San Francisco Department of Public Health Laboratory HIV

San Francisco Department of Public Health Laboratory: HIV Testing

HIV Testing: Goals of the presentation: 1. General overview, principles 2. Antibody testing 3. Direct Detection of Virus 4. The (Near)-Future of Testing

Part 1: General Principles

Course of early HIV infection Knowing this make it much easier to understand the differences in HIV tests
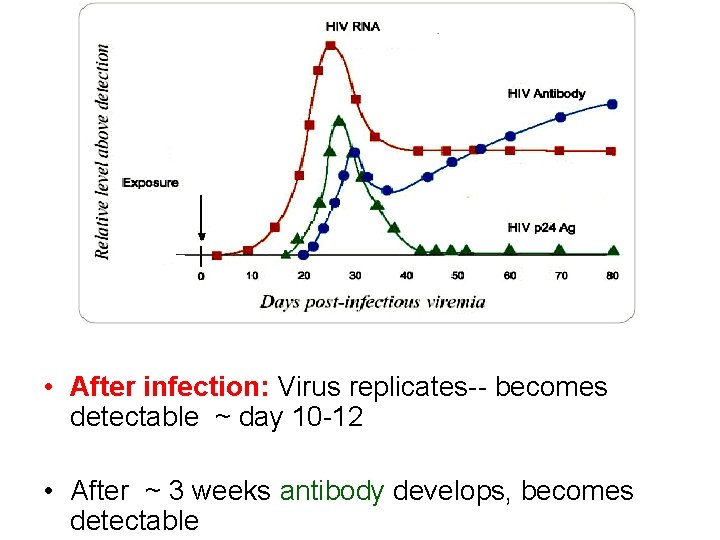
• After infection: Virus replicates-- becomes detectable ~ day 10 -12 • After ~ 3 weeks antibody develops, becomes detectable
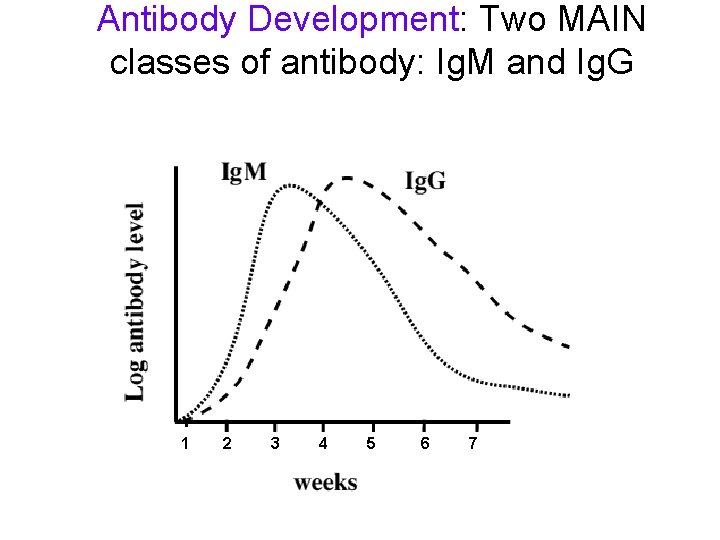
Antibody Development: Two MAIN classes of antibody: Ig. M and Ig. G 1 2 3 4 5 6 7

So, the order of things is: • Infection • Virus growth (RNA and antigen levels rise) • Antibody development -(Ig. M and then Ig. G)

HIV Testing and the “Window Period” • After infection, markers of infection take time to develop: tests will not detect infection during this time • People can be infectious during this window period Virus becomes detectable Infection event Day 0 Virus Detectable Day 11 Antibody becomes detectable Ig. M Detectable Day 22 Ig. G Detectable Day 36

Part 2: HIV Antibody Testing

HIV Testing is still accomplished through an algorithm recommended in 1989 by CDC : • All HIV testing must involve: --an antibody screening test And if the screening test is positive: --a confirmation test is performed The FDA approves certain tests as “screening” and others as “confirmation”

HIV Screening Tests

Screening tests: • Laboratory based antibody tests --called: “immunoassays” or “EIA” for “enzyme immunoassays”) • Point-of-Care, “rapid” tests: examples: Ora. Quick, Stat-Pak

Screening tests 1. Laboratory based 2. Point of Care (Rapid)

Laboratory Screening: peformed by EIA / ELISA: Enzyme Immuno. Assay • The original, long-standing workhorse of HIV testing (1985 - present) • Relatively easy to perform (can automate) • Sensitive • Can process many (~90) specimens at once

