Sargus HISTORY OF THE ATOM 460 BC Democritus

Sargus

HISTORY OF THE ATOM 460 BC Democritus develops the idea of atoms he pounded up materials in his pestle and mortar until he had reduced them to smaller and smaller particles which he called ATOMA (greek for indivisible)

Antoine Lavoisier 1743 -1794 • Law of Conservation of Mass • Law of Definite Proportions • Law of Multiple Proportions

HISTORY OF THE ATOM 1808 John Dalton suggested that all matter was made up of tiny spheres that were able to bounce around with perfect elasticity and called them ATOMS

Dalton’s Postulates • All matter is composed of atoms • The atoms of a given element are all identical* and different from atoms of another element • Atoms cannot be created, divided, or destroyed by chemical processes. • Chemical reactions change the way atoms are combined

Humphrey Davy 1778 -1829 • Developed electrolysis where a battery was used to apply an electric field to decompose compounds into elements • Showed that atoms are held together by electrical forces

Faraday and Stoney • Michael Faraday’s work on electricity also showed that electrical charge is associated with chemical bonds • In 1874 George Stoney determines that discrete units of electrical charge are associated with atoms, and in 1891 he coins the term “electrons”.

HISTORY OF THE ATOM 1898 Joseph John Thompson Performs various experiments in which he manipulates a cathode ray tube to demonstrate that: -cathode rays are made up of electrons -electrons have mass -electrons have a negative charge. He determined the charge to mass ratio of electrons http: //g. web. umkc. edu/gounevt/Animations 211 /Cathode. Ray. Tube. swf

HISTORY OF THE ATOM 1904 Thompson develops the idea that an atom was made up of electrons scattered unevenly within an elastic sphere surrounded by a soup of positive charge to balance the electron's charge like plums surrounded by pudding. PLUM PUDDING MODEL

Robert Millikan 1909 • Performs the famous “Millikan oil drop experiment” in which he experimentally determined the charge of an electron. 1. 602 × 10− 19 coulomb • Using Thompson’s charge to mass ratio allows for the determination of an electron’s mass. • http: //www. georgiacenter. uga. edu/idl/webid/portfolio/flash/an imation/milikan. html

Cautionary tale in science R. Feynman • We have learned a lot from experience about how to handle some of the ways we fool ourselves. One example: Millikan measured the charge on an electron by an experiment with falling oil drops, and got an answer which we now know not to be quite right. It's a little bit off because he had the incorrect value for the viscosity of air. It's interesting to look at the history of measurements of the charge of an electron, after Millikan. If you plot them as a function of time, you find that one is a little bit bigger than Millikan's, and the next one's a little bit bigger than that, until finally they settle down to a number which is higher. • Why didn't they discover the new number was higher right away? It's a thing that scientists are ashamed of - this history - because it's apparent that people did things like this: When they got a number that was too high above Millikan's, they thought something must be wrong - and they would look for and find a reason why something might be wrong. When they got a number close to Millikan's value they didn't look so hard. And so they eliminated the numbers that were too far off, and did other things like that. We've learned those tricks nowadays, and now we don't have that kind of a disease.

HISTORY OF THE ATOM 1910 Ernest Rutherford oversaw Geiger and Marsden carrying out his famous experiment. they fired Helium nuclei at a piece of gold foil which was only a few atoms thick. they found that although most of them passed through. About 1 in 10, 000 hit
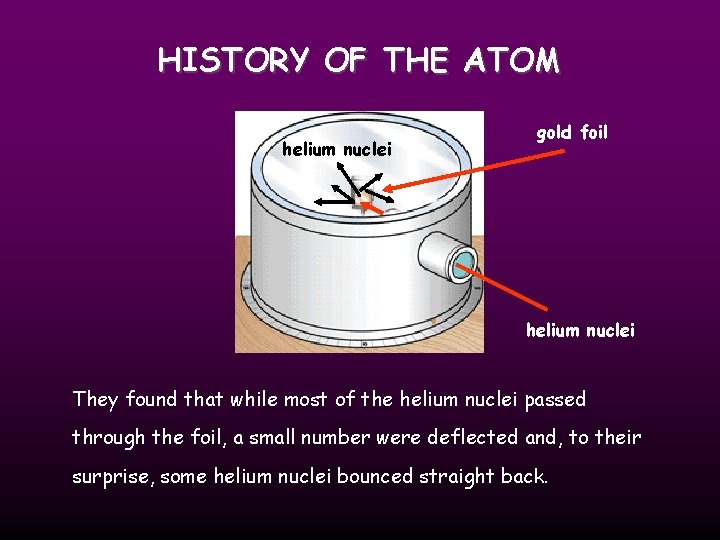
HISTORY OF THE ATOM helium nuclei gold foil helium nuclei They found that while most of the helium nuclei passed through the foil, a small number were deflected and, to their surprise, some helium nuclei bounced straight back.

