Safety and Immunogenicity Study of Rabies G Protein

Safety and Immunogenicity Study of Rabies G Protein Vaccine Administered to Healthy Volunteers: A Preliminary Report RI Modi 1, B Khamar 1, D Sawhney 1, M Talpada 2, Ashwini Shetty 3, SN Madhusudana 3, G Glenn 4, L Fries 4 1 2 3 4 Cadila Pharmaceuticals Ltd, Ahmedabad, India. Apollo Hospital, Ahmedabad, India NIMHANS, Banglore, India Novavax Inc, MD, USA

Rabies-Understanding the Problem • Rabies is a vaccine-preventable viral disease which occurs in more than 150 countries and territories 1 – Dog rabies potentially threatens over 3 billion people in Asia and Africa – People most at risk live in rural areas where human vaccines and immunoglobulin are not readily available or accessible • Rabies is a neglected disease of poor and vulnerable populations whose deaths are rarely reported – More than 95% of human deaths occur in Asia and Africa • 40% of people who are bitten by suspect rabid animals are children under 15 years of age • Compliance is a major issue with the currently available schedules – Just 33% of the category III bites complete the 5 injection regimen 2 • Large scale pre-exposure immunization of children has been advocated but the implementation is hindered by financial and logistic barriers 3 1. Source: WHO; 2. Study on the compliance and economic cost of rabies vaccination]. Zhongguo Yi Miao He Mian Yi. 2010 Jun; 16(3): 254 -7; 3. Cost comparison of rabies pre-exposure vaccination with post-exposure treatment in Thai children Vaccine. 2006 Feb 27; 24(9): 1478 -82. Epub 2005 Sep 29. 2

3

4
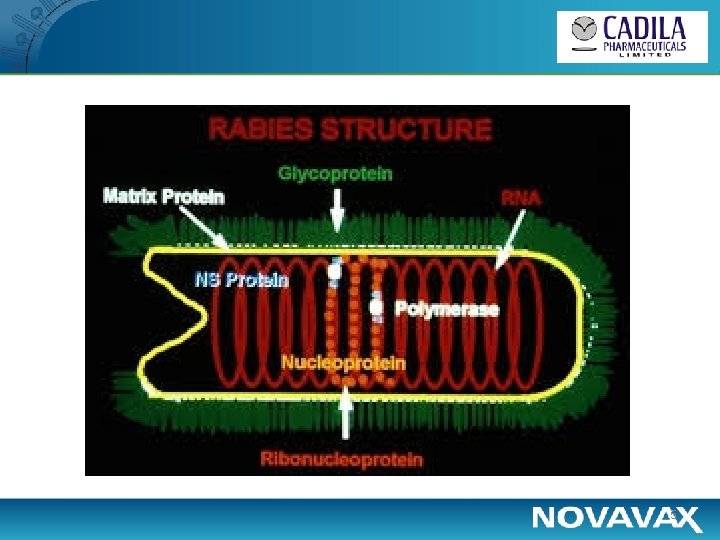
5

Rationale for a Novel Vaccine • The key target for neutralizing antibodies is rabies virus glycoprotein (Rabies G Protein) • The currently available vaccines are inactivated whole virus based vaccines: – 3 to 5 dose regimens – variable potency • Cost-effective improvements allowing fewer inoculations with good and sustained antibody induction would be highly desirable for both pre-exposure and post-exposure vaccination • Recombinant rabies G protein vaccine (sub-unit vaccine) may offer seroprotection with fewer doses (2 or 3) and simplified production 3
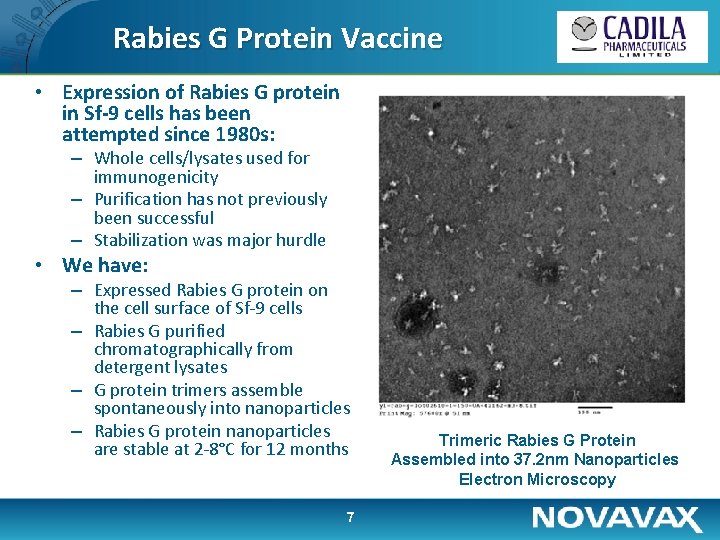
Rabies G Protein Vaccine • Expression of Rabies G protein in Sf-9 cells has been attempted since 1980 s: – Whole cells/lysates used for immunogenicity – Purification has not previously been successful – Stabilization was major hurdle • We have: – Expressed Rabies G protein on the cell surface of Sf-9 cells – Rabies G purified chromatographically from detergent lysates – G protein trimers assemble spontaneously into nanoparticles – Rabies G protein nanoparticles are stable at 2 -8°C for 12 months 7 Trimeric Rabies G Protein Assembled into 37. 2 nm Nanoparticles Electron Microscopy

