Safety Profile of Biologic Agents in Rheumatoid Arthritis

Safety Profile of Biologic Agents in Rheumatoid Arthritis: A Systematic Review Ten Topics in Rheumatology Manila, Philippines Karina D. Torralba, MD Los Angeles County Medical Center University of Southern California

Disclosures • Wyeth, Medical Education Grant • Roche, Medical Education Grant • American College of Rheumatology Research and Education Foundation Clinician-Scholar Educator Award

Objectives, Clarification of Focus • To review updated safety issues with regard to the use of biologic therapy – Rheumatoid arthritis populations – Sources of data: • Registry studies • Metanalysis • Major Randomized controlled trials for more recently approved drugs • Case reports, case series for unique situations
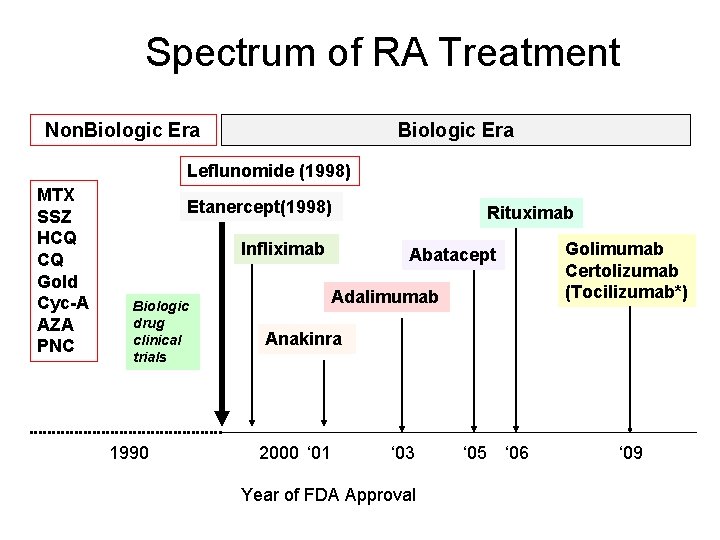
Spectrum of RA Treatment Non. Biologic Era Leflunomide (1998) MTX SSZ HCQ CQ Gold Cyc-A AZA PNC Etanercept(1998) Infliximab Biologic drug clinical trials 1990 Rituximab Abatacept Adalimumab Golimumab Certolizumab (Tocilizumab*) Anakinra 2000 ‘ 01 ‘ 03 Year of FDA Approval ‘ 05 ‘ 06 ‘ 09
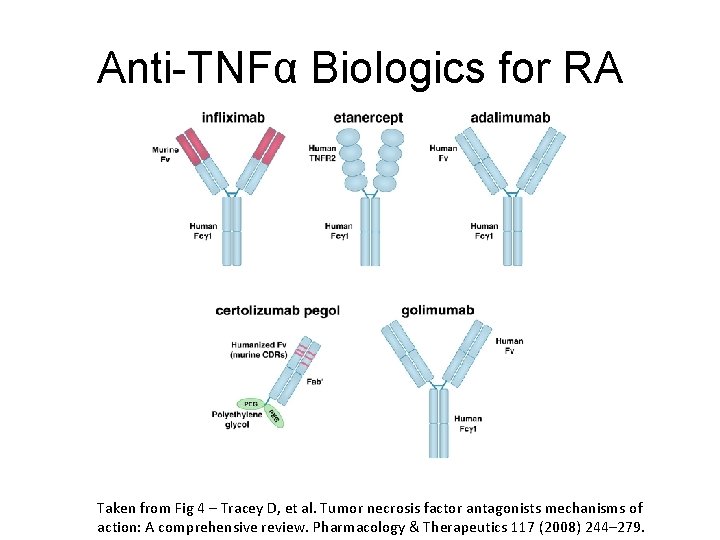
Anti-TNFα Biologics for RA Taken from Fig 4 – Tracey D, et al. Tumor necrosis factor antagonists mechanisms of action: A comprehensive review. Pharmacology & Therapeutics 117 (2008) 244– 279.

Biologic Therapy: Major Safety Issues • • Infections Infusion/injection-site reactions Autoimmune diseases Malignancy Immunogenicity, blocking antibodies Use in pregnancy Use in patients with congestive heart failure Use in patients with cardiovascular diseases

