Safety and Efficacy of Mavacamten in Symptomatic NonObstructive

Safety and Efficacy of Mavacamten in Symptomatic Non-Obstructive Hypertrophic Cardiomyopathy: The MAVERICK-HCM Study Carolyn Y. Ho, MD Cardiovascular Division, Brigham and Women's Hospital, Boston, Massachusetts On behalf of the MAVERICK investigators

Disclosures Presenting Author: • Consultant for Myo. Kardia • SHa. Re Registry lead investigator (funded by Myo. Kardia) • Advisory board for Ambry Genetics and Novartis This study was funded by Myo. Kardia, Inc. 2

Introduction: Non-Obstructive HCM Hypertrophic cardiomyopathy (HCM) is a primary myocardial disorder: • Unexplained left ventricular (LV) hypertrophy • Often caused by pathogenic variants in sarcomeric genes ~ 30% of patients have non-obstructive HCM (LVOT gradient <30 mm. Hg) No proven pharmacologic therapies. Current therapies include beta-blockers, verapamil, & diuretics: 1 -2 • Focus on symptomatic relief • Manage atrial fibrillation • Reduce congestion Cardiac transplantation may be the only option if severe, refractory symptoms By altering the contractile mechanics of the cardiomyocyte, myosin inhibitors have the potential to modify pathophysiology and improve symptoms associated with non-obstructive HCM. 1 Elliot PM. Eur Heart J. 2014; 35(39): 2733 -2779. BJ. Circulation. 2011; 124(24): 2761 -2796. 2 Gersh 3
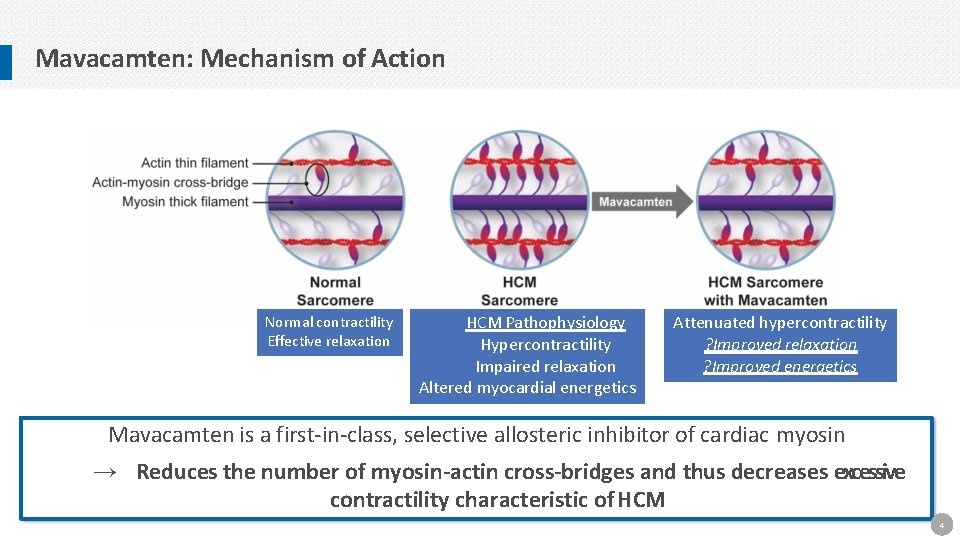