Rabies G Protein Vaccine Phase I trial (CTRI/2014/04/004538) • Randomized, open label immunogenicity and safety study in healthy adult volunteers • Study Center: − Apollo Hospital, Gandhinagar, Gujarat, India • Blinded immunogenicity analysis by RFFIT at independent lab: − Department of Neurovirology − National Institute of Mental Health and Neurological Sciences(NIMHANS), India • WHO Collaborating Centre for Reference and Research in Rabies • All immunogenicity sample analysis was blinded 8
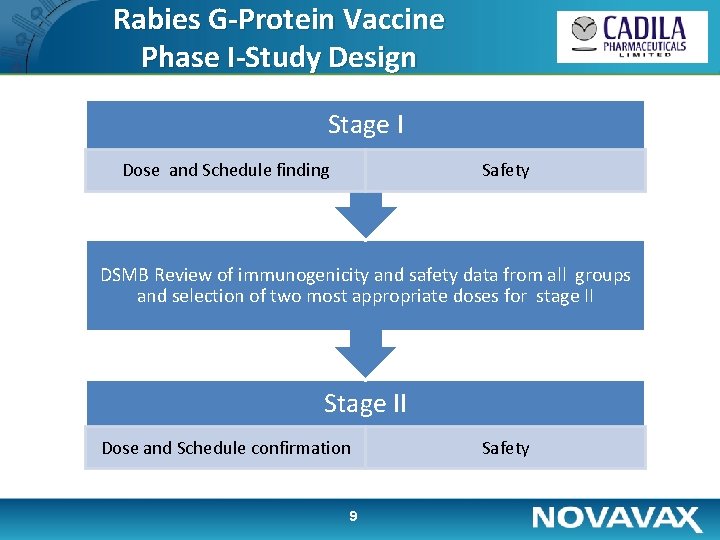
Rabies G-Protein Vaccine Phase I-Study Design Stage I Dose and Schedule finding Safety DSMB Review of immunogenicity and safety data from all groups and selection of two most appropriate doses for stage II Stage II Dose and Schedule confirmation 9 Safety
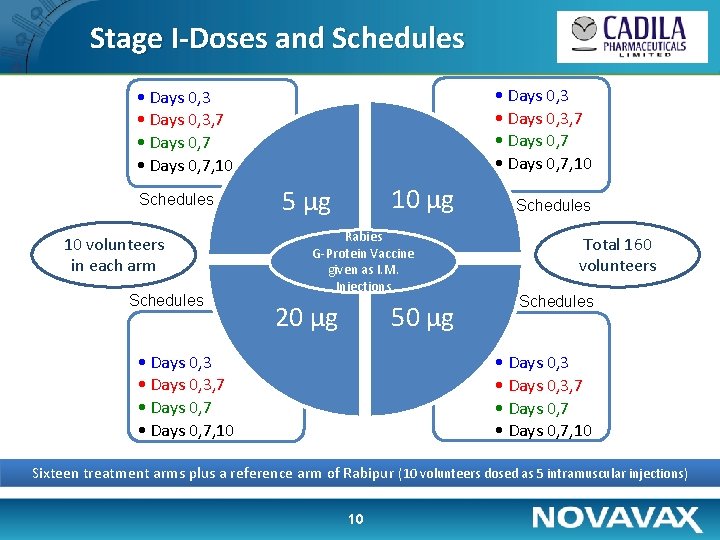
Stage I-Doses and Schedules • Days 0, 3, 7 • Days 0, 7, 10 Schedules 10 volunteers in each arm Schedules 10 µg 5 µg Rabies G-Protein Vaccine given as I. M. Injections 20 µg 50 µg • Days 0, 3, 7 • Days 0, 7, 10 Schedules Total 160 volunteers Schedules • Days 0, 3, 7 • Days 0, 7, 10 Sixteen treatment arms plus a reference arm of Rabipur (10 volunteers dosed as 5 intramuscular injections) 10