An Actual ELISA

HIV EIA: The First Generation Tests • Don’t detect antibody to all HIV-1 strains, or HIV-2 reliably • Only Ig. G detected • had window periods of ~6 weeks • No longer available in marketplace

HIV EIA: Third Generation • Ig. M and Ig. G detection • Detects Group O, Detects HIV-2 antibodies • Shorter Window Periods (22 days)
Point-of-care, “Rapid” HIV testing • Rapid Tests, also known as “point of care” tests are considered “Low Complexity” by CLIA • Tests are routinely done outside of lab; positive results go to a lab for confirmation --Some Labs are using rapid tests as their screening test (SFGH Clin. Lab)

Rapid testing options (in U. S. ):
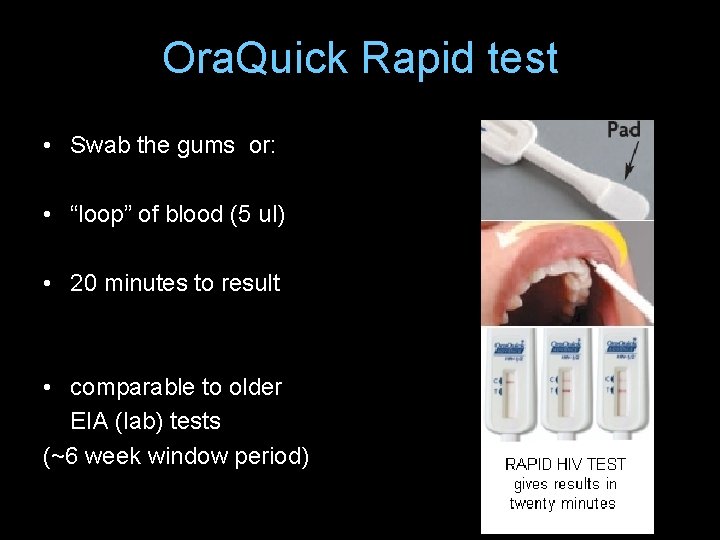
Ora. Quick Rapid test • Swab the gums or: • “loop” of blood (5 ul) • 20 minutes to result • comparable to older EIA (lab) tests (~6 week window period)

Clearview Stat-pak • Similar sensitivity and functionality to Ora. Quick • Can only use blood / plasma
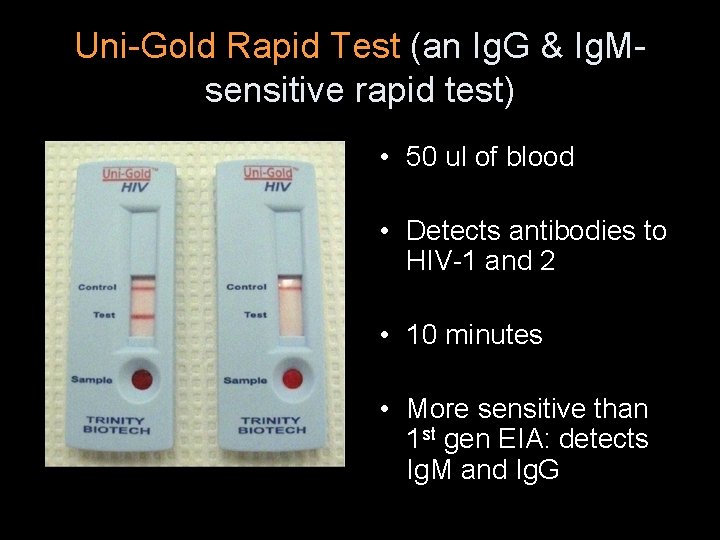
Uni-Gold Rapid Test (an Ig. G & Ig. Msensitive rapid test) • 50 ul of blood • Detects antibodies to HIV-1 and 2 • 10 minutes • More sensitive than 1 st gen EIA: detects Ig. M and Ig. G
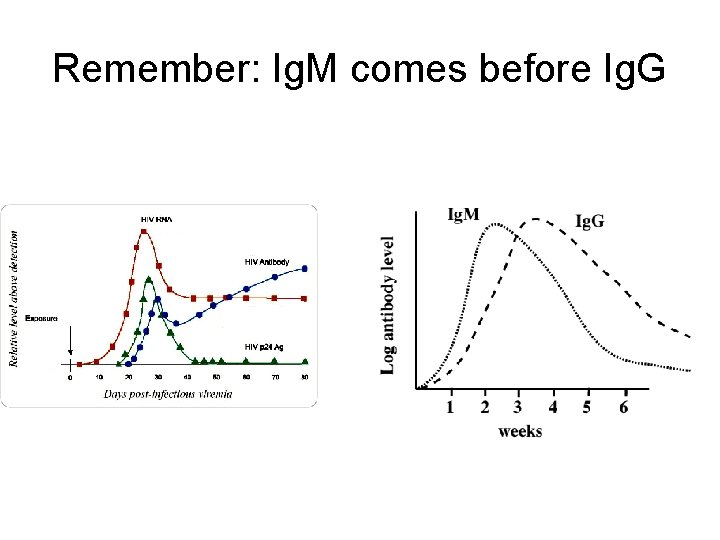
Remember: Ig. M comes before Ig. G

