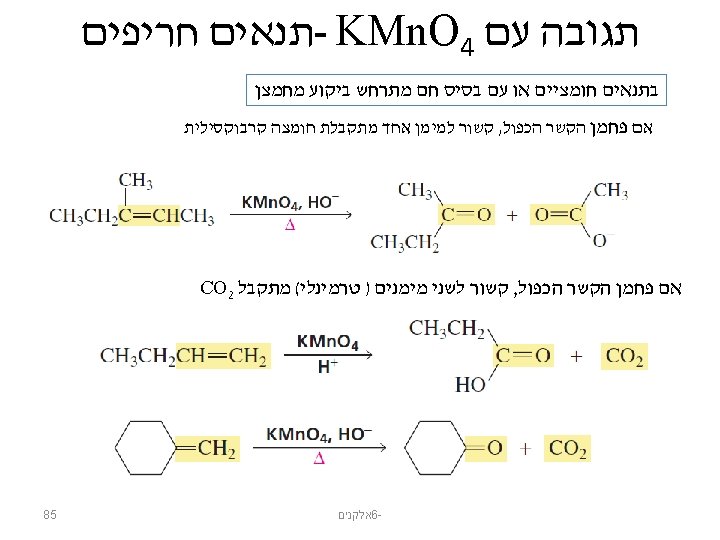
KMn. O 4 תגובה עם potassium permanganate: KMn. O 4 תנאים עדינים 1, 2 -diols are also known as vicinal diols or vicinal glycols רשות : מנגנון סיפוח סין 84 אלקנים 6 -
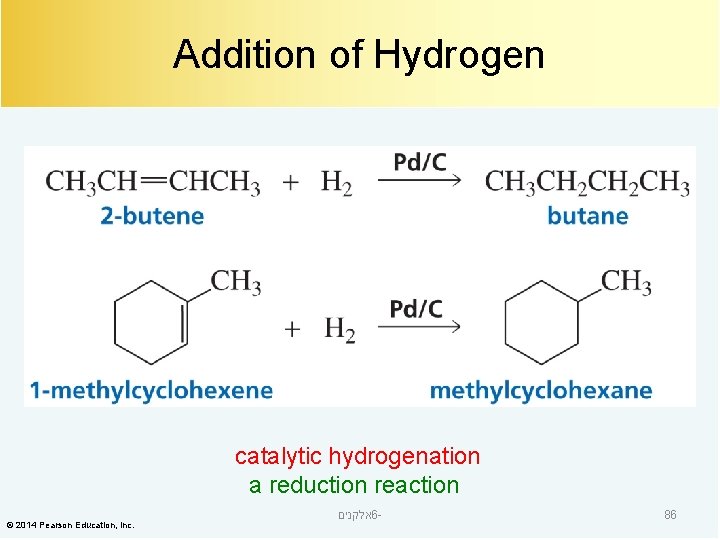
Addition of Hydrogen catalytic hydrogenation a reduction reaction © 2014 Pearson Education, Inc. אלקנים 6 - 86
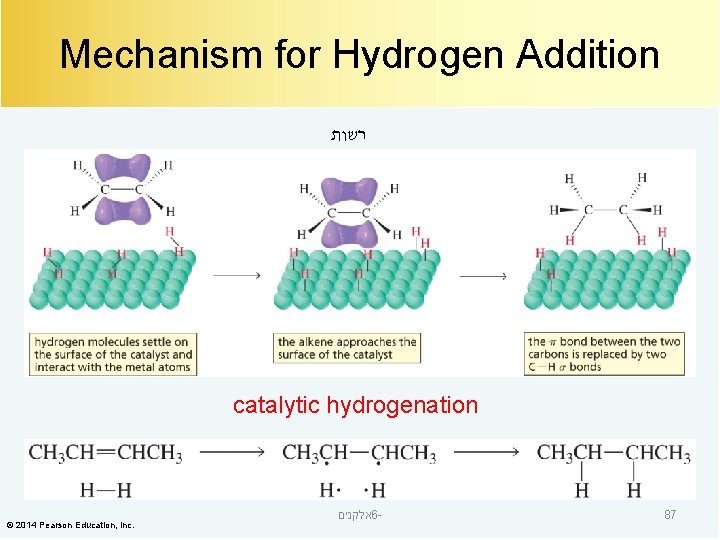
Mechanism for Hydrogen Addition רשות catalytic hydrogenation © 2014 Pearson Education, Inc. אלקנים 6 - 87
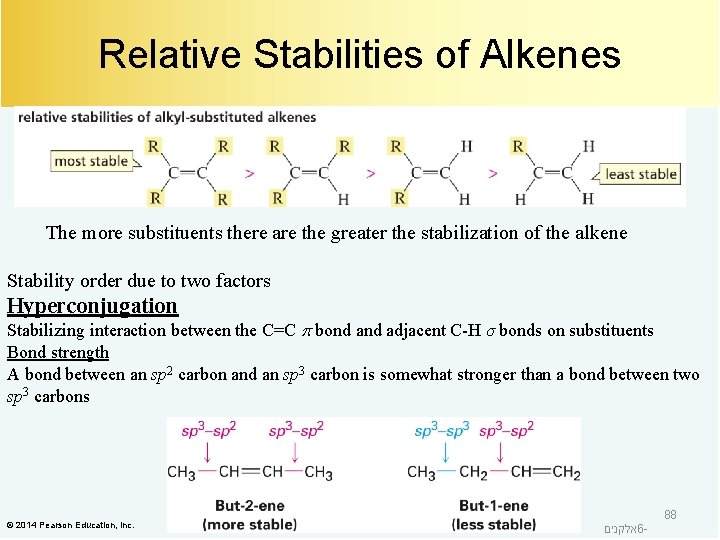
Relative Stabilities of Alkenes The more substituents there are the greater the stabilization of the alkene Stability order due