SASB Titration Initial p H pts A and
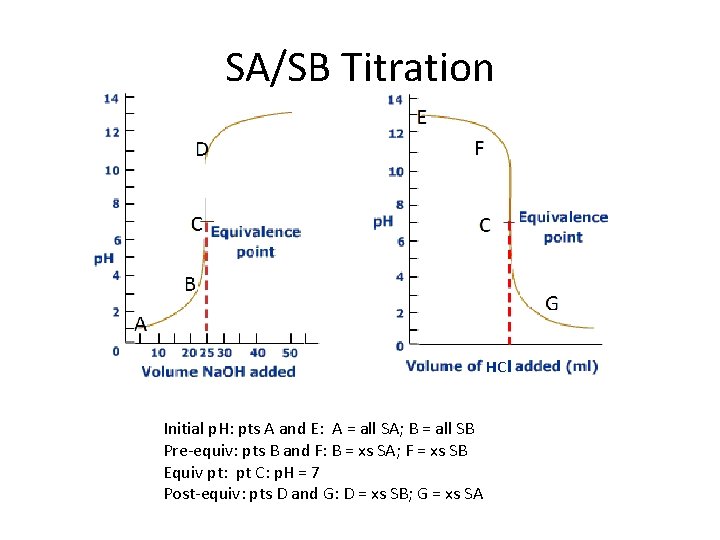
SA/SB Titration Initial p. H: pts A and E: A = all SA; B = all SB Pre-equiv: pts B and F: B = xs SA; F = xs SB Equiv pt: pt C: p. H = 7 Post-equiv: pts D and G: D = xs SB; G = xs SA

WA/SB Titration Initial p. H: pts A and E: A = all WA; B = all WB (ICE box) Pre-equiv: pts B and F: buffer (modified ICE) Equiv pt: pts C and G: switcheroo (use ICE for conjugate) Post-equiv: pts D and G: D = xs SB; G = xs SA

WA/SB Problem Titration of 40. 0 m. L of 0. 100 M HPr (propanoic acid, CH 3 CH 2 CO 2 H) with 0. 100 M Na. OH. Ka HPr = 1. 3 x 10 -5 • How many m. L Na. OH to reach equiv pt? • Calc initial p. H.

p. H for pre-equiv pt • Calc p. H when add 10. 0 m. L of Na. OH.
p. H at equiv pt = “switcheroo” • Calc p. H at equiv pt.

p. H post-equiv pt • Calc p. H when add 60. 0 m. L of 0. 100 M Na. OH.

Additional Problem Calculate p. H for the titration of 25. 0 m. L of 0. 100 M ethylamine (CH 3 CH 2 NH 2) with 0. 05 M HCl. Kb for ethylamine = 6. 4 x 10 -4 1. Identify the analyte and the titrant. 2. How many m. L of HCl must be added to reach equiv pt? 3. Calc initial p. H. 4. Calc p. H if add 5. 00 m. L of HCl. 5. Calc p. H at equiv pt. 6. Calc p. H if add 60. 0 m. L of HCl.

Solubility Equilibria • Looks at equil between (s) and ions (salts dissolved in water at saturation point) Ex. Na. Cl (s) Na+ (aq) + Cl- (aq) – When put salt in water, it dissolves. – As ions build up, they can reform the salt. – At some point, establish eq = saturation point. • Since we have equil, we can write a K expression. Ksp (solubility product constant) Ksp = [Na+][Cl-]

Ksp • Ksp tells about how soluble or not a salt is-explain • Solubility is the same as [ ] so for Na. Cl, Na. Cl (s) Na+ (aq) + Cl- (aq) if 1 M, then … if S, then … Ksp= [Na+][Cl-] = (S)(S) = S 2 Solubility, S = √Ksp = M units