Mavacamten: Mechanism of Action Normal contractility Effective relaxation HCM Pathophysiology Hypercontractility Impaired relaxation Altered myocardial energetics Attenuated hypercontractility ? Improved relaxation ? Improved energetics Mavacamten is a first-in-class, selective allosteric inhibitor of cardiac myosin → Reduces the number of myosin-actin cross-bridges and thus decreases excessive contractility characteristic of HCM 4
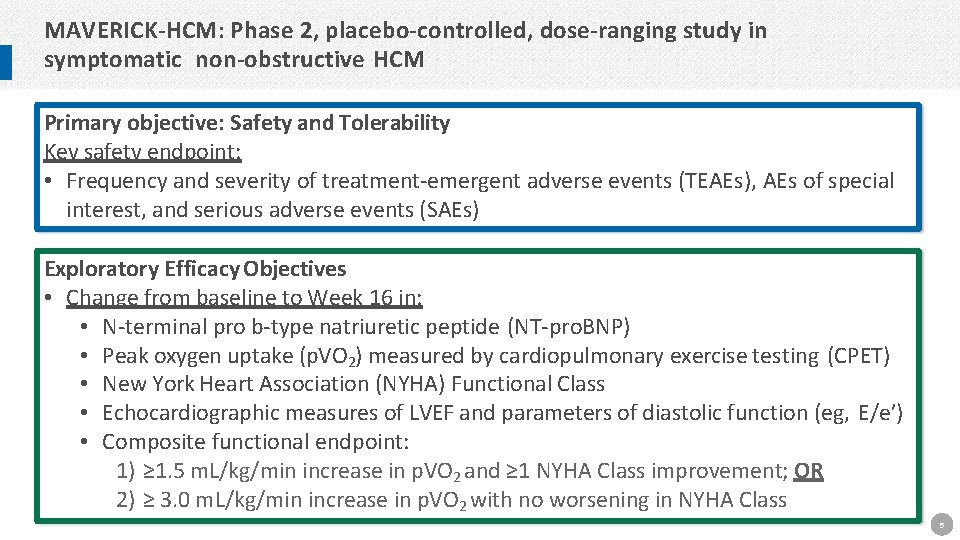
MAVERICK-HCM: Phase 2, placebo-controlled, dose-ranging study in symptomatic non-obstructive HCM Primary objective: Safety and Tolerability Key safety endpoint: • Frequency and severity of treatment-emergent adverse events (TEAEs), AEs of special interest, and serious adverse events (SAEs) Exploratory Efficacy Objectives • Change from baseline to Week 16 in: • N-terminal pro b-type natriuretic peptide (NT-pro. BNP) • Peak oxygen uptake (p. VO 2) measured by cardiopulmonary exercise testing (CPET) • New York Heart Association (NYHA) Functional Class • Echocardiographic measures of LVEF and parameters of diastolic function (eg, E/e’) • Composite functional endpoint: 1) ≥ 1. 5 m. L/kg/min increase in p. VO 2 and ≥ 1 NYHA Class improvement; OR 2) ≥ 3. 0 m. L/kg/min increase in p. VO 2 with no worsening in NYHA Class 5
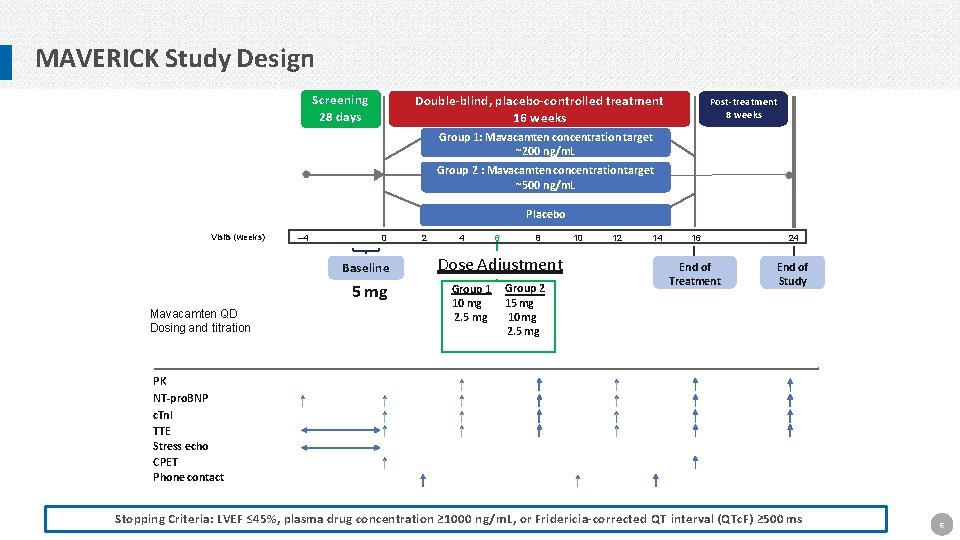
