Sanitary and Phytosanitary Meaures SPS and Technical Barriers

Sanitary and Phytosanitary Meaures (SPS) and Technical Barriers to Trade (TBT) 1

History: GATT 1947 • Food safety, animal and plant health measures were subject to GATT rules: – most-favoured nation – national treatment • Exception (Article XX: b) – measures necessary to protect human, animal or plant life of health 2

History: TBT and SPS • Tokyo Round (1970 s): “Standards Code” • Uruguay Round (1986 -94) – Agreement on Agriculture – SPS Agreement – New TBT Agreement 3
SPS and TBT SPS 4

How to tell what is SPS or TBT? First: • check if it is an SPS measure 5

Definition of an SPS Measure to protect: human or animal life human life animal or plant life a country from: risks arising from additives, contaminants, toxins or disease-causing organisms in their food, beverages, feedstuffs; plant- or animal-carried diseases (zoonoses); pests, diseases, or disease-causing organisms; damage caused by the entry, establishment or spread of pests 6

SPS: An important footnote • “animal” includes wild fauna, fish • “plant” includes forests, wild flora • “pests” include weeds • “contaminants” include pesticides, veterinary drug residues, extraneous matter 7

What type of measures? All measures with these SPS purposes, including: • product criteria • quarantine measures • processing requirements • certification • inspection • testing • health-related labelling 8

Measures intended to protect, for example • the environment, per se • consumer interests (not health) • animal welfare NOT covered by the SPS Agreement 9

SPS or TBT? SPS measures • protect human or animal health from food-borne risks • protect human health from animal- or plant-carried diseases • protect animals and plants from pests or diseases • examples: – microbiological contamination of food – pesticide or veterinary drug residues – food additives TBT measures • • human disease control nutritional claims food packaging and quality examples: – pharmaceuticals – labelling (unless related to food safety) – pesticide handling or quality 10

Example 1: SPS or TBT? Bottled water • safety for human consumption • bottle size and shape • bottle material – breaking – toxic substances contaminating water 11

Example 2: SPS or TBT? • Pesticides – safe handling instructions – quality – residue limits in food 12

Why does it matter which Agreement applies? Different rules apply to SPS and TBT measures 13

The TBT Agreement covers. . . • Standards • Technical Regulations • Conformity Assessment Procedures 14
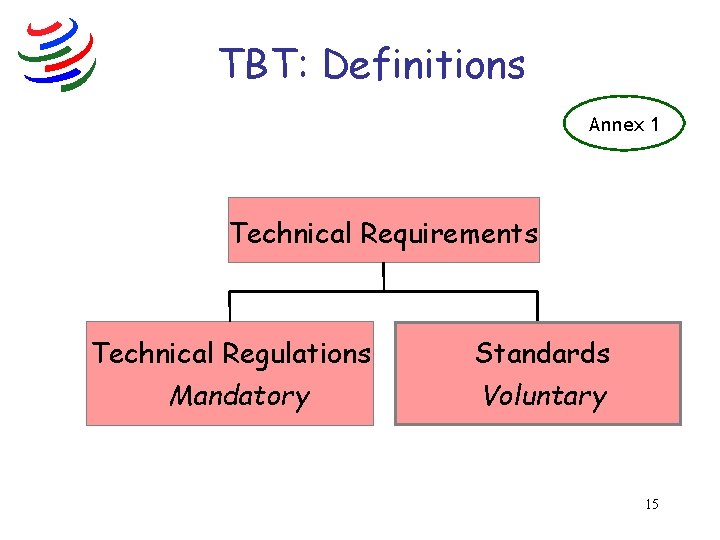
TBT: Definitions Annex 1 Technical Requirements Technical Regulations Mandatory Standards Voluntary 15

What is conformity assessment? • Measures taken by manufacturers, their customers, regulatory authorities, and independent third parties to assess compliance with standards 16

TBT: Basis • WTO Members have the right to adopt technical regulations, standards and conformity assessment procedures, • BUT: they must not constitute unnecessary obstacles to trade 17

SPS: Basis • Members have the right to take sanitary and phytosanitary measures necessary for the protection of human, animal and plant life or health… • BUT: - no unjustifiable discrimination – no disguised restrictions on international trade 18

SPS & TBT: main provisions SPS TBT • • • scientific justification harmonization least trade-restrictive equivalence regional conditions transparency technical assistance, special and differential treatment legitimate objective harmonization least trade-restrictive equivalence and mutual recognition • transparency • technical assistance, special and differential treatment 19

SPS: Scientific Justification Article 2. 2 • “applied only to the extent necessary to protect life or health” • “based on scientific principles” • “not maintained without sufficient scientific evidence” • “except… paragraph 7 of Article 5” 20

SPS: Provisional Measures Article 5. 7 Members may provisionally adopt measures. . . • if relevant scientific evidence is insufficient • on the basis of available information But: • shall seek to obtain the additional information • review within a reasonable period of time 21

