SABR in lung cancer Treatment of early stage

SABR in lung cancer

�Treatment of early stage disease (stage 1 disease) �Suitable for medically inoperable patients/dependent on patient choice
Inclusion criteria for SABR �MDT diagnosis of NSCLC- +histology/positive PET/growth on serial CTs �Clinical stages of T 1/T 2 (<5 cm)/T 3 (<5 cm) N 0 M 0 �Not suitable for surgery �PS 0 -2 �Peripheral lesions outside a 2 cm radius of main airways and proximal bronchial tree.

Exclusion criteria �Metastatic lung tumours �Tumour that cannot be clinically defined �If tumour has respiratory motion > 1 cm �Previous radiotherapy within planned treatment volume �Presence of pulmonary fibrosis �Chemotherapy within or planned within 6 weeks �Pregnant/lactating females �Inability to consent or comply with treatment
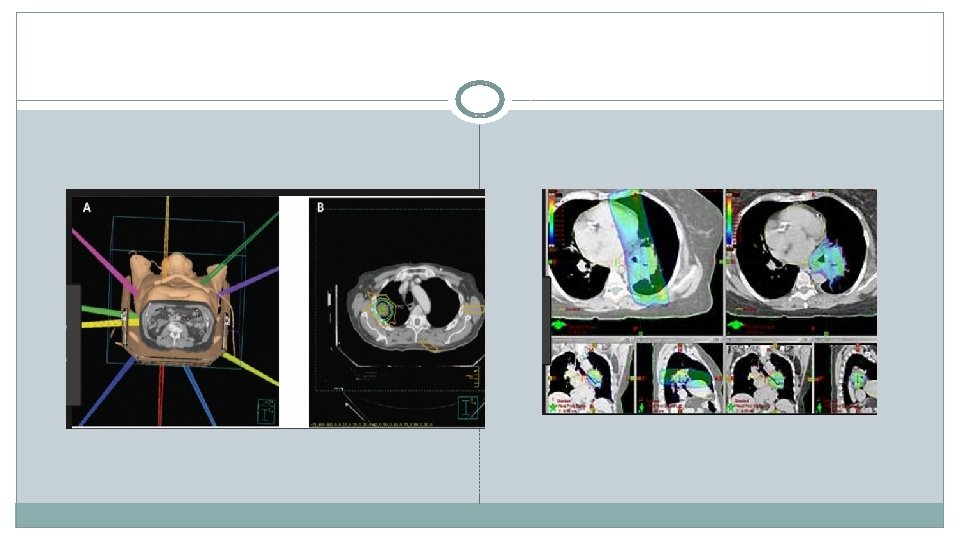
External bean radiation Stereotactic Ablative Body Radiotherapy (SABR) � 5 year survival rates of 10 -30% � Non- randomised evidence �High local recurrence rates (up supporting SABR as superior to conventional RT or surgery �Fewer side effects � 3 x week for 2 weeks to a third) �Higher dose radiation associated with more side effects �More frequent sessions

Surgery vs SABR �Comparable 1 and 3 year survival (surgery vs SABR) in COPD patients. Better 30 day mortality in COPD patients (10% in surgical group vs 0% in SABR group)- Palma et al, 2012 �Surgical resection of larger tumours vs SABR for larger tumours (3 year survival of 90% vs 80%)- Subroto et al, 2016 �Meta-analysis of 40 SBRT studies and 20 surgical studies- Adjusted for age and operability. Survival outcomes not significantly different between the groups. 5 year estimates for overrall survival 82% (SABR), 66% (limited lung resection), 68% (lobectomy) – Zheng at al, 2014
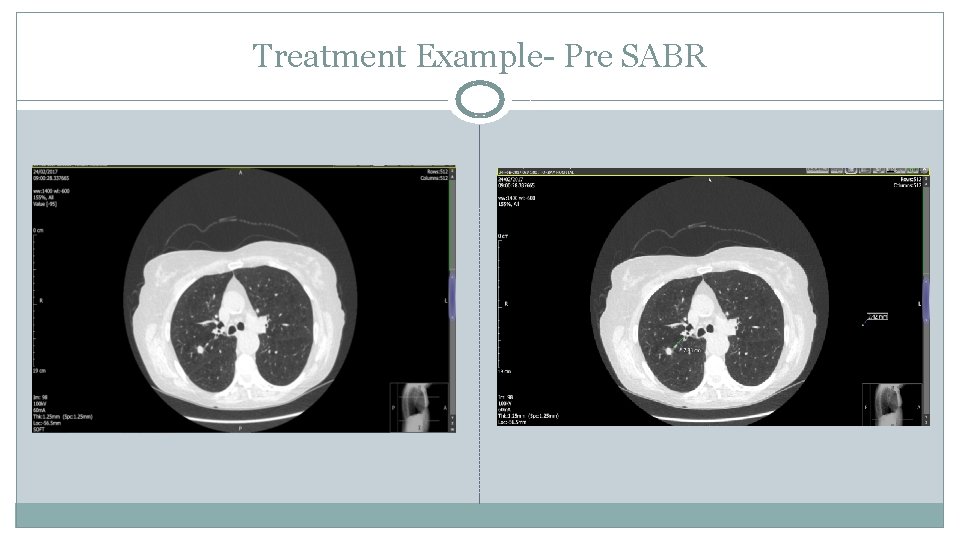
Treatment Example- Pre SABR
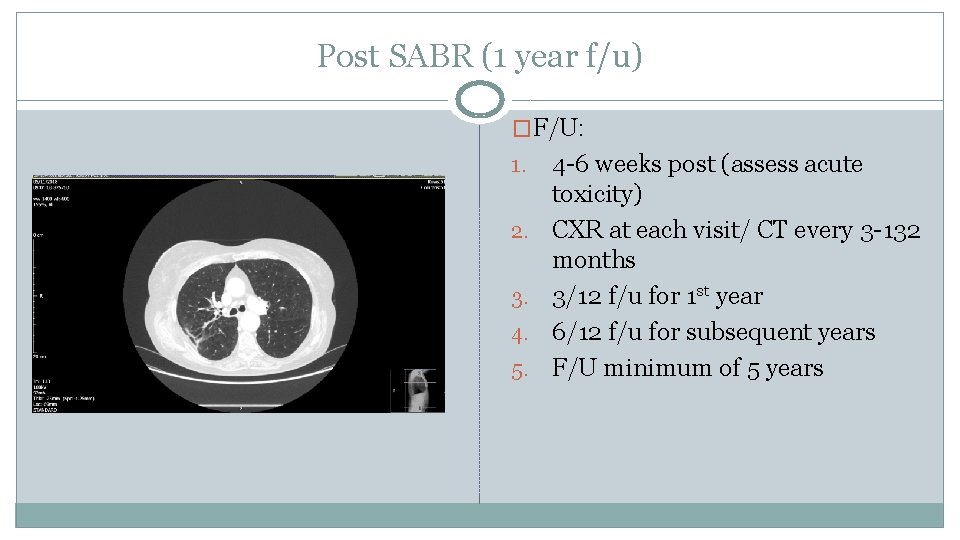
Post SABR (1 year f/u) �F/U: 1. 2. 3. 4. 5. 4 -6 weeks post (assess acute toxicity) CXR at each visit/ CT every 3 -132 months 3/12 f/u for 1 st year 6/12 f/u for subsequent years F/U minimum of 5 years
- Slides: 9