to two factors Hyperconjugation Stabilizing interaction between the C=C bond adjacent C-H σ bonds on substituents Bond strength A bond between an sp 2 carbon and an sp 3 carbon is somewhat stronger than a bond between two sp 3 carbons © 2014 Pearson Education, Inc. אלקנים 6 - 88
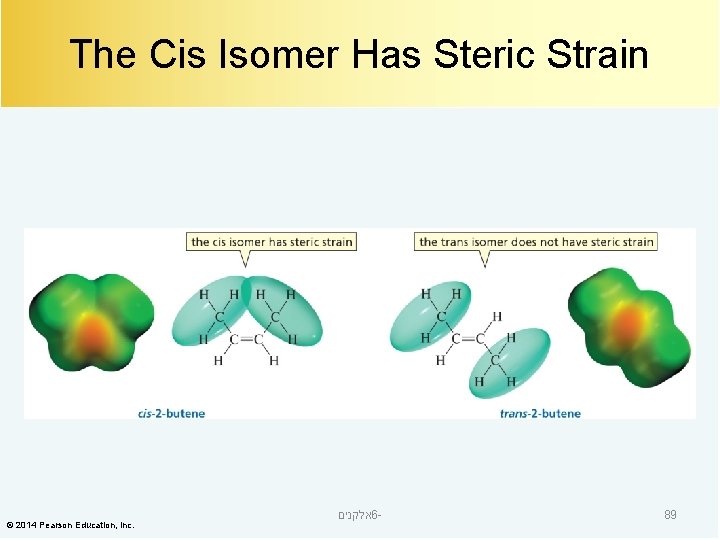
The Cis Isomer Has Steric Strain © 2014 Pearson Education, Inc. אלקנים 6 - 89
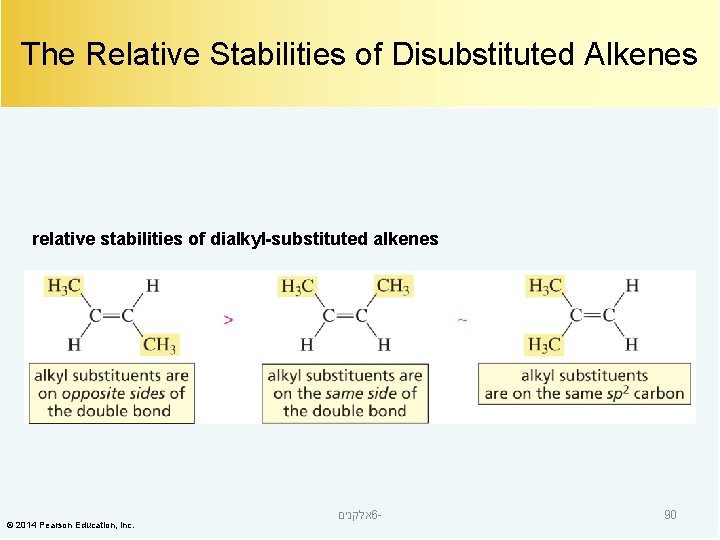
The Relative Stabilities of Disubstituted Alkenes relative stabilities of dialkyl-substituted alkenes © 2014 Pearson Education, Inc. אלקנים 6 - 90

The Product Does Not Have Stereoisomers © 2014 Pearson Education, Inc. אלקנים 6 - 91

