Safe Care Everywhere Infection Prevention in Outpatient Settings















































- Slides: 56

Safe Care Everywhere: Infection Prevention in Outpatient Settings Matthew Crist, MD, MPH Medical Officer Quality Standards & Safety Team DHQP/PRB cwu 0@cdc. gov July 14, 2016 National Center for Emerging and Zoonotic Infectious Diseases Division of Healthcare Quality Promotion

Outline • Background & Vision • Outbreak Investigations • Outpatient Policy Options -- Overview • Engaging Outpatient Providers • Q&A

Germs spread… within and across health care facilities… Hospitals Dialysis facilities Outpatient facilities Longterm care 3

Vision SAFE CARE EVERYWHERE

Outpatient Settings § Provide healthcare to patients who do not remain overnight • Examples include: o Physician offices o Urgent care centers o Cancer clinics and infusion centers o Imaging centers o Alternative medicine clinics o Ambulatory surgical centers § Increasingly complex procedures, vulnerable patients • Each year more than one million cancer patients receive outpatient chemotherapy and/or radiation therapy • May provide similar services as hospitals 5

Ambulatory Care – Growth, Concerns • Approximately 1. 2 billion outpatient visits / year – average person makes four visits/year • Expansion of services without parallel increases in attention to and oversight of infection control – Many lack written infection control policies and procedures • Lack systematic surveillance to detect infections

OUTBREAK INVESTIGATIONS IN OUTPATIENT SETTINGS

CDC Healthcare Outbreak Response Activities Division of Healthcare Quality Promotion (DHQP) § Investigate infections and other adverse events related to healthcare delivery § Consults (mostly with state health departments) • Cases, clusters, infection control breaches § About one field investigation (“Epi-Aid”) per month § Medical product-associated outbreaks • Many outbreaks and clusters potentially involve contamination of devices or injectable medications

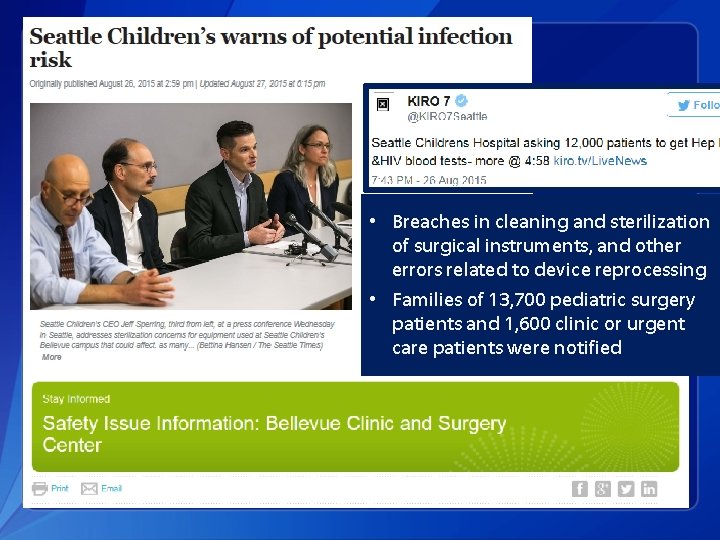
• Breaches in cleaning and sterilization of surgical instruments, and other errors related to device reprocessing • Families of 13, 700 pediatric surgery patients and 1, 600 clinic or urgent care patients were notified 9

http: //www. washingtonpost. com/national/health-science/woman-who-hadliposuction-in-baltimore-county-dies-from-infection/2012/09/19/bb 8991 de 027 d-11 e 2 -9 b 24 -ff 730 c 7 f 6312_story. html