MAVERICK Study Design Screening 28 days Double-blind, placebo-controlled treatment 16 weeks Post-treatment 8 weeks Group 1: Mavacamten concentration target ~200 ng/m. L Group 2 : Mavacamten concentration target ~500 ng/m. L Placebo Visits (weeks) – 4 0 Baseline 5 mg Mavacamten QD Dosing and titration 2 4 6 8 Dose Adjustment Group 1 10 mg 2. 5 mg Group 2 15 mg 10 mg 2. 5 mg 10 12 14 16 24 End of Treatment End of Study PK NT-pro. BNP c. Tnl TTE Stress echo CPET Phone contact Stopping Criteria: LVEF ≤ 45%, plasma drug concentration ≥ 1000 ng/m. L, or Fridericia-corrected QT interval (QTc. F) ≥ 500 ms 6
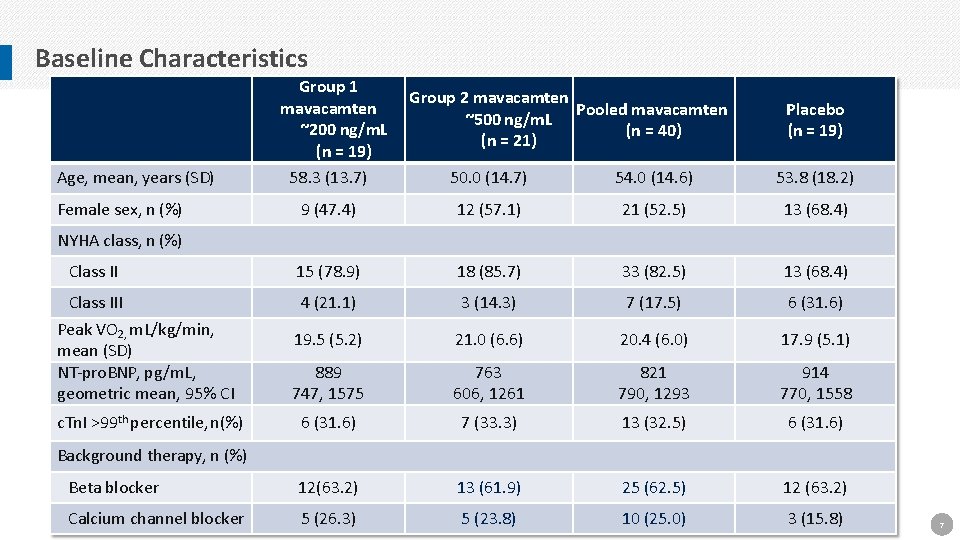
Baseline Characteristics Age, mean, years (SD) Female sex, n (%) Group 1 mavacamten ~200 ng/m. L (n = 19) 58. 3 (13. 7) Group 2 mavacamten Pooled mavacamten ~500 ng/m. L (n = 40) (n = 21) Placebo (n = 19) 50. 0 (14. 7) 54. 0 (14. 6) 53. 8 (18. 2) 9 (47. 4) 12 (57. 1) 21 (52. 5) 13 (68. 4) 15 (78. 9) 18 (85. 7) 33 (82. 5) 13 (68. 4) 4 (21. 1) 3 (14. 3) 7 (17. 5) 6 (31. 6) NYHA class, n (%) Class III Peak VO 2, m. L/kg/min, mean (SD) NT-pro. BNP, pg/m. L, geometric mean, 95% CI 19. 5 (5. 2) 21. 0 (6. 6) 20. 4 (6. 0) 17. 9 (5. 1) 889 747, 1575 763 606, 1261 821 790, 1293 914 770, 1558 c. Tn. I >99 th percentile, n(%) 6 (31. 6) 7 (33. 3) 13 (32. 5) 6 (31. 6) Beta blocker 12(63. 2) 13 (61. 9) 25 (62. 5) 12 (63. 2) Calcium channel blocker 5 (26. 3) 5 (23. 8) 10 (25. 0) 3 (15. 8) Background therapy, n (%) 7
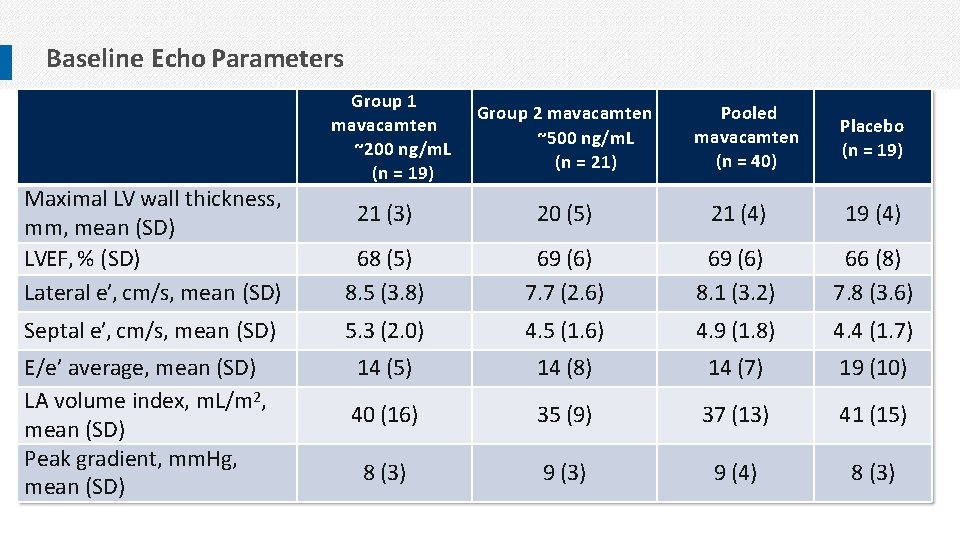