The Product is a Racemic Mixture When a reactant that does not have an asymmetric center forms a product that has one asymmetric center, the product is a racemic mixture. © 2014 Pearson Education, Inc. אלקנים 6 - 92
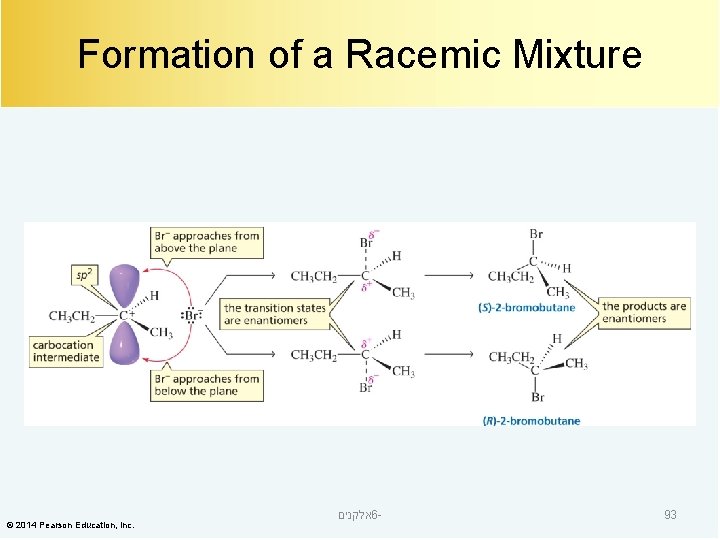
Formation of a Racemic Mixture © 2014 Pearson Education, Inc. אלקנים 6 - 93
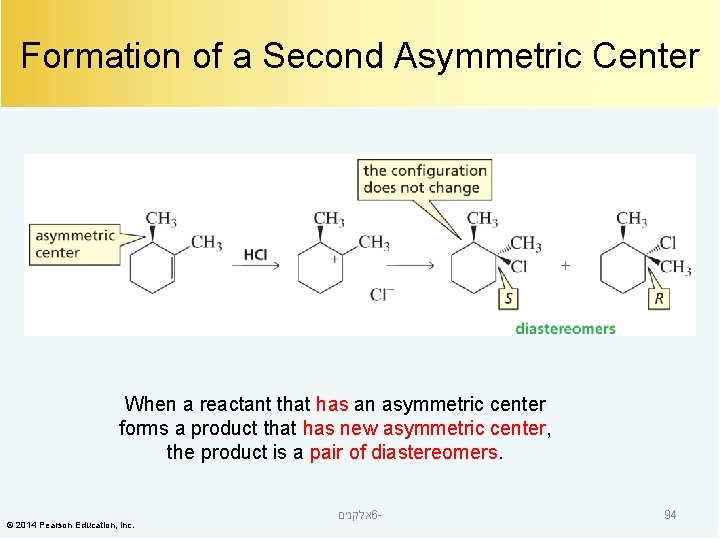
Formation of a Second Asymmetric Center When a reactant that has an asymmetric center forms a product that has new asymmetric center, the product is a pair of diastereomers. © 2014 Pearson Education, Inc. אלקנים 6 - 94
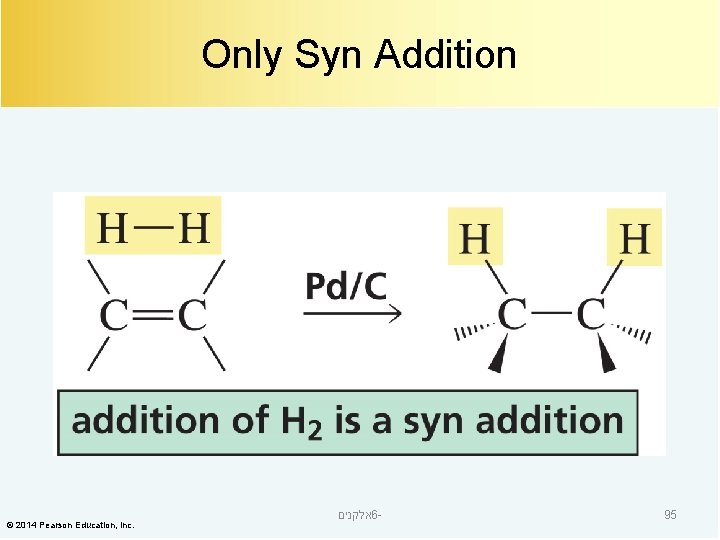
Only Syn Addition © 2014 Pearson Education, Inc. אלקנים 6 - 95