Study Population and Trial Activities • Study Population – Stage I • Healthy adult males aged 18 -64 years – Stage II • Healthy adult males and non-pregnant females 18 -64 years • Blood sampling for immunogenicity by RFFIT at: – Day 0 (pre-vaccination) and Days 14, 42, 90, 135 and 180 (post vaccination) • Safety – Safety follow-ups were done at all visits till day 180 – Volunteers maintained an adverse event diary till day 35 post vaccination • Both solicited and unsolicited adverse events were recorded 11
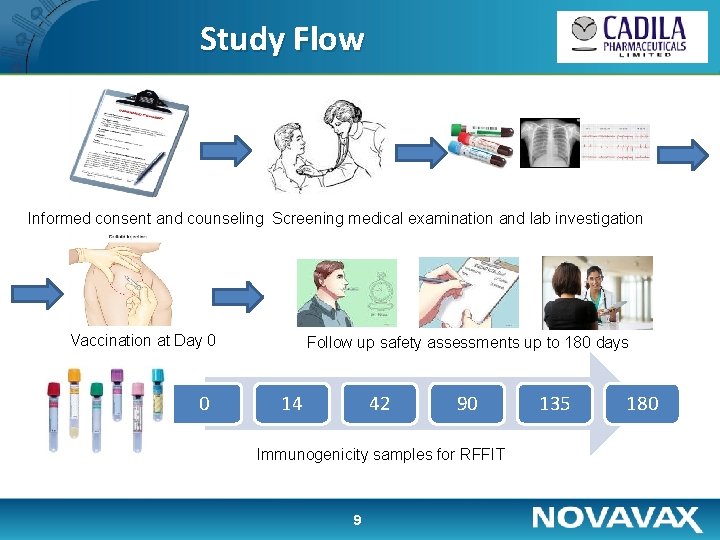
Study Flow Informed consent and counseling Screening medical examination and lab investigation Vaccination at Day 0 0 Follow up safety assessments up to 180 days 14 42 90 Immunogenicity samples for RFFIT 9 135 180