Baseline Echo Parameters Maximal LV wall thickness, mm, mean (SD) LVEF, % (SD) Lateral e’, cm/s, mean (SD) Group 1 mavacamten ~200 ng/m. L (n = 19) Group 2 mavacamten ~500 ng/m. L (n = 21) Pooled mavacamten (n = 40) Placebo (n = 19) 21 (3) 20 (5) 21 (4) 19 (4) 68 (5) 8. 5 (3. 8) 69 (6) 7. 7 (2. 6) 69 (6) 8. 1 (3. 2) 66 (8) 7. 8 (3. 6) Septal e’, cm/s, mean (SD) 5. 3 (2. 0) 4. 5 (1. 6) 4. 9 (1. 8) 4. 4 (1. 7) E/e’ average, mean (SD) LA volume index, m. L/m 2, mean (SD) Peak gradient, mm. Hg, mean (SD) 14 (5) 14 (8) 14 (7) 19 (10) 40 (16) 35 (9) 37 (13) 41 (15) 8 (3) 9 (4) 8 (3)
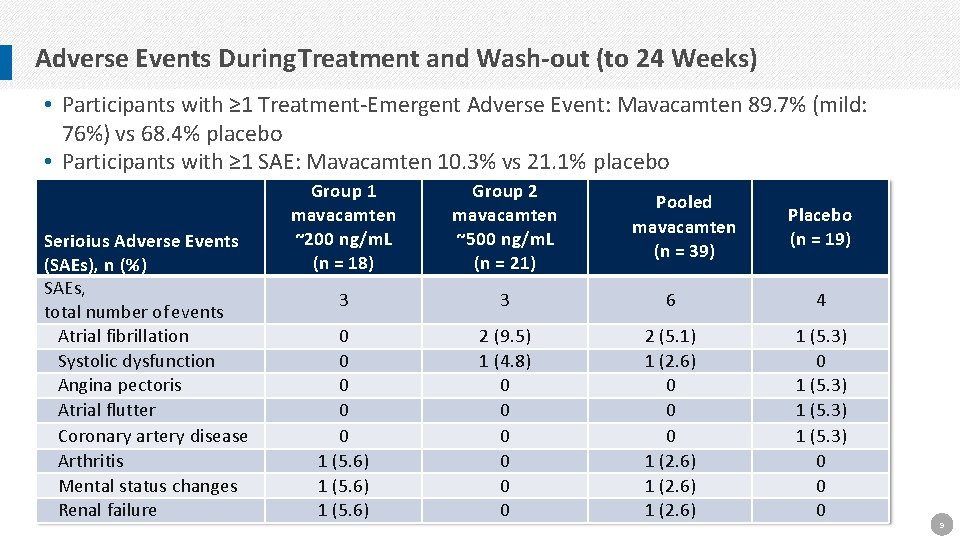
Adverse Events During Treatment and Wash-out (to 24 Weeks) • Participants with ≥ 1 Treatment-Emergent Adverse Event: Mavacamten 89. 7% (mild: 76%) vs 68. 4% placebo • Participants with ≥ 1 SAE: Mavacamten 10. 3% vs 21. 1% placebo Serioius Adverse Events (SAEs), n (%) SAEs, total number of events Atrial fibrillation Systolic dysfunction Angina pectoris Atrial flutter Coronary artery disease Arthritis Mental status changes Renal failure Group 1 mavacamten ~200 ng/m. L (n = 18) Group 2 mavacamten ~500 ng/m. L (n = 21) 3 3 6 4 0 0 0 1 (5. 6) 2 (9. 5) 1 (4. 8) 0 0 0 2 (5. 1) 1 (2. 6) 0 0 0 1 (2. 6) 1 (5. 3) 0 1 (5. 3) 0 0 0 Pooled mavacamten (n = 39) Placebo (n = 19) 9