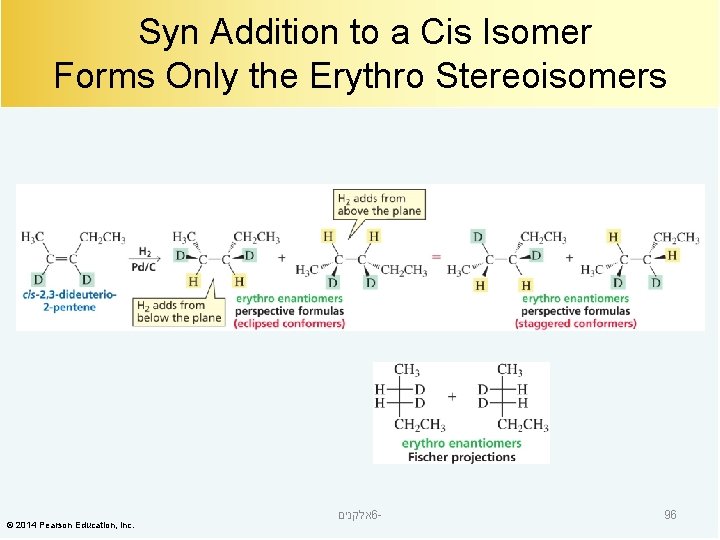
Syn Addition to a Cis Isomer Forms Only the Erythro Stereoisomers © 2014 Pearson Education, Inc. אלקנים 6 - 96
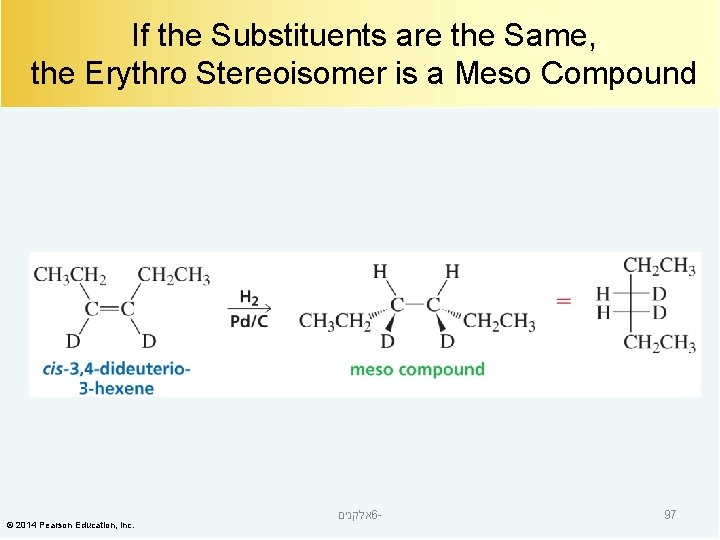
If the Substituents are the Same, the Erythro Stereoisomer is a Meso Compound © 2014 Pearson Education, Inc. אלקנים 6 - 97
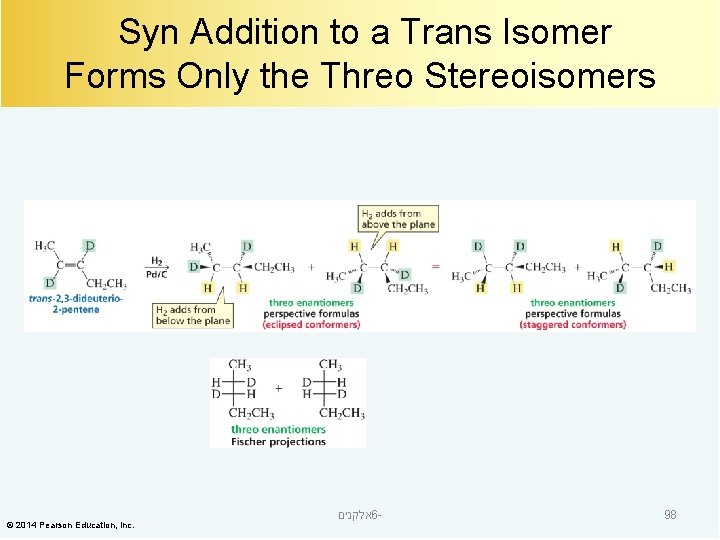
Syn Addition to a Trans Isomer Forms Only the Threo Stereoisomers © 2014 Pearson Education, Inc. אלקנים 6 - 98
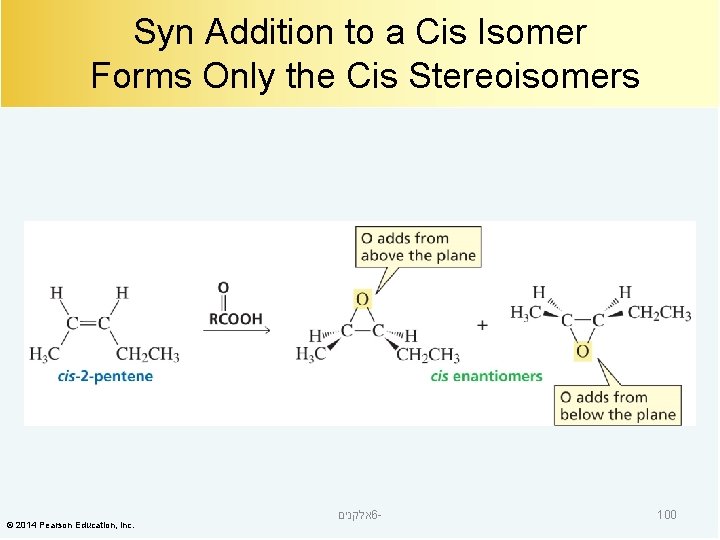
Syn Addition to a Cis Isomer Forms Only the Cis Stereoisomers © 2014 Pearson Education, Inc. אלקנים 6 - 100
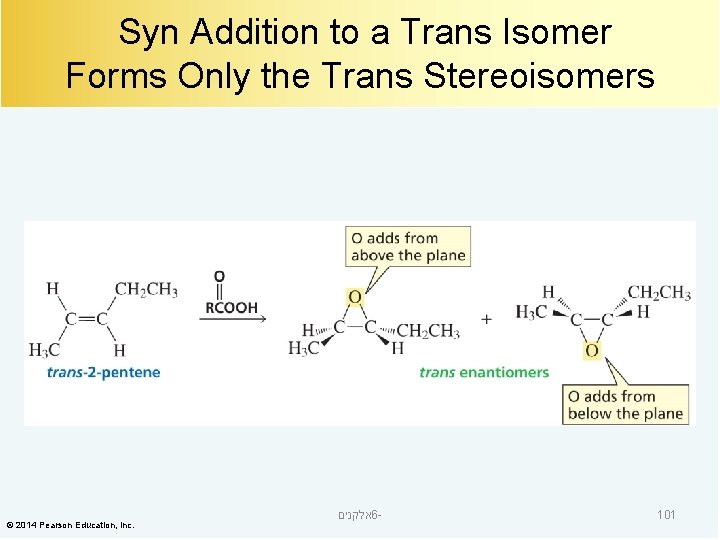