Predictive Factors of Serious Infections in RA • • • ↑Age +RF Nodules ↑ESR ↓WBC • • Extraarticular Features Corticosteroid use Diabetes mellitus Alcoholism • Chronic Lung Disease • Organic Brain Disease • Serious Infections (Definition) Life-threatening, fatal, requiring hospitalization, intravenous antibiotics, or resulting in persistent of significant disability Doran MF, et al. Predictors of infection n rheumatoid arthritis. Arthritis Rheum 2002; 46: 2294 -300.
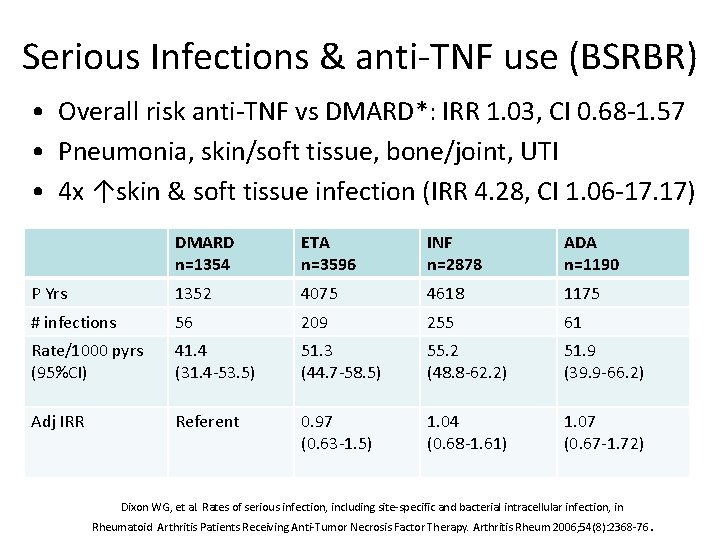
Serious Infections & anti-TNF use (BSRBR) • Overall risk anti-TNF vs DMARD*: IRR 1. 03, CI 0. 68 -1. 57 • Pneumonia, skin/soft tissue, bone/joint, UTI • 4 x ↑skin & soft tissue infection (IRR 4. 28, CI 1. 06 -17. 17) DMARD n=1354 ETA n=3596 INF n=2878 ADA n=1190 P Yrs 1352 4075 4618 1175 # infections 56 209 255 61 Rate/1000 pyrs (95%CI) 41. 4 (31. 4 -53. 5) 51. 3 (44. 7 -58. 5) 55. 2 (48. 8 -62. 2) 51. 9 (39. 9 -66. 2) Adj IRR Referent 0. 97 (0. 63 -1. 5) 1. 04 (0. 68 -1. 61) 1. 07 (0. 67 -1. 72) Dixon WG, et al. Rates of serious infection, including site-specific and bacterial intracellular infection, in Rheumatoid Arthritis Patients Receiving Anti-Tumor Necrosis Factor Therapy. Arthritis Rheum 2006; 54(8): 2368 -76.

Serious Infections with Rituximab, Abatacept, Anakinra • Metanalysis: 495 → 12 RCTs (3 RIT, 5 ABA, 4 ANA) Risk of serious infections, according to dose, OR (95% CI) High dose vs placebo Low dose vs placebo High dose vs low dose RIT (1000 vs 500 mg) 1. 68 (0. 64 -4. 35) 0. 24 (0. 01 -4. 33) 7. 20 (0. 43 -120. 66) ABA (<2 vs 10 mg/kg) 1. 35 (0. 78 -2. 33) 0. 84 (0. 13 -5. 3) 2. 16 (0. 52 -8. 98) DMARD users excluded 1. 24 (0. 70 -2. 29) ANA (<100 vs ≥ 100 mg) Comorbidity factors excluded 3. 40 (1. 11 -10. 46) 1. 67 (0. 51 -5. 41) 2. 0 (0. 48 -8. 33) 0. 51 (0. 03 -8. 27) 9. 63 (1. 31 -70. 91) 6. 41 (0. 81 -50. 30) Salliot C, et al. Risk of serious infections during rituximab, abatacept and anakinra treatments for rheumatoid arthritis: meta-analyses of randomised p lacebo-controlled trials. ARD 2009; 68: 25 -32.