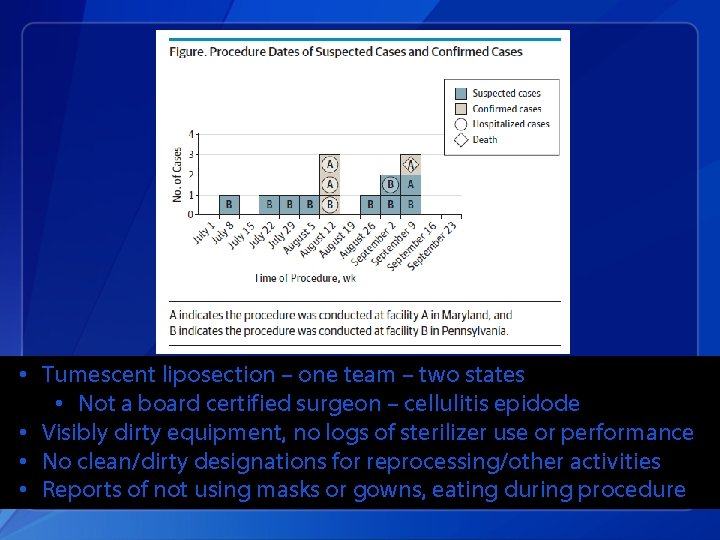
• Tumescent liposection – one team – two states • Not a board certified surgeon – cellulitis epidode • Visibly dirty equipment, no logs of sterilizer use or performance • No clean/dirty designations for reprocessing/other activities • Reports of not using masks or gowns, eating during procedure


Summary of existing guidance that provides basic infection prevention recommendations for nearly any outpatient setting www. cdc. gov/HAI/settings/outpatient/

Unsafe Injection Practices q Mishandling and inappropriate sharing of medication vials and containers § Administration of medication from a single-dose vial to multiple patients § Intravenous solution bags used as a common source of supply (e. g. , for flush) § Handled in a contaminated environment q Reuse of syringes and needles § To access shared medications (“indirect reuse” a. k. a. “double dipping”) § For multiple patients (“direct reuse”)

Injectable Medications § Safe healthcare delivery relies on supply of safe sterile injectable medications (free of intrinsic contamination ) • Oversight required to assure injectable medications are supplied in a sterile form o Majority are manufactured FDA Standards • Extrinsic contamination can result from mishandling of medications during preparation and administration CDC addresses and promotes “Injection Safety” through guidelines and educational efforts

http: //www. oneandonlycampaign. org/content/bloodbornepathogens-training

Outbreak of Pseudomonas aeruginosa and Klebsiella pneumoniae BSI, outpatient chemotherapy center • Fourteen cases (17%) identified among 84 active clinic patients • Unqualified/unlicensed staff (e. g. , administrator with “on-the-job training” provided port access and infusion services) • Cost-containment measures recently instituted • Switched to common-source 1 -L saline and 1 -L heparin flush • Bags were used over several days for multiple patients • A single syringe was dedicated to each patient that could be reused multiple times to access the common bag of saline solution • Other syringes were dedicated to drawing up heparin flush and shared among multiple patients over an indeterminate period (discarded only if visible blood) Dobbs et al, AJIC (2014) 731 -4 WJTV TV

Tsukamurella q Independent clinic located inside a hospital complex q The chemotherapy hood was adjacent to a window that was opened intermittently q Single-dose medication vials (outside of the chemotherapy hood) were stored and reused over multiple days q Nonchemotherapy medications were prepared next to a sink, which could contaminate medications with tap water q Clinical impacts • 14/15 patients hospitalized • 10 patients had to have their central lines removed

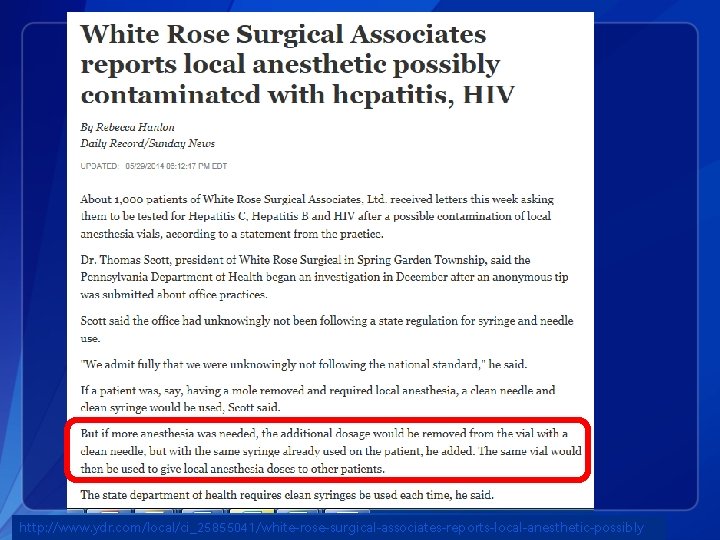
http: //www. ydr. com/local/ci_25855041/white-rose-surgical-associates-reports-local-anesthetic-possibly