Syn Addition to a Trans Isomer Forms Only the Trans Stereoisomers © 2014 Pearson Education, Inc. אלקנים 6 - 101
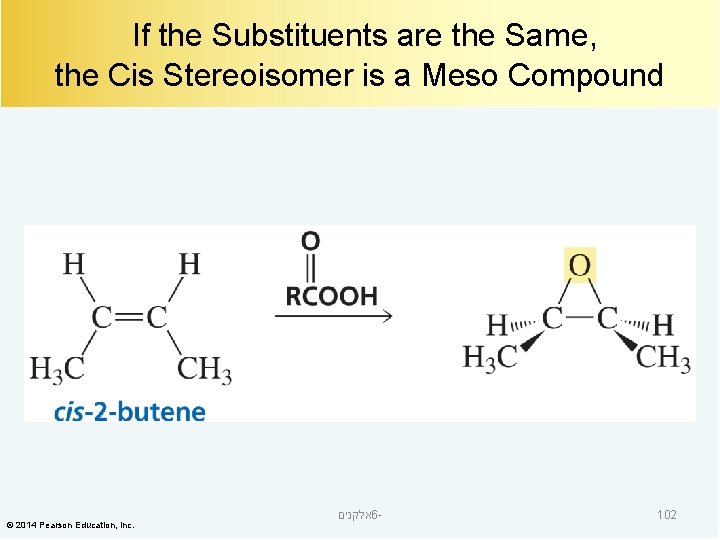
If the Substituents are the Same, the Cis Stereoisomer is a Meso Compound © 2014 Pearson Education, Inc. אלקנים 6 - 102
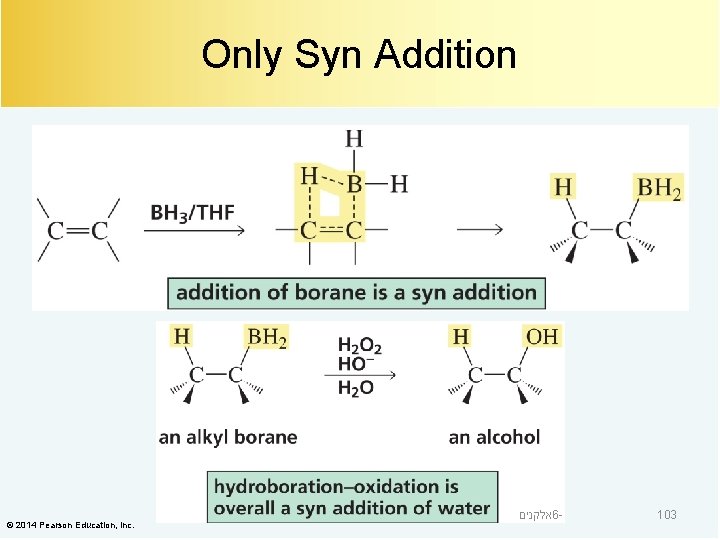
Only Syn Addition © 2014 Pearson Education, Inc. אלקנים 6 - 103
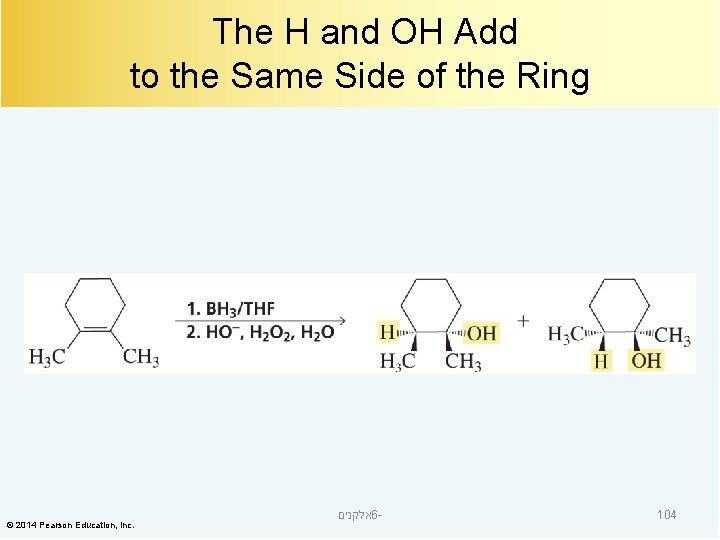
The H and OH Add to the Same Side of the Ring © 2014 Pearson Education, Inc. אלקנים 6 - 104
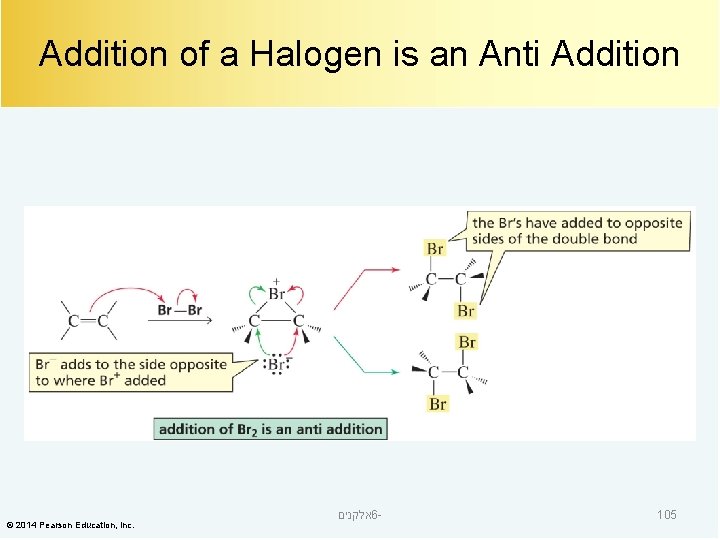
Addition of a Halogen is an Anti Addition © 2014 Pearson Education, Inc. אלקנים 6 - 105
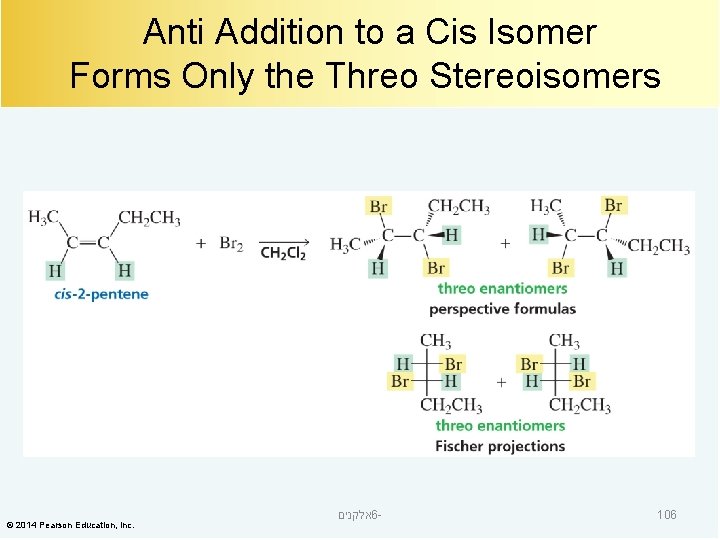