TB risk and anti-TNFα therapy • 10712 anti-TNF α vs 3232 DMARD cohort • 34026 p-yrs vs 7345 p-yrs – 28447 pyrs actively on anti-TNF α • 40 episodes in 39 patients on anti-TNF α • Median time to diagnosis (mos) • 5. 5 (INF), 11 -13(ETN), 15 -18. 5 (ADA) • ↑↑ 3 -4 -fold among INF, ADA users vs ETA – 62% extrapulmonary, 28% disseminated – 10/39 deaths within 12 months of diagnosis Dixon WG, et al. Drug-Specific risk of Tuberculosis in patients with rheumatoid arthritis treated with anti-TNF therapy: Results from the BSRBR. ARD Oct 2009.
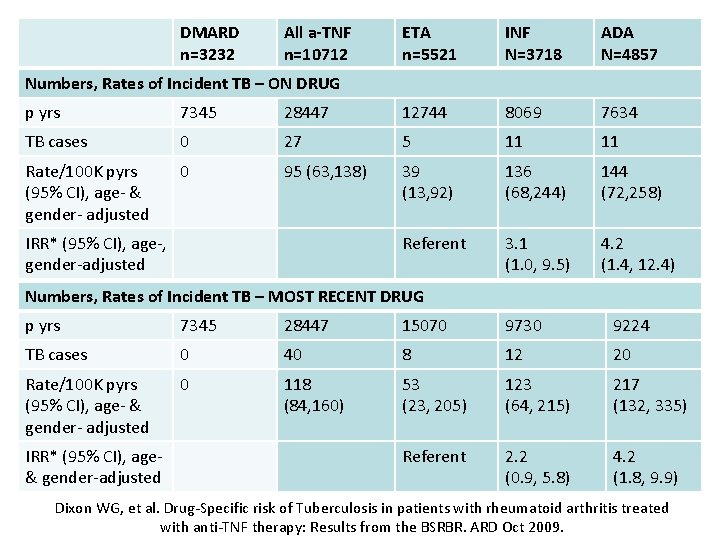
DMARD n=3232 All a-TNF n=10712 ETA n=5521 INF N=3718 ADA N=4857 Numbers, Rates of Incident TB – ON DRUG p yrs 7345 28447 12744 8069 7634 TB cases 0 27 5 11 11 Rate/100 K pyrs (95% CI), age- & gender- adjusted 0 95 (63, 138) 39 (13, 92) 136 (68, 244) 144 (72, 258) Referent 3. 1 (1. 0, 9. 5) 4. 2 (1. 4, 12. 4) IRR* (95% CI), age-, gender-adjusted Numbers, Rates of Incident TB – MOST RECENT DRUG p yrs 7345 28447 15070 9730 9224 TB cases 0 40 8 12 20 Rate/100 K pyrs (95% CI), age- & gender- adjusted 0 118 (84, 160) 53 (23, 205) 123 (64, 215) 217 (132, 335) Referent 2. 2 (0. 9, 5. 8) 4. 2 (1. 8, 9. 9) IRR* (95% CI), age& gender-adjusted Dixon WG, et al. Drug-Specific risk of Tuberculosis in patients with rheumatoid arthritis treated with anti-TNF therapy: Results from the BSRBR. ARD Oct 2009.
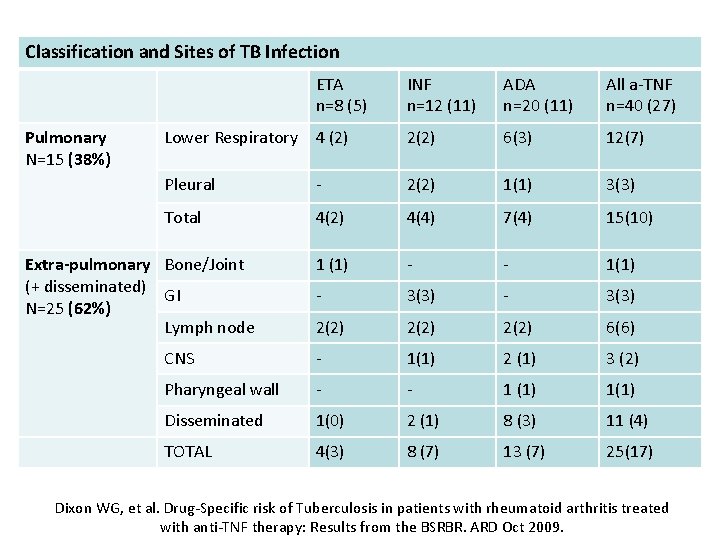
Classification and Sites of TB Infection ETA n=8 (5) Pulmonary N=15 (38%) INF n=12 (11) ADA n=20 (11) All a-TNF n=40 (27) Lower Respiratory 4 (2) 2(2) 6(3) 12(7) Pleural - 2(2) 1(1) 3(3) Total 4(2) 4(4) 7(4) 15(10) 1 (1) - - 1(1) - 3(3) 2(2) 6(6) CNS - 1(1) 2 (1) 3 (2) Pharyngeal wall - - 1 (1) 1(1) Disseminated 1(0) 2 (1) 8 (3) 11 (4) TOTAL 4(3) 8 (7) 13 (7) 25(17) Extra-pulmonary Bone/Joint (+ disseminated) GI N=25 (62%) Lymph node Dixon WG, et al. Drug-Specific risk of Tuberculosis in patients with rheumatoid arthritis treated with anti-TNF therapy: Results from the BSRBR. ARD Oct 2009.
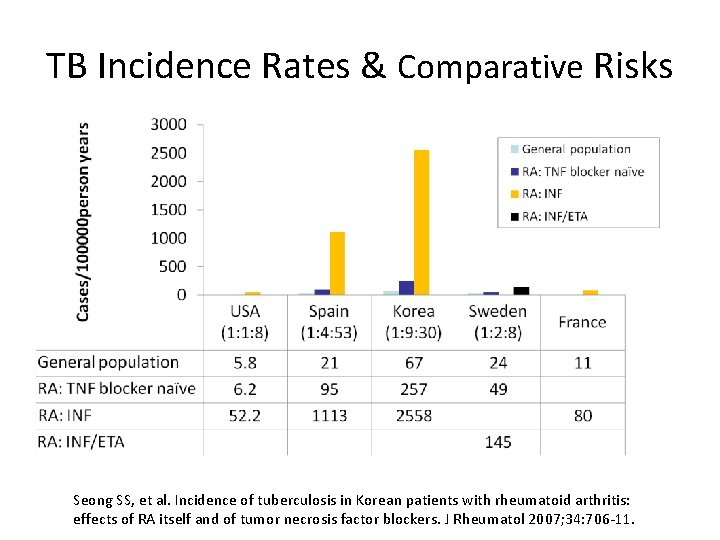
TB Incidence Rates & Comparative Risks Seong SS, et al. Incidence of tuberculosis in Korean patients with rheumatoid arthritis: effects of RA itself and of tumor necrosis factor blockers. J Rheumatol 2007; 34: 706 -11.