1. If Ag. Br (s) has Ksp = 1. 6 x 10 -10 at 25 o. C, what is solubility in M? 2. What does that # mean in words?

• Write Ksp expression for magnesium bromide (s) dissolving in water in terms of [ ] and S. • Calculate Ksp if, at 25 o. C, S = 0. 0159 M.
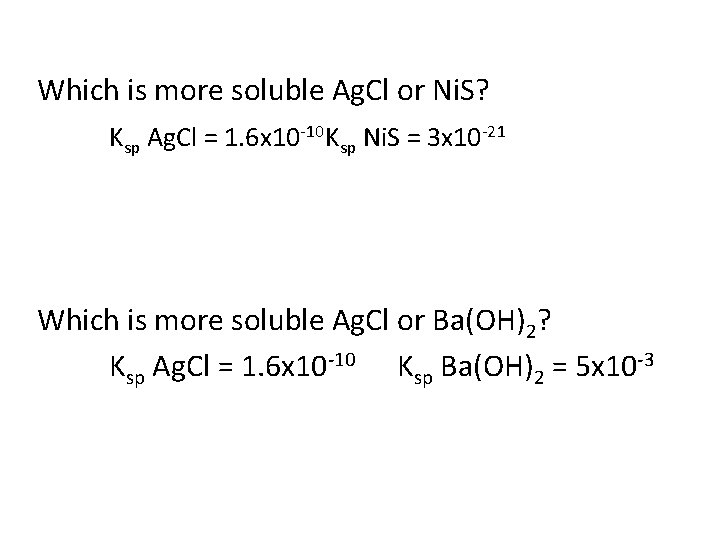
Which is more soluble Ag. Cl or Ni. S? Ksp Ag. Cl = 1. 6 x 10 -10 Ksp Ni. S = 3 x 10 -21 Which is more soluble Ag. Cl or Ba(OH)2? Ksp Ag. Cl = 1. 6 x 10 -10 Ksp Ba(OH)2 = 5 x 10 -3
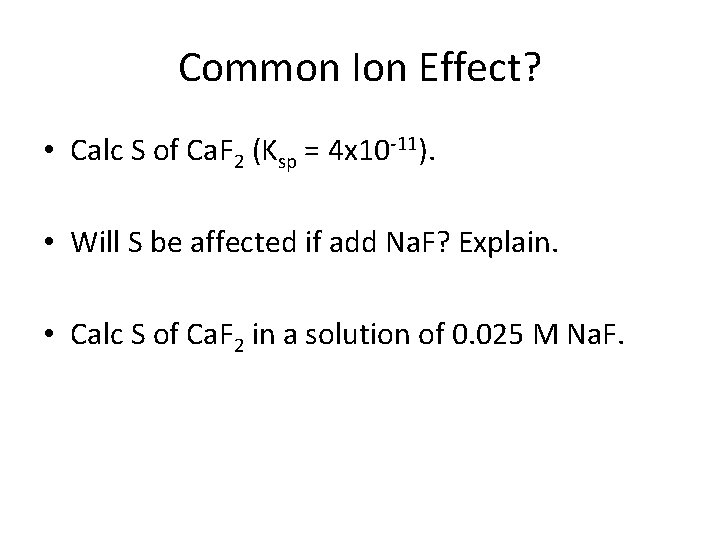
Common Ion Effect? • Calc S of Ca. F 2 (Ksp = 4 x 10 -11). • Will S be affected if add Na. F? Explain. • Calc S of Ca. F 2 in a solution of 0. 025 M Na. F.

Does p. H affect solubility? • Ba(OH)2 (s) Ba 2+ (aq) + 2 OH- (aq) If add HCl (↓p. H), would solubility change? What about adding Na. OH?
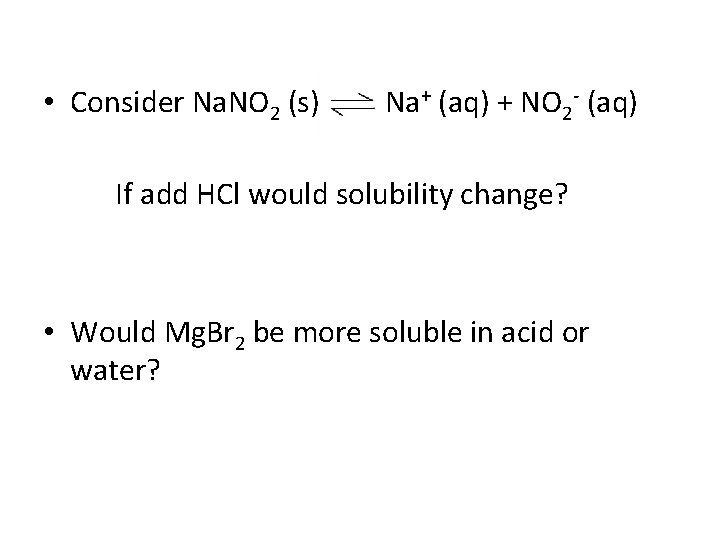
• Consider Na. NO 2 (s) Na+ (aq) + NO 2 - (aq) If add HCl would solubility change? • Would Mg. Br 2 be more soluble in acid or water?

Will ppt form? • Compare Qsp to Ksp – If Qsp > Ksp, ppt will form – If Qsp < Ksp, no ppt • Will ppt form when 100. m. L of 4 x 10 -4 M Mg(NO 3)2 is mixed with 100. m. L of 2 x 10 -4 M Na. OH? Ksp Mg(OH)2 = 1. 8 x 10 -11
- Slides: 16