Anti Addition to a Cis Isomer Forms Only the Threo Stereoisomers © 2014 Pearson Education, Inc. אלקנים 6 - 106
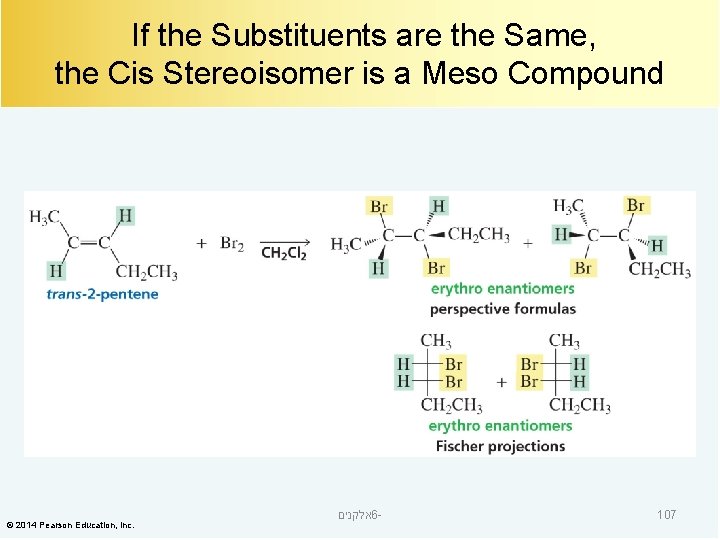
If the Substituents are the Same, the Cis Stereoisomer is a Meso Compound © 2014 Pearson Education, Inc. אלקנים 6 - 107
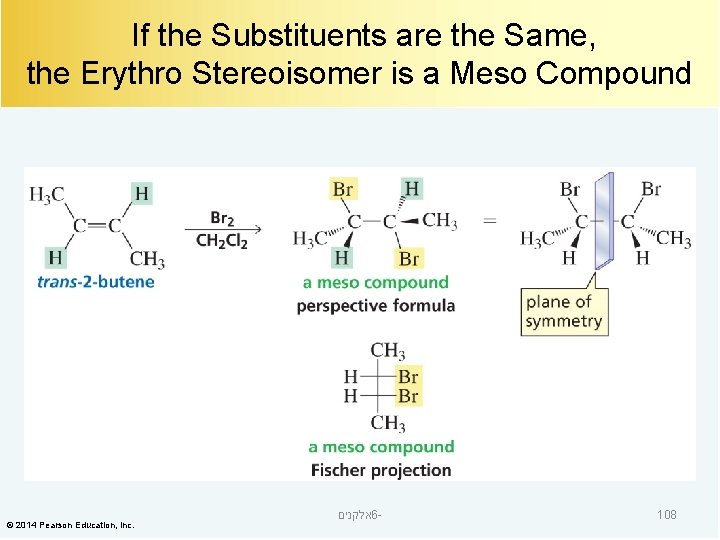
If the Substituents are the Same, the Erythro Stereoisomer is a Meso Compound © 2014 Pearson Education, Inc. אלקנים 6 - 108
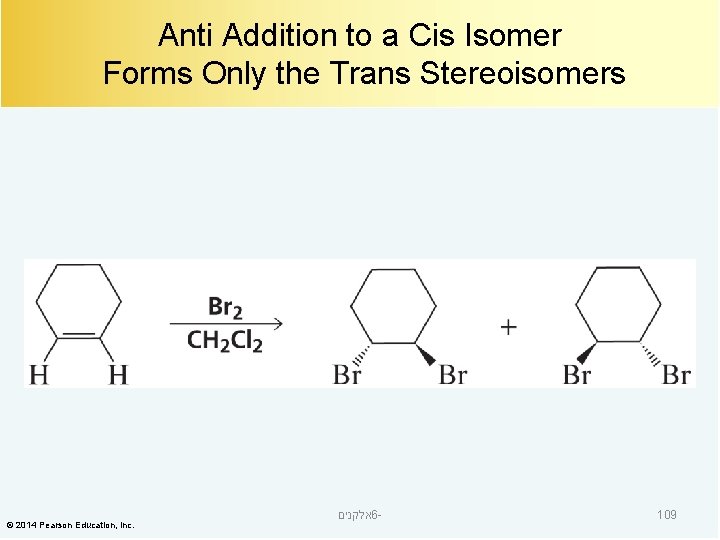
Anti Addition to a Cis Isomer Forms Only the Trans Stereoisomers © 2014 Pearson Education, Inc. אלקנים 6 - 109
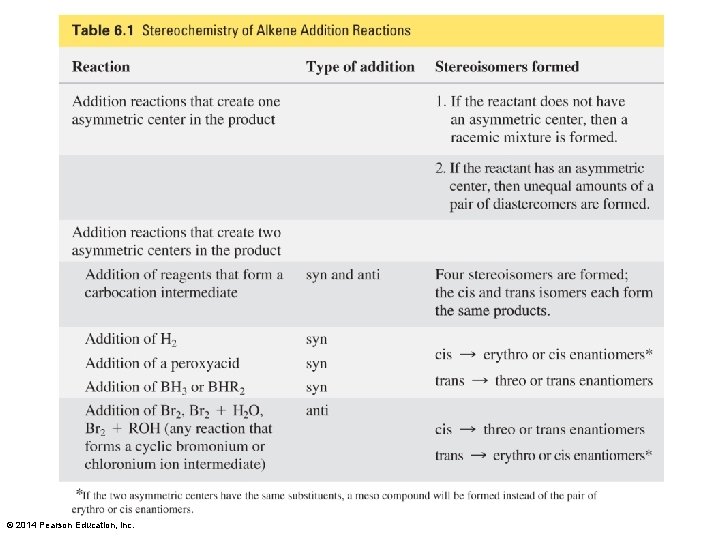
© 2014 Pearson Education, Inc.