TBT: legitimate objectives • National security • Prevention of deceptive practices • Human health or safety • Animal and plant life or health • Environment Inter alia ! 22

TBT: no unnecessary obstacle to trade (1) • Not more trade restrictive than necessary – risks of non-fulfilment – relevant factors: scientific and technical information, processing technology, intended end-uses of product, risks of non -fulfilment 23

TBT: no unnecessary obstacle to trade (2) Three step analysis 1. Is the policy objective legitimate? 2. Can this objective be achieved with an alternative measure that is less trade-restrictive? 3. What are the risks of non-fulfillment of the originally proposed measure? 24
Consequently, where possible Performance-based preferable to design-based or descriptive product requirements 25

TBT: Notification obligations (1) One time + changes: • Statement on the Implementation and Administration of the Agreement (art. 15. 2) • Designation of a Government Authority for Implementation of Notification Obligations (art. 10 paras. 10 and 11) • Enquiry Point(s): one or more (art. 10 paras. 1 and 2) G/TBT/1/Rev. 10 Notifications circulated as G/TBT/2/Add. # 26

TBT: Notification obligations (2) • Proposed or Adopted Technical Regulations or Conformity Assessment Procedures by Central or Local Governments (art. 2. 9, 2. 10, 3. 2, 5. 6, 5. 7, 7. 2) – ad hoc when: – Relevant international standard does not exist OR the technical content of a proposed technical regulation or conformity assessment procedure is not in accordance with the technical content of relevant international standards, guides or recommendations – Significant effect on trade of other Member(s) Notifications circulated as G/TBT/N/Country/# 27

TBT: Notification obligations (3) • Bilateral or Multilateral Agreements (art. 10. 7) – ad hoc “Whenever a Member has reached an agreement with any other country or countries on issues related to technical regulations, standards or conformity assessment procedures which may have a significant effect on trade, at least one Member party to the agreement shall notify other Members through the Secretariat of the products to be covered by the agreement and include a brief description of the agreement. ” Notifications circulated under G/TBT/10. 7/N/# 28

TBT: Notification obligations (4) Code of Good Practice • Document G/TBT/W/4/rev. 1 – Acceptance of the Code: form A Notifications made to the ISO/IEC Information Center and circulated in WTO as G/TBT/CS/N/# – Withdrawal from the Code: form B – Work programme: form C – Notification every 6 months made to the ISO/IEC Information Center 29

TBT: Enquiry Point - RF • DSTANDARTINFORM 4 Granatny per. , G/TBT/ENQ/38/Rev. 1 Moscow, K-1, GSP-5 123995 Russian Federation Tel. : +7 (495) 225 61 89 Fax: +7 (495) 332 56 59/+7(495) 719 78 20 Email: enpoint@gostinfo. ru 30

SPS: Transparency provisions • Publication of regulations (Annex B) • Notification (Article 7 and Annex B) • Explanation (Article 5. 8) 31

SPS: Notification obligations • National Notification Authority (NNA) – one time + changes • SPS measures – ad hoc • Enquiry Point – one time + changes G/SPS/7/Rev. 3 32
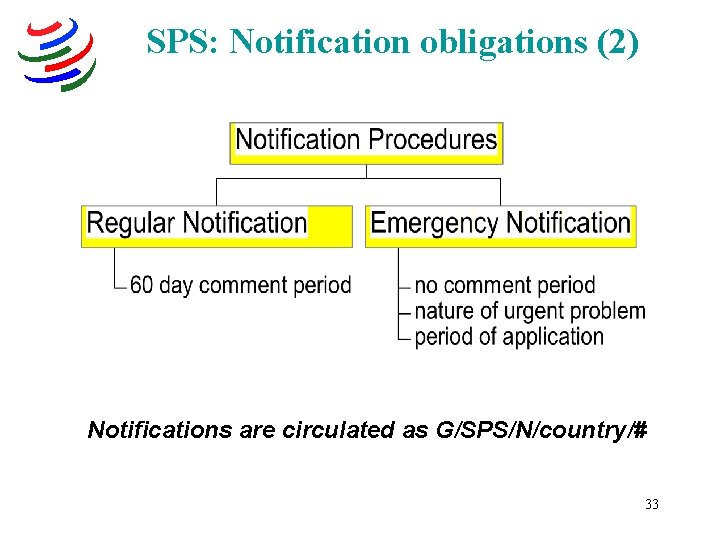
SPS: Notification obligations (2) Notifications are circulated as G/SPS/N/country/# 33

SPS: Enquiry Point - RF Russian Information Center on Standardization, Certification G/SPS/ENQ/26 Contact person: Ms Nina Khromova 4 Granatny per. Moscow 103 001 Telephone: +(7 495) 332 5628/332 5629/225 6189 Telefax: +(7 495) 332 5659 E-mail/Internet: enpoint@gostinfo. ru 34
- Slides: 34