PPD screening, TB risk in US Immigrant Population • • • D Cooray, G Karpouzas, Harbor-UCLA Baseline and yearly TST ADA, ETA, IFX (INF) 27% (109/400) TST+ 30 conversions Cultures, PCR, CT Chest – 5 NTM, 2 MTB DV Cooray, GA Karpouzas, Harbor-UCLA, Los Angeles, CA ACR 2009 Plenary Session, Abstract 1153
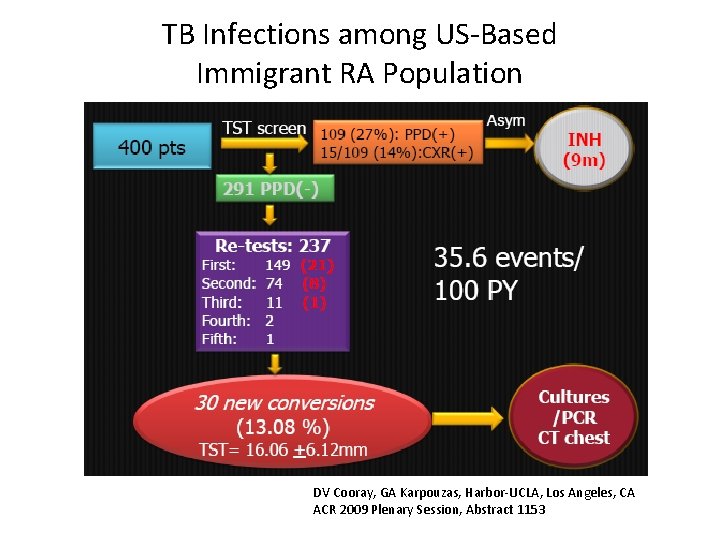
TB Infections among US-Based Immigrant RA Population DV Cooray, GA Karpouzas, Harbor-UCLA, Los Angeles, CA ACR 2009 Plenary Session, Abstract 1153
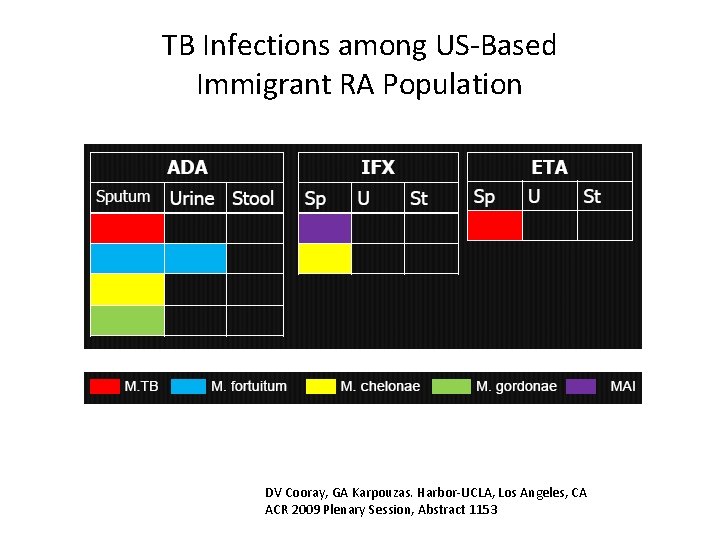
TB Infections among US-Based Immigrant RA Population DV Cooray, GA Karpouzas. Harbor-UCLA, Los Angeles, CA ACR 2009 Plenary Session, Abstract 1153