Effect of Mavacamten on LVEF Mean reduction in LVEF, % (SD) (baseline to week 16) Group 1 mavacamten ~200 ng/m. L (n = 19) Group 2 mavacamten ~500 ng/m. L (n = 21) Pooled mavacamten (n = 40) Placebo (n = 19) -2. 3% (5. 3) -5. 6% (9. 7) -4. 1% (8. 0) -2. 3% (4. 9) 10
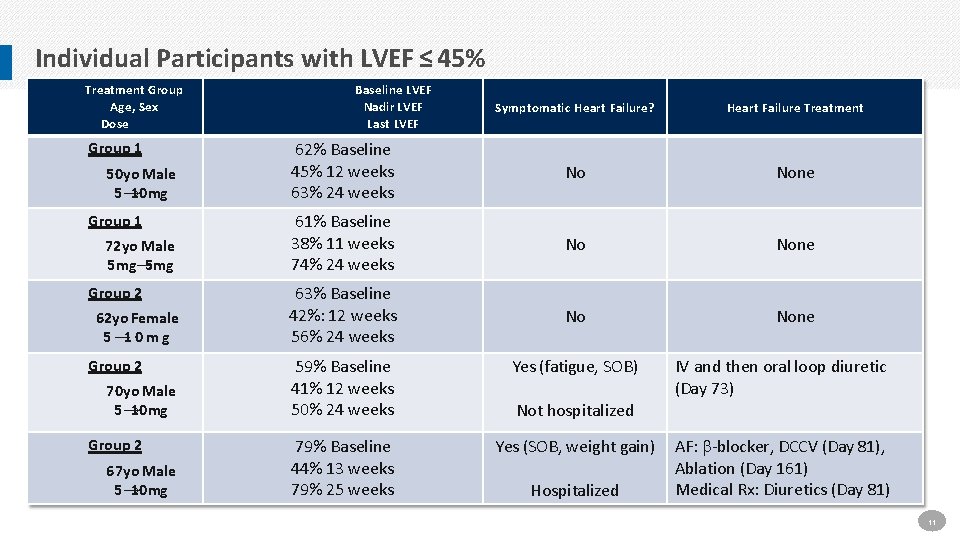
Individual Participants with LVEF ≤ 45% Treatment Group Age, Sex Dose Group 1 50 yo Male 5→ 10 mg Group 1 72 yo Male 5 mg→ 5 mg Group 2 62 yo Female 5 → 1 0 m g Group 2 70 yo Male 5→ 10 mg Group 2 67 yo Male 5→ 10 mg Baseline LVEF Nadir LVEF Last LVEF Symptomatic Heart Failure? Heart Failure Treatment 62% Baseline 45% 12 weeks 63% 24 weeks No None 61% Baseline 38% 11 weeks 74% 24 weeks No None 63% Baseline 42%: 12 weeks 56% 24 weeks No None 59% Baseline 41% 12 weeks 50% 24 weeks Yes (fatigue, SOB) 79% Baseline 44% 13 weeks 79% 25 weeks Yes (SOB, weight gain) IV and then oral loop diuretic (Day 73) Not hospitalized Hospitalized AF: -blocker, DCCV (Day 81), Ablation (Day 161) Medical Rx: Diuretics (Day 81) 11

Exploratory Efficacy Results 12
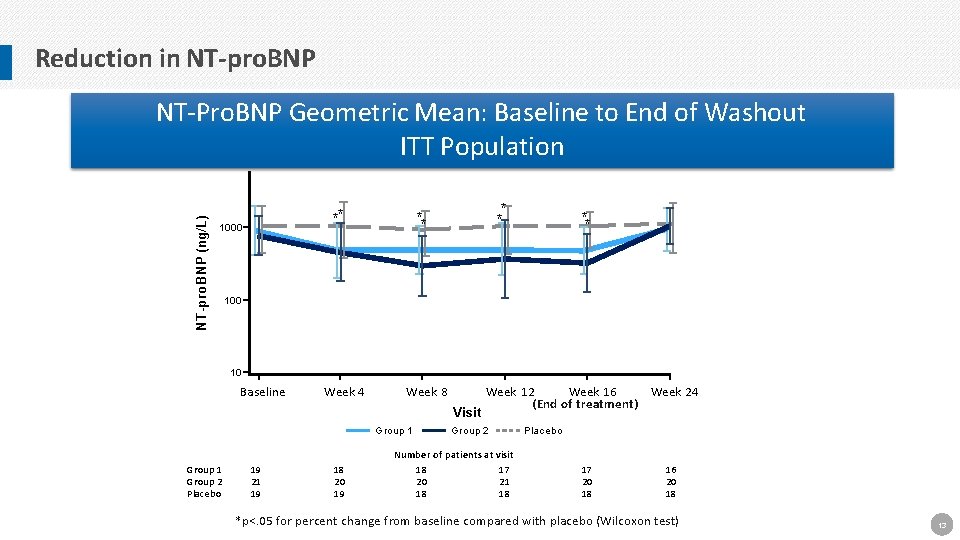
Reduction in NT-pro. BNP (ng/L) NT-Pro. BNP Geometric Mean: Baseline to End of Washout ITT Population ** 1000 * * ** ** 100 10 Baseline Week 4 Week 8 Visit Week 12 Week 16 Week 24 (End of treatment) Group 2 Group 1 Placebo Number of patients at visit Group 1 Group 2 Placebo 19 21 19 18 20 18 17 21 18 17 20 18 16 20 18 *p<. 05 for percent change from baseline compared with placebo (Wilcoxon test) 13
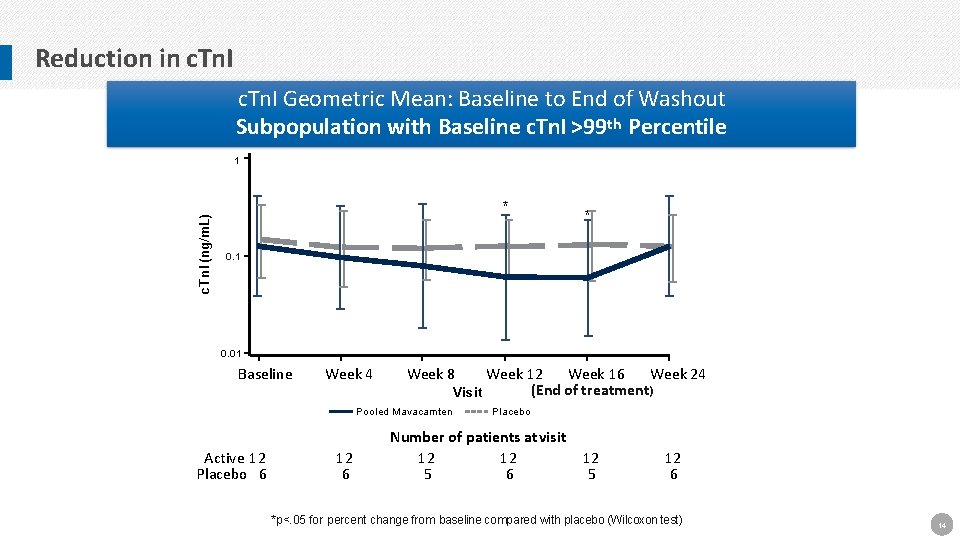