Insulin Pen Reuse Incidents § Reuse of insulin pens for multiple patients, reportedly after changing needles has resulted in large notifications § NY hospital, 2008: 185 patients notified § TX hospital, 2009: 2, 114 patients notified § WI hospital and outpatient clinic, 2011: 2, 401 patients notified § 2013: multiple incidents involving NY and NC, including 2 VA Medical Centers and a private hospital Schaefer et al. Diabetes Care. 2013 Nov; 36(11): e 188 -9 www. newsday. com/news/health/south-nassau-hospital-alerting-4 -247 -patients-ofpossible-blood-contamination-1. 7359969



MSSA Cluster – Rheumatology Practice § Dec 2011: hospital IP notified health department • 4 patients admitted (LOS 1 -8 days) for surgical debridement of labconfirmed MSSA infections • HD identified 5 th patient treated at different hospital ED § Cases all received joint injections at an independent outpatient rheumatology clinic on same afternoon • 3 exam rooms; poor records management § Steroid (MPA) from a compounding pharmacy • labeled as “MDV” containing preservatives § Opened MDVs and SDVs kept on top of towel dispenser Drezner et al. Infect Control Hosp Epidemiol 2014; 35(2): 187 -189

MSSA Cluster – Rheumatology Practice § Evidence pointed to extrinsic contamination • Single dose vials used for multiple patients • Reuse of multidose vials (including the pharmacy product which was not a true MDV) stored in a contamination prone area • Failure to cleanse/disinfect vial septum with alcohol • Hand hygiene deficiencies • Refrigerators had no thermometers, were disorganized, overfilled, and poorly maintained Drezner et al. Infect Control Hosp Epidemiol 2014; 35(2): 187 -189


Diversion & Tampering Patient safety is compromised whenever diversion by healthcare personnel occurs Harms can include: § Failure to receive prescribed medication • Resulting in failure to obtain adequate pain management § Exposure to substandard care from an impaired provider § Exposure to life-threatening infections Berge KH et al. Mayo Clin Proc. 2012; 87(7): Schaefer MK, Perz JF. Mayo Clinic Proc. 2014; 89 (6)

Mechanisms of Diversion § False documentation (e. g. , medication dose not actually administered to the patient or “wasted” but instead saved for use by the provider) § Scavenging of wasted medication (e. g. , removal of residual medication from used syringes) § Theft by tampering (e. g. , removal of medication from a medication container or syringe and replacement with saline or other similarly appearing solution that may be administered to patients )

Hepatitis C outbreak, Colorado 2009 § CO Department of Public Health and Environment received 2 reports of acute HCV infection • Patients had undergone surgical procedures at same hospital § § § HCV-infected surgical technician stole fentanyl syringes that had been predrawn by anesthesia staff and left unlocked in the OR Tech refilled contaminated syringes with saline to swap with additional fentanyl syringes At least 18 patients infected; >8, 000 patients notified • Notification included ASC that employed tech after she was fired from hospital and NY hospital where tech worked prior to the CO hospital § Tech sentenced to 30 -year prison term http: //www. mayoclinicproceedings. org/article/S 0025 -6196(14)00342 -5/fulltext