Autoimmune diseases induced by biologics • SLE or lupus-like syndromes • Vasculitis • Psoriasis • Sardoidosis • Demyelinating CNS Disease • Demyelinating peripheral neuropathies • Antiphospholipid syndrome or APS-like features • Interstitial lung diseases • Ocular Autoimmne Diseases • Autoimmune Hepatitis • Inflammatory myopathies Ramos-Casals M, et al. Best Prac Res Clin Rheumatol 2008 Torralba KD, Quismorio FP. Curr Op Rheumatol 2009
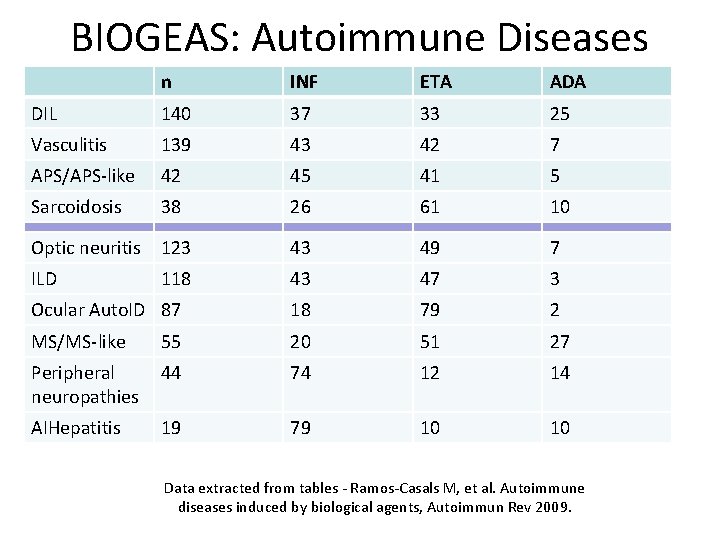
BIOGEAS: Autoimmune Diseases n INF ETA ADA DIL 140 37 33 25 Vasculitis 139 43 42 7 APS/APS-like 42 45 41 5 Sarcoidosis 38 26 61 10 Optic neuritis 123 43 49 7 ILD 118 43 47 3 Ocular Auto. ID 87 18 79 2 MS/MS-like 55 20 51 27 Peripheral neuropathies 44 74 12 14 AIHepatitis 19 79 10 10 Data extracted from tables - Ramos-Casals M, et al. Autoimmune diseases induced by biological agents, Autoimmun Rev 2009.
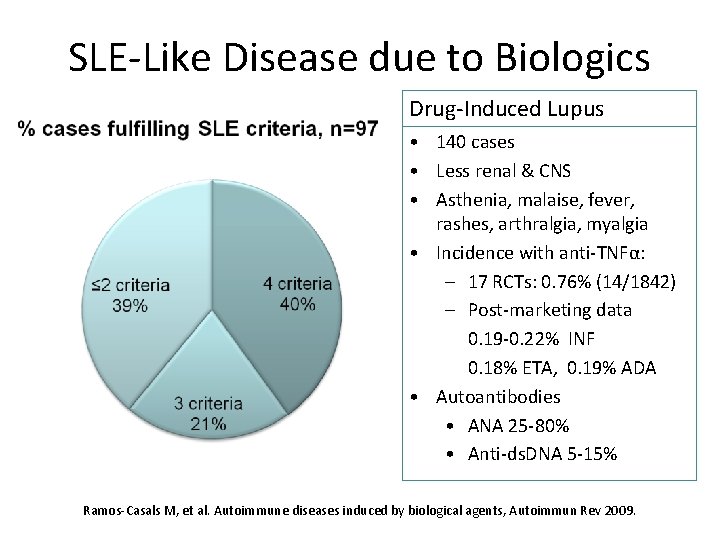
SLE-Like Disease due to Biologics Drug-Induced Lupus • 140 cases • Less renal & CNS • Asthenia, malaise, fever, rashes, arthralgia, myalgia • Incidence with anti-TNFα: – 17 RCTs: 0. 76% (14/1842) – Post-marketing data 0. 19 -0. 22% INF 0. 18% ETA, 0. 19% ADA • Autoantibodies • ANA 25 -80% • Anti-ds. DNA 5 -15% Ramos-Casals M, et al. Autoimmune diseases induced by biological agents, Autoimmun Rev 2009.

Systemic Autoimmune Diseases due to Biologics • Vasculitis – 88% cutaneous • Sarcoidosis – 74% pulmonary, 29% cutaneous • APS – a. PL (+) - 8/13 cases – Thromboses (30), thrombocytopenia (9), thrombophlebitis (4) • Peripheral Neuropathy • EMG (n=28, INF) ↑amplitude, median nerve; ↓velocity tibial, sural • ILD – 66% on MTX ? Potentiate MTX lung toxicity Ramos-Casals M, et al. Autoimmune diseases induced by biological agents, Autoimmun Rev 2009. Torralba KD, Quismorio FP. Sarcoidosis and the Rheumatologist. Curr Op Rheumatol 2009.

Psoriasis & anti-TNF α therapy: The Paradox • Cytokine alteration: IFN-α production by plasmacytoid dendritic cells • 25/9826 anti-TNF α group – IR: 1. 04 (95% CI 0. 67 -1. 54)/1000 pyrs • Majority – due to ADA • 79% continue anti-TNFα therapy – 25% resolution while on therapy • May respond anti-psoriatics • Resolves with drug discontinuation – 4% with continued psoriasis Harrison MJ, et al. Rates of new-onset psoriasis in patients with rheumatoid arthritis receiving anti-tumour necrosis factor α therapy: BSRBR. ARD 1009; 68: 209 -15. Collamer AN, et al. Psoriatic Skin Lesions Induced by Tumor Necrosis Factor Antagonist Therapy: A Literature Review and Potential Mechanisms of Action. Arthritis & Rheumatism 2008; 59: 996 -1001.

Immunogenicity: Antidrug antibodies Immunogenicity with anti-TNF agents INF ETA ADA CZP GOL Monotherapy +++ + + ND ND With MTX + +/- + ND Clinical Consequences • Drug resistance – Increased clearance – Inactivation of product • Drug Reactions – definite mechanism unclear Taken from Table 1, Fig 6 – Tracey D, et al. Tumor necrosis factor antagonists mechanisms of action: A comprehensive review. Pharmacology & Therapeutics 117

Malignancy risk with Biologics • 13001 subjects, 49000 p yrs (1998 -2005) • US NDB data compared with US NCI SEER • No increased risk for lymphoma, lung, breast, and colon cancer • Increased risk for skin cancer Nonmelanotic skin cancer – OR 1. 5 (95%CI 1. 2 -1. 8) 623 incident cases Melanoma - OR 2. 3 (95% CI 0. 9 -5. 4) Wolfe F, Michaud K. Biologic treatment of rheumatoid arthritis and the risk of malignancy: Analyses from a large US observational study. Arthritis Rheum 2007; 56(9): 2886 -95. .