Results Stage 1 10
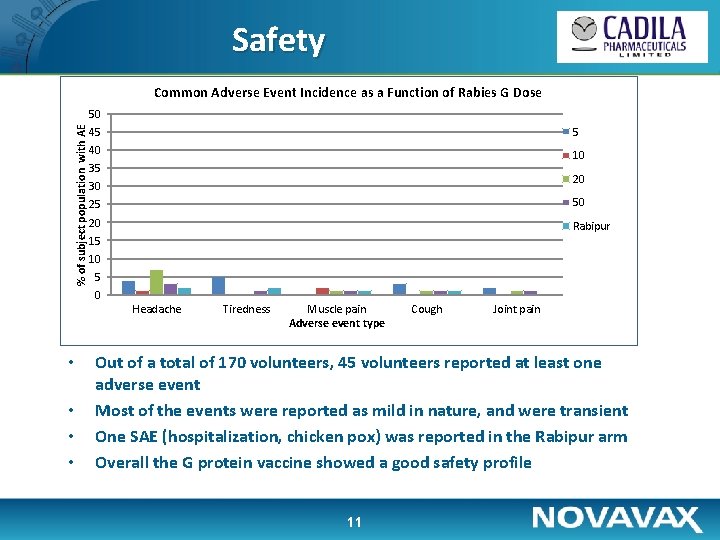
Safety Common Adverse Event Incidence as a Function of Rabies G Dose % of subject population with AE 50 45 5 40 10 35 20 30 25 50 20 Rabipur 15 10 5 0 Headache • • Tiredness Muscle pain Adverse event type Cough Joint pain Out of a total of 170 volunteers, 45 volunteers reported at least one adverse event Most of the events were reported as mild in nature, and were transient One SAE (hospitalization, chicken pox) was reported in the Rabipur arm Overall the G protein vaccine showed a good safety profile 11
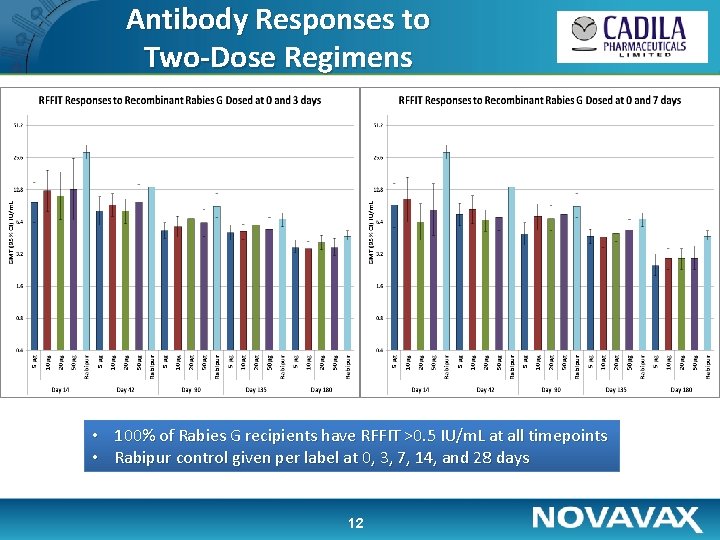
Antibody Responses to Two-Dose Regimens • 100% of Rabies G recipients have RFFIT >0. 5 IU/m. L at all timepoints • Rabipur control given per label at 0, 3, 7, 14, and 28 days 12
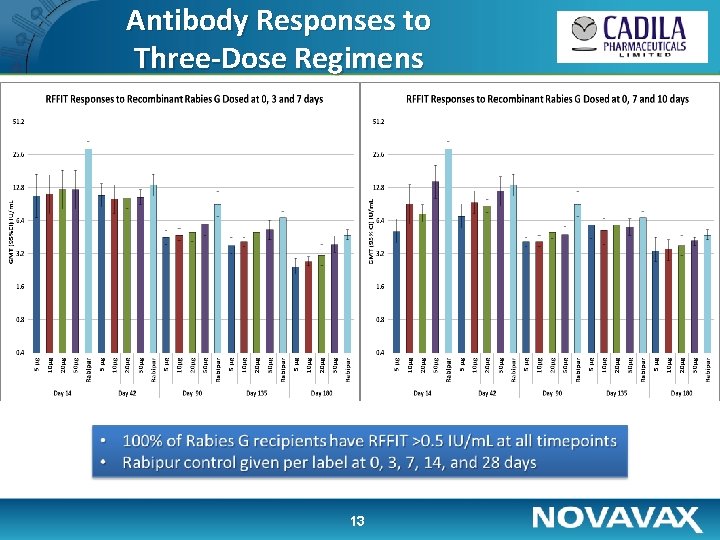
Antibody Responses to Three-Dose Regimens 13

Immunogenicity • Antibody titer ≥ 0. 5 IU/ml was considered as sero-protective – Three out of 160 volunteers (1. 9%) had titers ≥ 0. 5 IU/ml at baseline; these were excluded from analyses • 100% Sero-protection was achieved in all groups at day 14 with titers sustained well above 0. 5 IU/m. L at day 180 • 10 to 50µg of recombinant rabies G protein show almost similar dose response • Three-dose regimens notably more immunogenic than two doses • Both two- and three-dose regimens which include day 0 + 3 exposures appear somewhat more advantageous 17

Summary and Conclusions • For the first time a recombinant Rabies G protein vaccine has been evaluated in human subjects • All volunteers at all doses and schedules showed seroprotection at days 14, 42, 90, 135 and 180 • The vaccine was found to be safe and well tolerated • Rabies G Protein vaccine induces sero-protective levels in human volunteers with two and three dose regimens with doses from 5µg to 50µg 18

Future Directions • Complete Stage II (ongoing) with optimal dose regimens • Phase III trial of a post-exposure prophylaxis regimen in healthy volunteers • Assess the immunosuppressive effect of RIG coadministration, if any • Evaluate a pre-exposure prophylaxis regimen Efficacy studies if feasible and induction of cellular immune responses ( Th 1 and Th 2) 19

Thank You 17 20
- Slides: 20