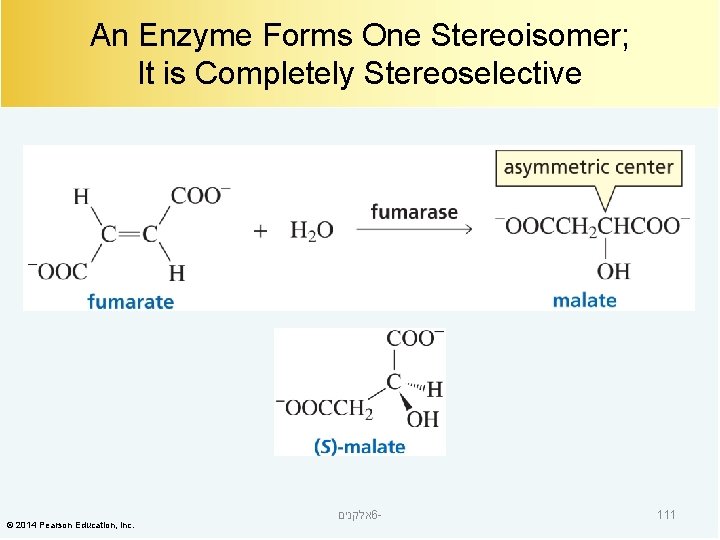
An Enzyme Forms One Stereoisomer; It is Completely Stereoselective © 2014 Pearson Education, Inc. אלקנים 6 - 111
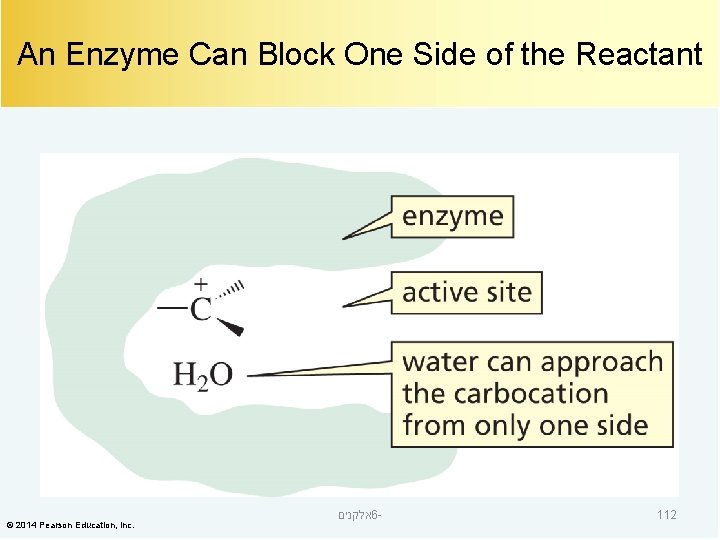
An Enzyme Can Block One Side of the Reactant © 2014 Pearson Education, Inc. אלקנים 6 - 112
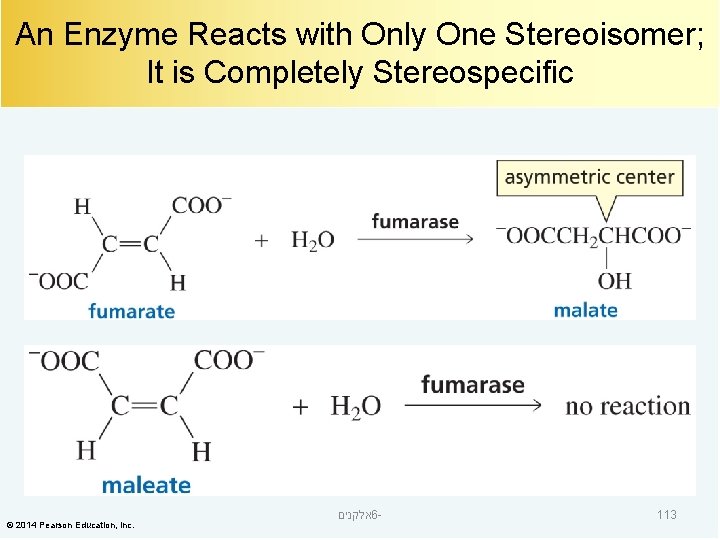
An Enzyme Reacts with Only One Stereoisomer; It is Completely Stereospecific © 2014 Pearson Education, Inc. אלקנים 6 - 113
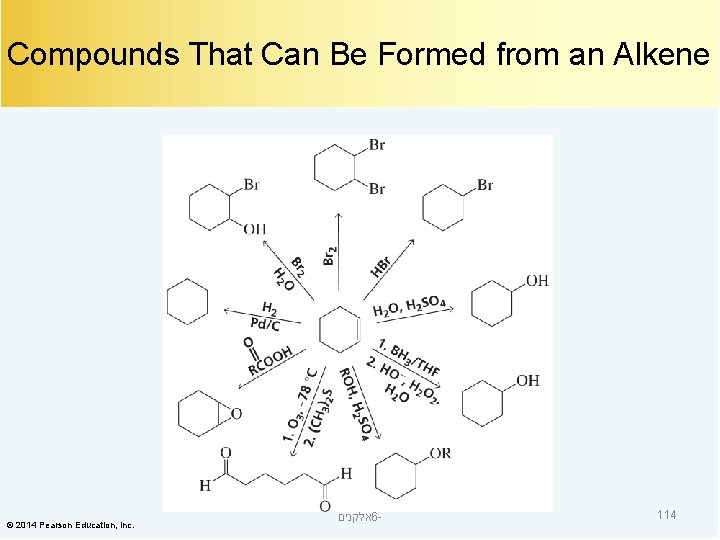
Compounds That Can Be Formed from an Alkene © 2014 Pearson Education, Inc. אלקנים 6 - 114
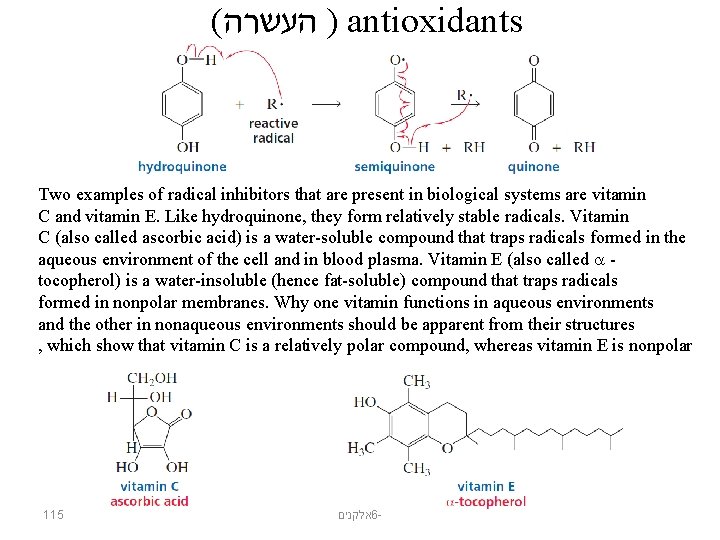
( ) העשרה antioxidants Two examples of radical inhibitors that are present in biological systems are vitamin C and vitamin E. Like hydroquinone, they form relatively stable radicals. Vitamin C (also called ascorbic acid) is a water-soluble compound that traps radicals formed in the aqueous environment of the cell and in blood plasma. Vitamin E (also called tocopherol) is a water-insoluble (hence fat-soluble) compound that traps radicals formed in nonpolar membranes. Why one vitamin functions in aqueous environments and the other in nonaqueous environments should be apparent from their structures , which show that vitamin C is a relatively polar compound, whereas vitamin E is nonpolar 115 אלקנים 6 -
- Slides: 115