Reduction in c. Tn. I Geometric Mean: Baseline to End of Washout Subpopulation with Baseline c. Tn. I >99 th Percentile c. Tn. I (ng/m. L) 1 * * 0. 1 0. 01 Baseline Week 4 Week 8 Week 12 Week 16 Week 24 (End of treatment) Visit Pooled Mavacamten Active 12 Placebo 6 12 6 Placebo Number of patients at visit 12 12 5 6 12 5 12 6 *p<. 05 for percent change from baseline compared with placebo (Wilcoxon test) 14
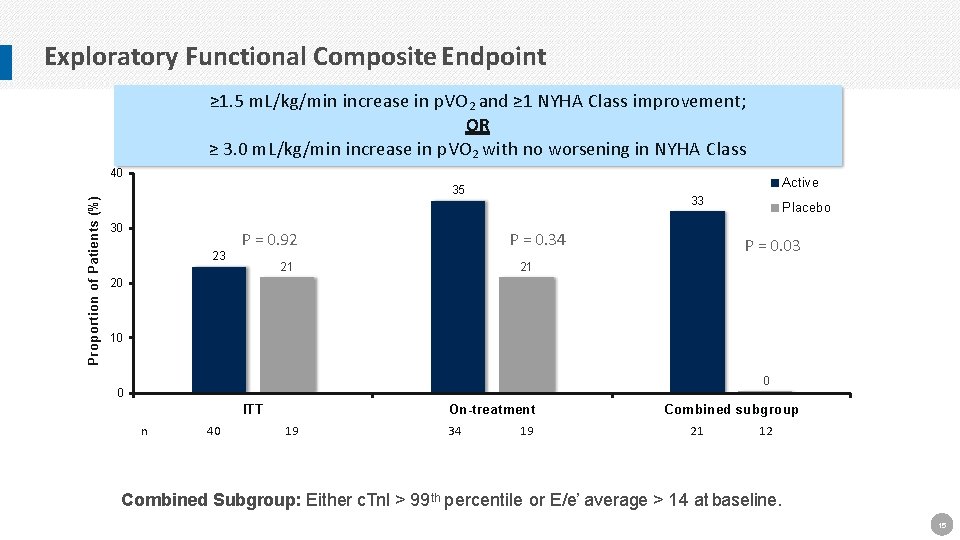
Exploratory Functional Composite Endpoint ≥ 1. 5 m. L/kg/min increase in p. VO 2 and ≥ 1 NYHA Class improvement; OR ≥ 3. 0 m. L/kg/min increase in p. VO 2 with no worsening in NYHA Class Proportion of Patients (%) 40 Active 35 30 23 P = 0. 92 33 P = 0. 34 Placebo P = 0. 03 21 21 20 10 0 0 On-treatment ITT n 40 19 34 19 Combined subgroup 21 12 Combined Subgroup: Either c. Tn. I > 99 th percentile or E/e’ average > 14 at baseline. 15
Conclusions Mavacamten was generally tolerated in most participants with non-obstructive HCM No excess of serious adverse events LVEF decreased by 4 (8)% in the pooled mavacamten group versus 2. 3 (5)% in placebo. 5 of 40 participants (12. 5%) had reversible reductions in LVEF<45% leading to protocol-driven treatment discontinuation. LVEF recovered within 4 -12 weeks. Treatment with mavacamten resulted in a dose-dependent reduction in serum levels of NT-pro. BNP and c. Tn. I suggesting physiological benefit. Exploratory