Public Health Lab assessed rapid tests for ability to detect recent infection • Most are about as good as older lab tests • 1 of them (Uni-Gold) that detects Ig. M is notably more sensitive for recent infection

Confirmation Testing
Confirmation Tests • tests that confirm a positive screening test result • Currently, all confirmation is lab-based • Efforts underway to allow for a rapid test to be confirmed by a second rapid, --but it is not yet allowed Immunofluorescence Assay
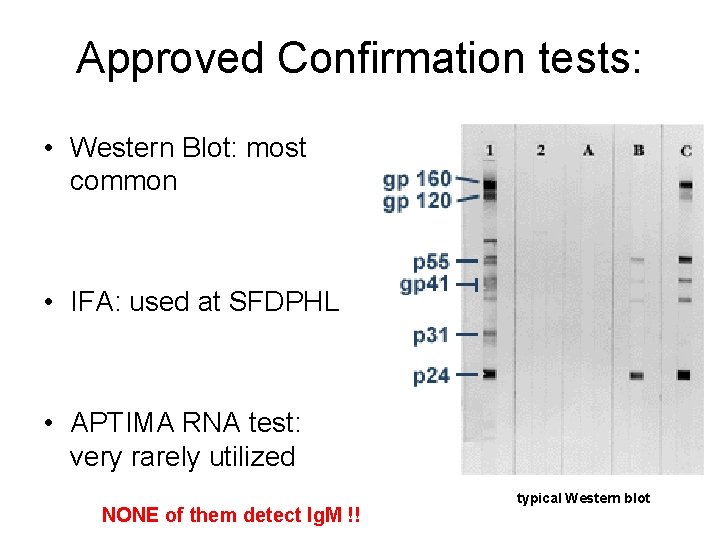
Approved Confirmation tests: • Western Blot: most common • IFA: used at SFDPHL • APTIMA RNA test: very rarely utilized NONE of them detect Ig. M !! typical Western blot
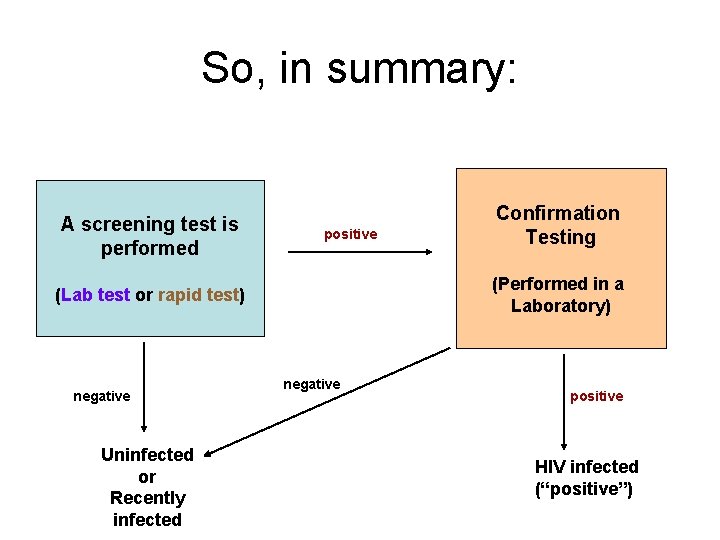
So, in summary: A screening test is performed positive (Performed in a Laboratory) (Lab test or rapid test) negative Uninfected or Recently infected Confirmation Testing negative positive HIV infected (“positive”)
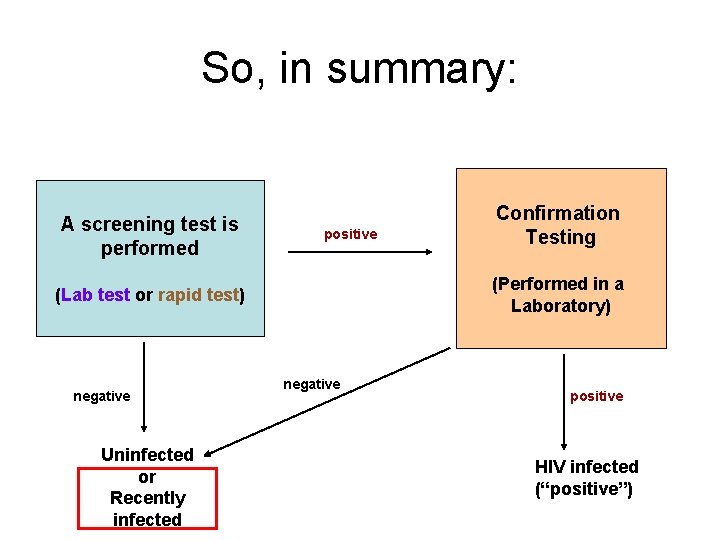
So, in summary: A screening test is performed positive (Performed in a Laboratory) (Lab test or rapid test) negative Uninfected or Recently infected Confirmation Testing negative positive HIV infected (“positive”)