http: //www. denverpost. com/news/ci_12790134

http: //www. cdc. gov/injectionsafety/drugdiversion/

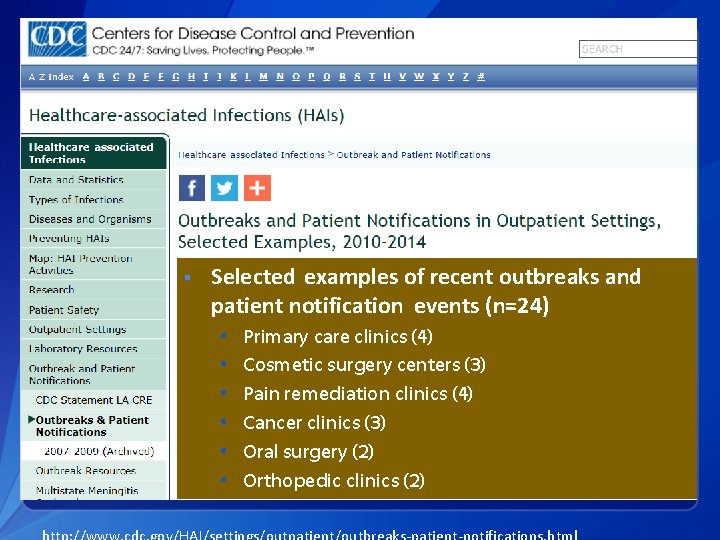
§ Selected examples of recent outbreaks and patient notification events (n=24) • • • Primary care clinics (4) Cosmetic surgery centers (3) Pain remediation clinics (4) Cancer clinics (3) Oral surgery (2) Orthopedic clinics (2)

POLICY OPTIONS IN OUTPATIENT SETTINGS


Outpatient Settings Policy Options: Four Key Policy Elements Consider more effective and proactive oversight of outpatient facilities through: (1) Facility licensing/accreditation (2) Provider training, licensing and certification (3) Reporting requirements (4) Investigation authorities

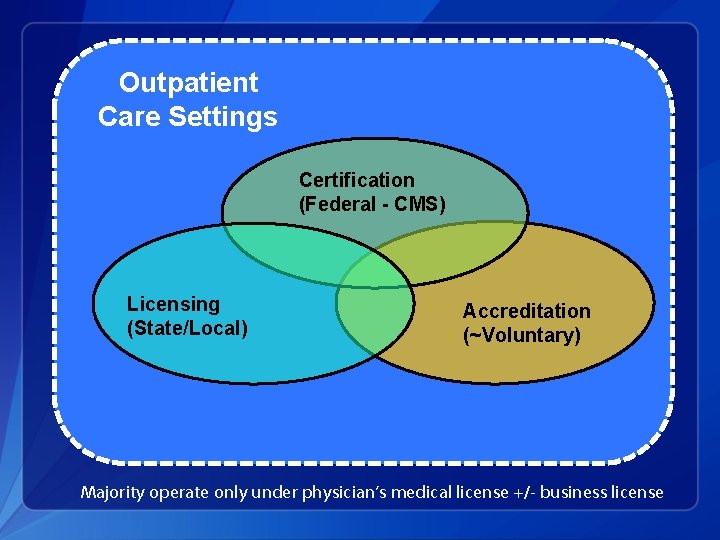
(1) Facility Licensing and Accreditation § Few outpatient healthcare facilities • Certified by CMS • Licensed by states • Maintain accreditation status

Outpatient Care Settings Certification (Federal - CMS) Licensing (State/Local) Accreditation (~Voluntary) Majority operate only under physician’s medical license +/- business license

(1) Facility Licensing and Accreditation § Few outpatient healthcare facilities • Certified by CMS • Licensed by states • Maintain accreditation status § Most lack an infection control plan and are not held to minimum safety standards for infection prevention § § Not subject to routine survey/inspections (vs. restaurants) Basic infection control practices as outlined in Standard Precautions should be the minimum expectation • CDC Guide to Infection Prevention in Outpatient Settings

Guide to Infection Prevention for Outpatient Settings: Minimum Expectations for Safe Care Summary of infection prevention recommendations & guidelines - Ensure that an outpatient facility has appropriate infection prevention policies and procedures to provide safe care Checklist: - Systematically assess infection prevention practices and competencies Version 2. 2 - November 2015