analyses suggest that patients with more severe disease expression (baseline elevated c. Tn. I or E/e’) may benefit more from mavacamten therapy Biomarkers and clinical parameters will inform dosing strategies in future studies. Results set the groundwork for future, larger scale studies in n. HCM and potentially in HFp. EF.

Acknowledgments Co-authors: Matthew E. Mealiffe, MD; Richard G. Bach, MD, FACC; Mondira Bhattacharya, MD; Lubna Choudhury, MD Jay M. Edelberg, MD, Ph. D; Sheila M. Hegde, MD; Daniel Jacoby, MD, BA; Neal K. Lakdawala, MD, MSc; Steven J. Lester, MD; Yanfei Ma, MS; Ali J. Marian, MD; Sherif F. Nagueh, MD; Anjali Owens, MD; Florian Rader, MD, MSc; Sara Saberi, MD; Amy J. Sehnert, MD; Mark V. Sherrid, MD; Scott D. Solomon, MD; Andrew Wang, MD; Omar Wever-Pinzon, MD; Timothy C. Wong, MD; Stephen B. Heitner, MD Site investigators T. Abraham, K. Afshar, P. Alvarez, K. Ananthasubramaniam, R. Bach, R. Becker, L. Choudhury, R. Daggubati, S. Day, S. Heitner, D. Jacoby, C. Kramer, N. Lakdawala, S. Lester, A. Marian, J. Moses, S. Nagueh, A. Owens, D. Owens, E. Popjes, F. Rader, A. Ravichandran, S. Saberi, K. Shah, M. Sherrid, J. Shirani, J. Symanski, A. Turer, A. Wang, L. Wang, O. Wever-Pinzon, M. Wheeler, M. Wolff, T. Wong) Study coordinators, cardiac sonographers, the Myo. Kardia study team, and especially, the patients who partnered with us and their families. 17
- Slides: 17