Part 3: Direct Detection of Virus
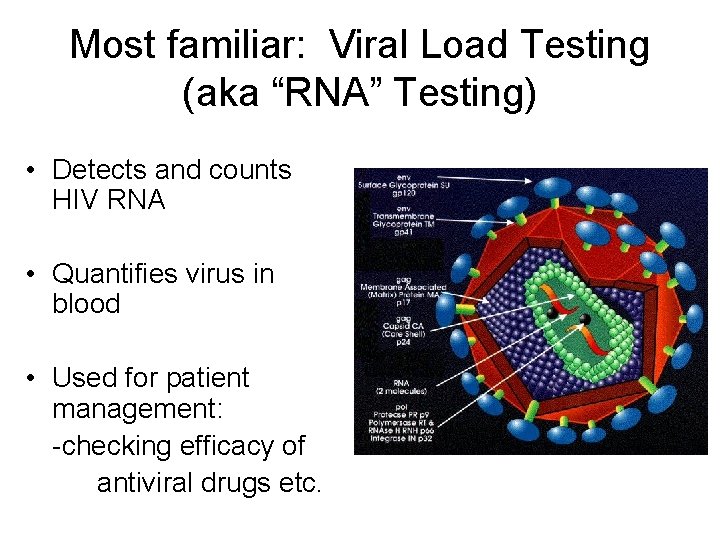
Most familiar: Viral Load Testing (aka “RNA” Testing) • Detects and counts HIV RNA • Quantifies virus in blood • Used for patient management: -checking efficacy of antiviral drugs etc.

Viral Load (RNA) tests: • FDA approved only for patient management • Not approved as a diagnostic test • One RNA test is approved as a confirmation test, but it is not widely utilized

Viral load (RNA) tests: on the window period timeline: Virus RNA becomes detectable Infection event Day 0 Virus Detectable Day 11 Antibody becomes detectable Ig. M Detectable Day 22 Ig. G Detectable Day 36
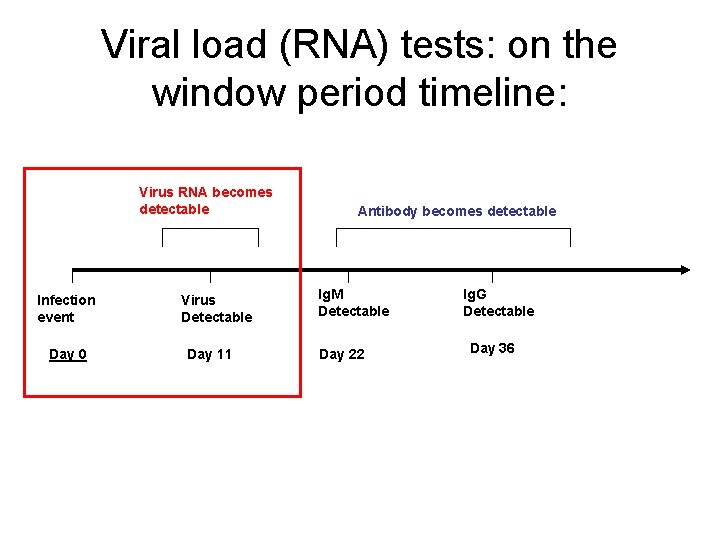
Viral load (RNA) tests: on the window period timeline: Virus RNA becomes detectable Infection event Day 0 Virus Detectable Day 11 Antibody becomes detectable Ig. M Detectable Day 22 Ig. G Detectable Day 36