Malignancy and anti-TNFα therapy • Swedish Cohort, multi-source (1999 -2006) • 240 cancers/6366 patients (25, 693 pyrs) – RR 1. 00 (95% CI 0. 87– 1. 17), c/w TNF-naïve – RR 0. 99 (95% CI 0. 79 -1. 24), c/w MTX starters • Organ-specific risk Not Increased • Agent-specific cancer risk • risk with follow-up (6 years) Askling J, et al. Cancer Risk in patients with rheumatoid arthritis treated with anti-tumor necrosis factor α therapies; Does the risk change with the time since start of treatment? Arthritis & Rheum 2009; 60(11); 3180 -9.

New Anti-TNFα agents: Safety Issues • Certolizumab Pegol – Pegylated Fab fragment, human anti-TNF Ab – t 1/2 14 days; q 2 week dosing – UTI, URTI (200 mg); Hypertension (400 mg); Headache • Golimumab – Humanized anti-TNF monoclonal antibody – SQ injection once monthly – URTI/Nasopharyngitis, Diarrhea – most common AEs Smolen J, et al. GO-AFTER. Lancet 2009; 374: 210– 21. Smolen J, et al. RAPID 2. Ann Rheum Dis. 2009 Jun; 68(6): 797 -804. Fleischmann R, et al. FAST 4 WARD. Ann Rheum Dis. 2009 Jun; 68(6): 805 -11.
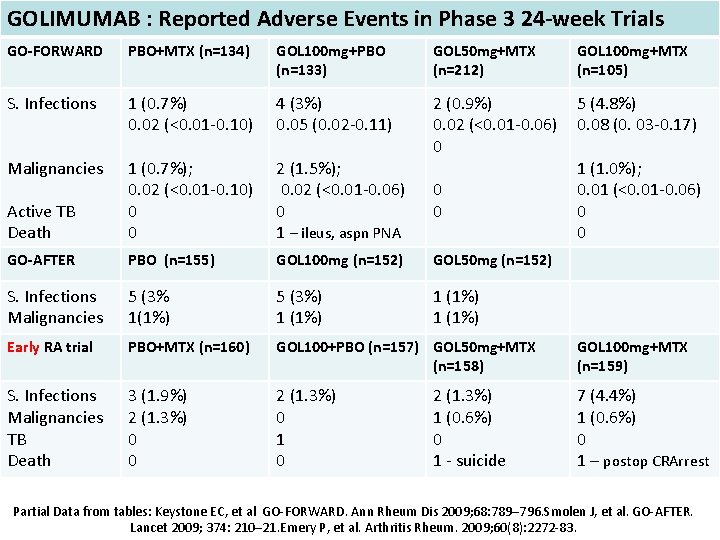
GOLIMUMAB : Reported Adverse Events in Phase 3 24 -week Trials GO-FORWARD PBO+MTX (n=134) GOL 100 mg+PBO (n=133) GOL 50 mg+MTX (n=212) GOL 100 mg+MTX (n=105) S. Infections 1 (0. 7%) 0. 02 (<0. 01 -0. 10) 4 (3%) 0. 05 (0. 02 -0. 11) 5 (4. 8%) 0. 08 (0. 03 -0. 17) Malignancies Active TB Death 1 (0. 7%); 0. 02 (<0. 01 -0. 10) 0 0 2 (1. 5%); 0. 02 (<0. 01 -0. 06) 0 1 – ileus, aspn PNA 2 (0. 9%) 0. 02 (<0. 01 -0. 06) 0 GO-AFTER PBO (n=155) GOL 100 mg (n=152) GOL 50 mg (n=152) S. Infections Malignancies 5 (3% 1(1%) 5 (3%) 1 (1%) Early RA trial PBO+MTX (n=160) GOL 100+PBO (n=157) GOL 50 mg+MTX (n=158) GOL 100 mg+MTX (n=159) S. Infections Malignancies TB Death 3 (1. 9%) 2 (1. 3%) 0 0 2 (1. 3%) 0 1 0 7 (4. 4%) 1 (0. 6%) 0 1 – postop CRArrest 0 0 2 (1. 3%) 1 (0. 6%) 0 1 - suicide 1 (1. 0%); 0. 01 (<0. 01 -0. 06) 0 0 Partial Data from tables: Keystone EC, et al GO-FORWARD. Ann Rheum Dis 2009; 68: 789– 796. Smolen J, et al. GO-AFTER. Lancet 2009; 374: 210– 21. Emery P, et al. Arthritis Rheum. 2009; 60(8): 2272 -83.
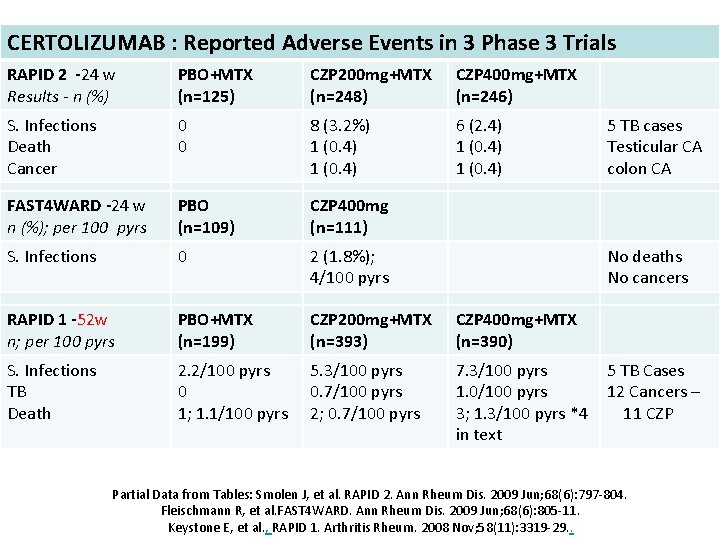
CERTOLIZUMAB : Reported Adverse Events in 3 Phase 3 Trials RAPID 2 -24 w Results - n (%) PBO+MTX (n=125) CZP 200 mg+MTX (n=248) CZP 400 mg+MTX (n=246) S. Infections Death Cancer 0 0 8 (3. 2%) 1 (0. 4) 6 (2. 4) 1 (0. 4) FAST 4 WARD -24 w n (%); per 100 pyrs PBO (n=109) CZP 400 mg (n=111) S. Infections 0 2 (1. 8%); 4/100 pyrs RAPID 1 -52 w n; per 100 pyrs PBO+MTX (n=199) CZP 200 mg+MTX (n=393) CZP 400 mg+MTX (n=390) S. Infections TB Death 2. 2/100 pyrs 0 1; 1. 1/100 pyrs 5. 3/100 pyrs 0. 7/100 pyrs 2; 0. 7/100 pyrs 7. 3/100 pyrs 1. 0/100 pyrs 3; 1. 3/100 pyrs *4 in text 5 TB cases Testicular CA colon CA No deaths No cancers 5 TB Cases 12 Cancers – 11 CZP Partial Data from Tables: Smolen J, et al. RAPID 2. Ann Rheum Dis. 2009 Jun; 68(6): 797 -804. Fleischmann R, et al. FAST 4 WARD. Ann Rheum Dis. 2009 Jun; 68(6): 805 -11. Keystone E, et al. , RAPID 1. Arthritis Rheum. 2008 Nov; 58(11): 3319 -29. .