HISTORY OF THE ATOM Rutherford’s new evidence allowed him to propose a more detailed model with a central nucleus. He suggested that the positive charge was all in a central nucleus. With this holding the electrons in place by electrical attraction However, this was not the end of the story. http: //micro. magnet. fsu. edu/electromag/java/rutherford/ http: //www. mhhe. com/physsci/chemistry/essentialchemistry/flash/ruther 14. swf

HISTORY OF THE ATOM 1913 Niels Bohr studied under Rutherford at the Victoria University in Manchester. Bohr refined Rutherford's idea by adding that the electrons were in orbits. Rather like planets orbiting the sun. With each orbit only able to contain a set number of electrons. His attempts in explaining the atomic emission spectrum of hydrogen led to the refutation of the idea of orbits and the development of the quantum model of the atom. Bohr theorized that electrons occupy quantized energy levels within the atom.

Bohr’s Atom electrons in orbits nucleus
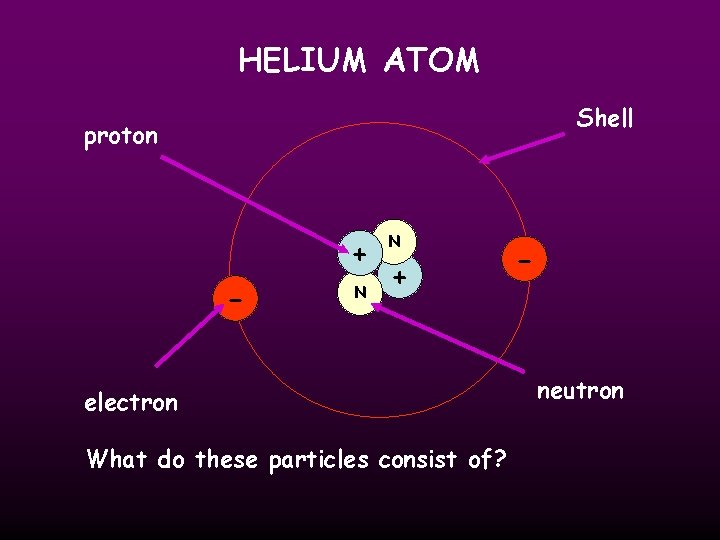
HELIUM ATOM Shell proton + - N N + electron What do these particles consist of? - neutron