Using Viral Load assays as screening tests? --- Why not? • Not FDA approved • Original viral load tests lacked specificity • Most infection in any given community is post-window period (established)

Using Viral Load assays as screening tests? ----Yes! • They are more sensitive for recent infection— shorter window periods • Newer methods have very high specificity— wouldn’t generate false positives at a dangerous rate • They detect infection when infected individuals are most infectious (very high viral loads)
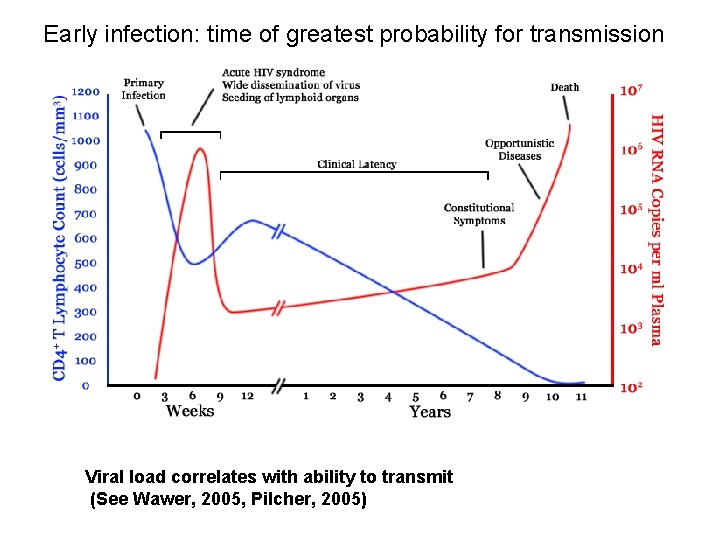
Early infection: time of greatest probability for transmission Viral load correlates with ability to transmit (See Wawer, 2005, Pilcher, 2005)

Implementation of RNA testing as HIV screening test at SF Public Health Lab: 2005 • Testing all patients from City Clinic, and now-- Magnet and AHP who were negative on their Antibody test and reported high risk • Specimens are “pooled”
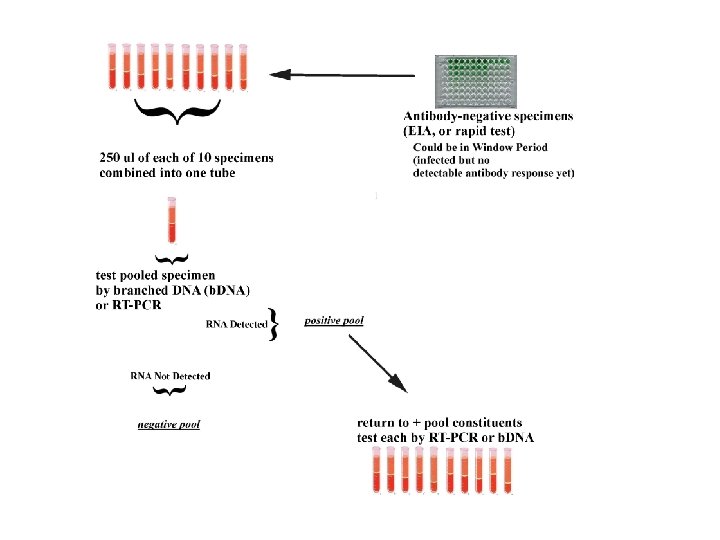

Results: so far (since 2005) • 102 individuals detected by RNA testing • Most have high (>100, 000) viral loads • Were negative by traditional testing • Zero false positives so far