Abatacept: Safety Issues • Acute infusion reactionsa – 9. 8% vs 6. 7% placebo, mild-moderate • Malignancy outcomes – 4134 Abatacept-treated patients compared with 41, 529 DMARD treated patients in 5 cohorts – No increased rates of malignancy, infection over 6 yearsb a. Sibilia J, Westhovens R. Safety of T-cell costimulation modulation with abatacept in patients with rheumatoid arthritis. Clin Exp Rheumatol 2007; 25 (5 Suppl 46): S 46 -56. b. Simon TA et al. Malignancies In RA Abatacept clinical development program. ARD 2008.
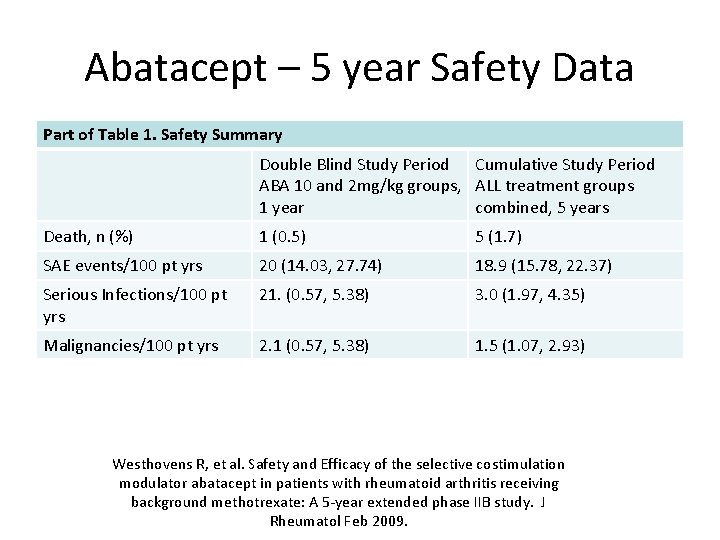
Abatacept – 5 year Safety Data Part of Table 1. Safety Summary Double Blind Study Period Cumulative Study Period ABA 10 and 2 mg/kg groups, ALL treatment groups 1 year combined, 5 years Death, n (%) 1 (0. 5) 5 (1. 7) SAE events/100 pt yrs 20 (14. 03, 27. 74) 18. 9 (15. 78, 22. 37) Serious Infections/100 pt yrs 21. (0. 57, 5. 38) 3. 0 (1. 97, 4. 35) Malignancies/100 pt yrs 2. 1 (0. 57, 5. 38) 1. 5 (1. 07, 2. 93) Westhovens R, et al. Safety and Efficacy of the selective costimulation modulator abatacept in patients with rheumatoid arthritis receiving background methotrexate: A 5 -year extended phase IIB study. J Rheumatol Feb 2009.
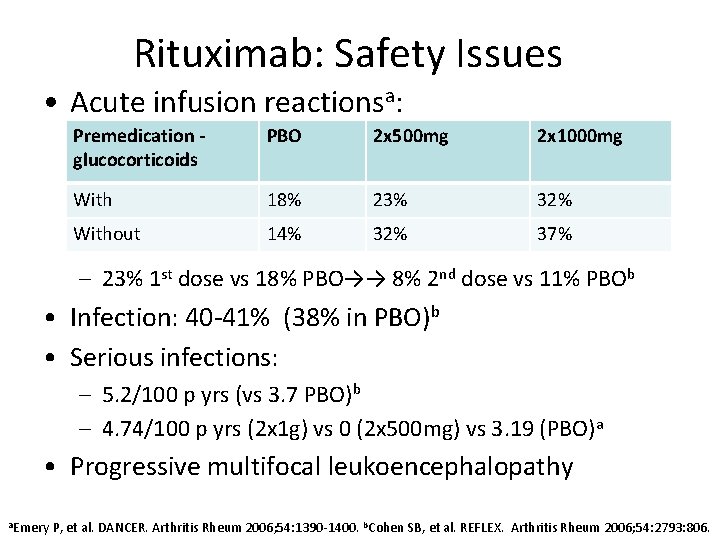
Rituximab: Safety Issues • Acute infusion reactionsa: Premedication glucocorticoids PBO 2 x 500 mg 2 x 1000 mg With 18% 23% 32% Without 14% 32% 37% – 23% 1 st dose vs 18% PBO→→ 8% 2 nd dose vs 11% PBOb • Infection: 40 -41% (38% in PBO)b • Serious infections: – 5. 2/100 p yrs (vs 3. 7 PBO)b – 4. 74/100 p yrs (2 x 1 g) vs 0 (2 x 500 mg) vs 3. 19 (PBO)a • Progressive multifocal leukoencephalopathy a. Emery P, et al. DANCER. Arthritis Rheum 2006; 54: 1390 -1400. b. Cohen SB, et al. REFLEX. Arthritis Rheum 2006; 54: 2793: 806.