(1) Facility Licensing and Accreditation -- Considerations -§ Maintaining accurate information on the locations, numbers, and types of facilities § Registration requirements • Accreditation requirements • Designation of an infection control coordinator § Enhancing collaboration with medical boards, licensure agencies, pharmacy boards and accrediting agencies § Designating fees or fines for the support of education, technical assistance, and inspection/monitoring § Using CDC-developed survey tools for expanded on-site assessments

(2) Provider Training, Licensure and Certification § Requirements for healthcare providers highly variable • Licensure generally clear for physicians and nurses; less so for allied health professionals § Allied health professionals may receive minimal formal infection control training • Annual OSHA blood borne pathogen training not enough § Delegation of healthcare delivery activities loosely governed by state practice standards

Provider Training, Licensure and Certification -- Considerations -§ Expanding licensure and certification requirements § Requiring training in HAI prevention and infection control as a condition of licensure or certification § Minimum levels of periodic continuing education credits related to infection control training § Requiring state-approved basic infection control training for anyone engaged in healthcare work under the supervision of a licensed healthcare professional, including workers not licensed by the state

JOE SOMEBODY

SOMEONE SOMEBODY


(3) Reporting Requirements § HAI and infectious disease reporting requirements variable across the 50 states • What, when and how to report • Healthcare provider vs. health facility vs. laboratory § Outbreak definitions variable • Just one HAI-associated case of certain diseases can signal serious problems § Other types of adverse events which may pose serious infection risks § Clear direction from state health departments is beneficial Partner with acute care IPs § • “See Something, Say Something”

(4) Investigation Authorities § State and local health departments require firm legal foundation to investigate: • Potential disease outbreaks • Cases of reportable disease • Other breakdowns in care delivery which may pose infection risk § Authority should clearly apply to all types of outpatient healthcare facilities § Shared authorities should be understood and discussed

Outpatient Settings Policy Options: Available for Download URL: http: //www. cdc. gov/hai/pdfs/prevent/Outpatient-Settings-Policy. Options. pdf

ICAR Activity A A. 1: Expand State HAI Plan and Advisory Group q Incorporate concept of on-site assessment, gap-assessment, spectrum of healthcare q Add partners including ASPR-funded Hospital Preparedness Program (HPP); others A. 2: Improve coordination between DOH and healthcare settings q Inventory of facilities, by type of setting q Identify regulatory/licensing and other oversight authorities q Infection control capacity and competency requirements A. 3: Assess readiness of designated Ebola facilities q Conduct on-site assessments of all designated Ebola assessment hospitals (or treatment centers, if any) q Determine gaps in readiness q Address gaps through consultation/training using CDC-based resources; develop mitigation and implement plan with hospital q Follow up to confirm mitigation of gaps A. 4: Assess and improve HAI outbreak reporting and response • Evaluate capacities to detect, report and respond • Standardize an approach to assessment • Across setting types

ENGAGING OUTPATIENT PROVIDERS (CSTE Roundtable Participants, We Heard You )

• Create model infection control plans and expand uptake of these and other infection prevention guidance materials in outpatient settings • Work with 12 outpatient-focused professional organizations (e. g. , medical specialties that primarily serve ambulatory patient populations), and 10 ambulatory health care systems


Activity B. 1 Outpatient Facility Engagement § A total of 26 health departments that received Activity B funding indicated they would target outpatient facilities • >1, 800 total assessments § Consider selecting/prioritizing assessments based on: • Types of facilities that have had outbreaks or complaints • Facilities that perform invasive procedures (e. g. , epidural injections, surgeries, endoscopies, chemotherapy) o Initial focus on those that have never received any type of certification, accreditation, or licensing visit 7


Thank You! cwu 0@cdc. gov For more information please contact Centers for Disease Control and Prevention 1600 Clifton Road NE, Atlanta, GA 30333 Telephone: 1 -800 -CDC-INFO (232 -4636)/TTY: 1 -888 -232 -6348 Visit: www. cdc. gov | Contact CDC at: 1 -800 -CDC-INFO or www. cdc. gov/info The findings and conclusions in this report are those of the authors and do not necessarily represent the official position of the Centers for Disease Control and Prevention. National Center for Emerging and Zoonotic Infectious Diseases Division Name in this space

Questions / Discussion