RNA testing: Not Perfect • 7 -10 day turnaround time for results • Expensive, even in pooled format • In pooled format, window period probably about 12 -14 days
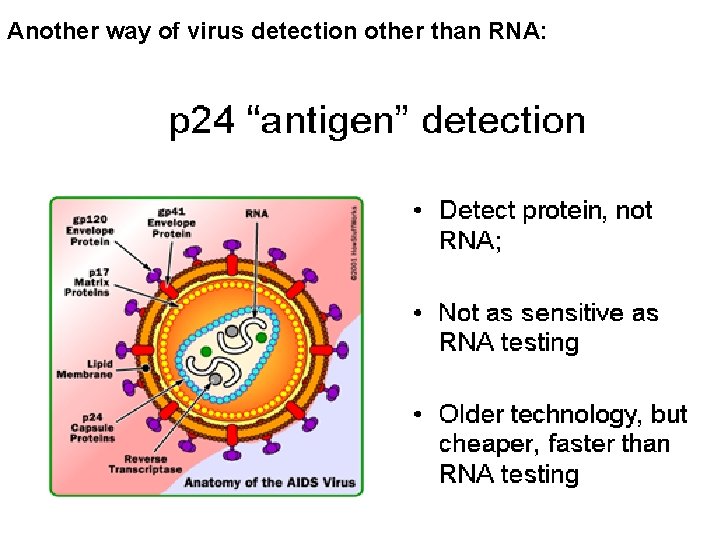
Another way of virus detection other than RNA:

“ 4 th Generation” EIA: Antigen-Antibody Combo testing • FDA Approved in June, 2010 • Detects virus and / or antibody simultaneously • Detects “antigen” of virus, not RNA

Window period for Antigen. Antibody Combo tests?

Window period for Antigen. Antibody Combo tests?

Study at SF Public Health Lab, in collaboration with Abbott • Tested panel of specimens found by RNA Testing in SF • Antigen-antibody combo test detected 89% of all those specimens

Advantages of antigen-antibody test: • Detects the great majority of recent infections • Fast turnaround time: ~3 -5 days • Cheaper than an RNA test • FDA approved (remember: RNA is not) – Only drawback? RNA is slightly more sensitive

Part 4: (Near) Future of Testing

Antigen-Antibody Rapid tests • ~20 minutes • More sensitive than laboratory antibody tests • Can detect virus and antibody • Entering clinical trials now

Point-of-Care RNA tests • A few companies working on this; fingerstick blood • Sensitivity around 5000 RNA copies • Compare this to ~50 copies/ml for lab based tests
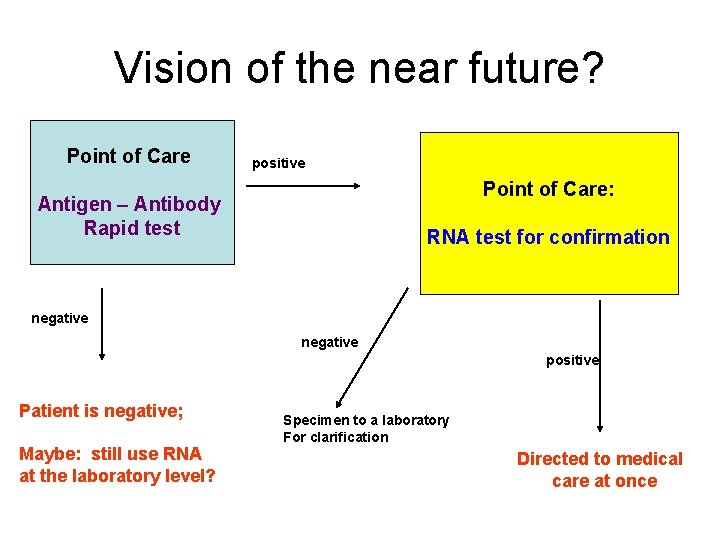
Vision of the near future? Point of Care positive Point of Care: Antigen – Antibody Rapid test RNA test for confirmation negative positive Patient is negative; Maybe: still use RNA at the laboratory level? Specimen to a laboratory For clarification Directed to medical care at once

Thank You for your Attention

The STOP STUDY

STOP Study (Screening Targeted Populations to Interrupt On-going Chain of Transmission) ◦ Specific Aims of the Study: To evaluate the yield, cost-effectiveness and feasibility of screening for acute HIV infection (AHI) with a 4 th generation enzyme immunoassay (EIA) in high-riskincidence settings compared to pooled nucleic acid amplification NAAT (aka pooled RNA). To evaluate the yield, cost-effectiveness and feasibility of enhanced partner notification/contact tracing techniques linked to AHI screening.

STOP Study CDC funded (SF, NYC, North Carolina) 4 year study – beginning June 2010 22, 000 specimens/49 AHI cases Working with high volume, high incidence HIV test sites All participants will be screened with a rapid HIV test, 4 th generation EIA test, and NAAT. Partner Services critical for persons with AHI.
- Slides: 55