ATOMIC STRUCTURE Particle Charge Mass proton + ve charge 1 neutron No charge 1 electron -ve charge nil
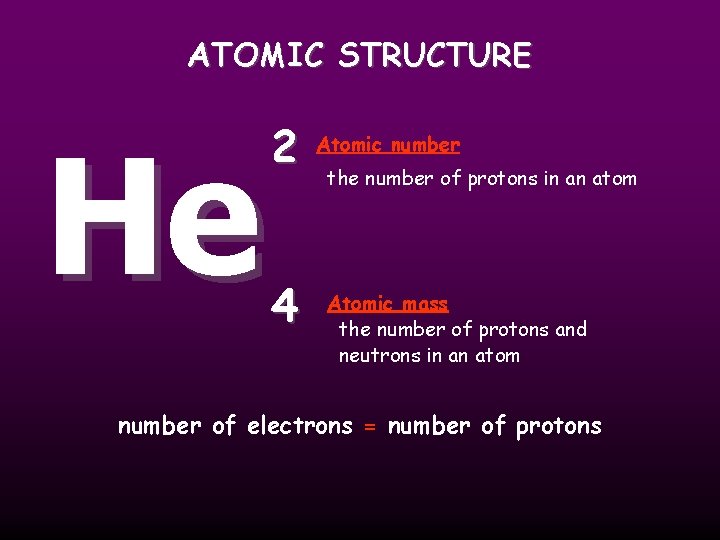
ATOMIC STRUCTURE He 2 4 Atomic number the number of protons in an atom Atomic mass the number of protons and neutrons in an atom number of electrons = number of protons
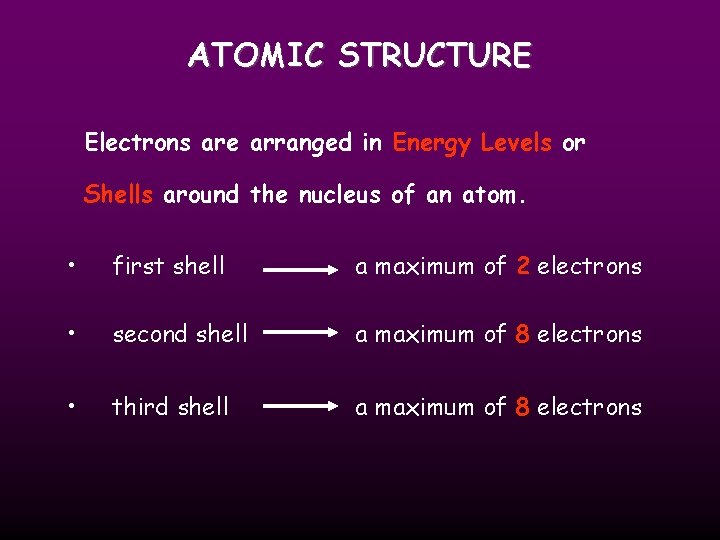
ATOMIC STRUCTURE Electrons are arranged in Energy Levels or Shells around the nucleus of an atom. • first shell a maximum of 2 electrons • second shell a maximum of 8 electrons • third shell a maximum of 8 electrons

ATOMIC STRUCTURE There are two ways to represent the atomic structure of an element or compound; 1. 2. Electronic Configuration Dot & Cross Diagrams

ELECTRONIC CONFIGURATION With electronic configuration elements are represented numerically by the number of electrons in their shells and number of shells. For example; Nitrogen 2 in 1 st shell 5 in 2 nd shell configuration = 2 , 5 2 + 5 = 7 N 7 14

ELECTRONIC CONFIGURATION Write the electronic configuration for the following elements; a) Ca 20 b) Na 40 2, 8, 8, 2 d) Cl 17 35 2, 8, 7 11 23 c) 2, 8, 1 e) Si 14 28 2, 8, 4 O 8 16 2, 6 f) B 5 11 2, 3

DOT & CROSS DIAGRAMS With Dot & Cross diagrams elements and compounds are represented by Dots or Crosses to show electrons, and circles to show the shells. For example; X Nitrogen X X N XX X X N 7 14
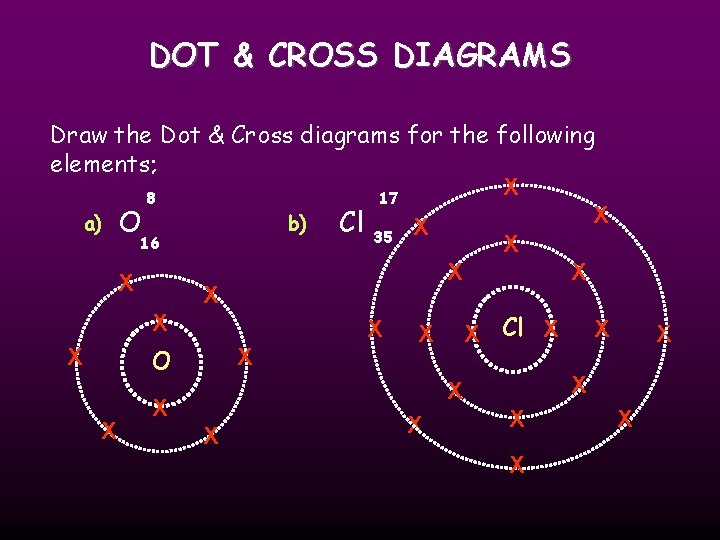
DOT & CROSS DIAGRAMS Draw the Dot & Cross diagrams for the following elements; X 8 17 X a) O b) Cl 35 X 16 X X X X X Cl X X X O X X X X X

SUMMARY 1. The Atomic Number of an atom = number of protons in the nucleus. 2. The Atomic Mass of an atom = number of Protons + Neutrons in the nucleus. 3. The number of Protons = Number of Electrons. 4. Electrons orbit the nucleus in shells. 5. Each shell can only carry a set number of electrons.
- Slides: 26