Tocilizumab: Safety Issues • Infections – Nasopharyngitis – No TB occurences • Laboratory Abnormalities – 57% (4 mg), 76% (8 mg) Liver enzyme elevations • CHARISMA: mild, transient • ↑↑TOC+MTX (11%) vs TOC alone (6%) Cholesterol elevation – 44% Maini R, et al. Arthritis Rheum 2003; 48 Suppl: S 652; Nishimoto N, et al. Arthritis Rheum 2004; 50: 1761 -9; Emery P, et al. Arthritis Rheum 2008; 58 Suppl: S 617.
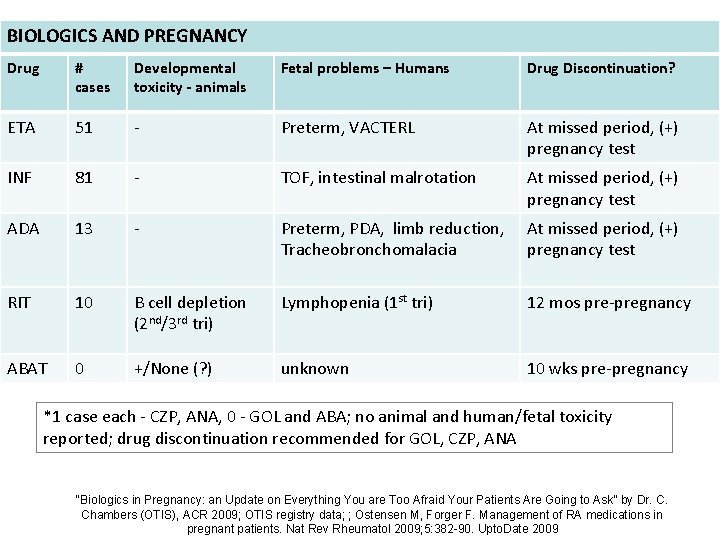
BIOLOGICS AND PREGNANCY Drug # cases Developmental toxicity - animals Fetal problems – Humans Drug Discontinuation? ETA 51 - Preterm, VACTERL At missed period, (+) pregnancy test INF 81 - TOF, intestinal malrotation At missed period, (+) pregnancy test ADA 13 - Preterm, PDA, limb reduction, Tracheobronchomalacia At missed period, (+) pregnancy test RIT 10 B cell depletion (2 nd/3 rd tri) Lymphopenia (1 st tri) 12 mos pre-pregnancy ABAT 0 +/None (? ) unknown 10 wks pre-pregnancy *1 case each - CZP, ANA, 0 - GOL and ABA; no animal and human/fetal toxicity reported; drug discontinuation recommended for GOL, CZP, ANA “Biologics in Pregnancy: an Update on Everything You are Too Afraid Your Patients Are Going to Ask” by Dr. C. Chambers (OTIS), ACR 2009; OTIS registry data; ; Ostensen M, Forger F. Management of RA medications in pregnant patients. Nat Rev Rheumatol 2009; 5: 382 -90. Upto. Date 2009

Take Home Points • Vigilant monitoring is needed for infections, malignancy, infusion/injection reactions, and other safety issues – Vaccination early into RA treatment should be considered – TB screening • Risk: benefit should be considered on an individual basis • Biologics are relatively safe, however long-term studies especially for recently approved drugs are needed • Use of biologics in pregnancy/lactation – needs further study
- Slides: 33