S 3 BGE Chemistry Chemical Changes and Structure

S 3 BGE Chemistry Chemical Changes and Structure ACID SPILL 02/11/2020

Lesson 1 Acids and Alkalis 02/11/2020

Common acids and alkalis 02/11/2020 We are learning to: • Recognise some common acids and alkalis in the lab and at home. I can: • Name some examples of household acids and alkalis • State the chemical formula of common lab acids and alkalis.

Lesson Starter: What do you already know about Acids and Alkalis? Discuss with your partner what you already know about acids and alkalis Write down any facts you know about acids and alkalis 02/11/2020

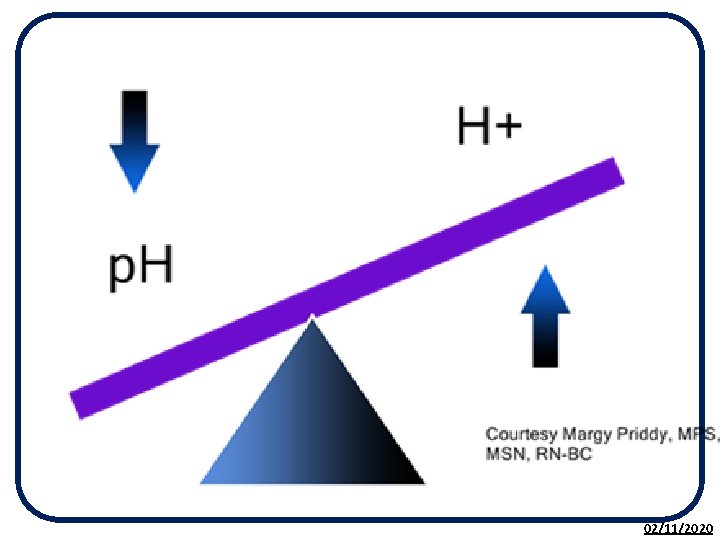
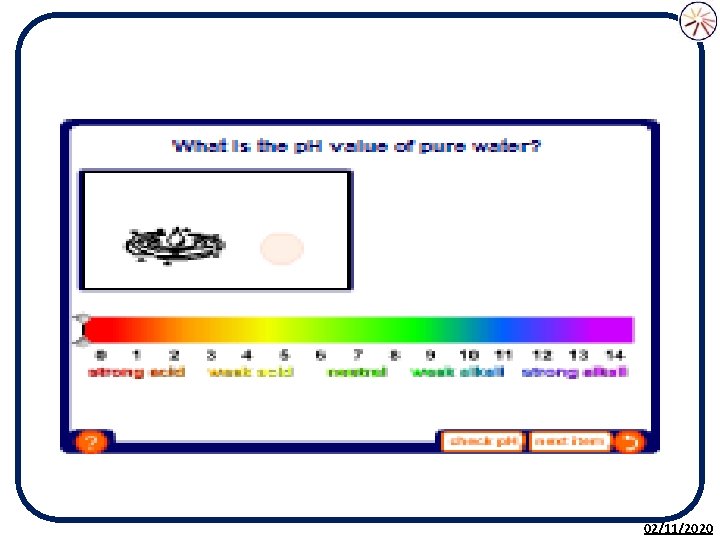
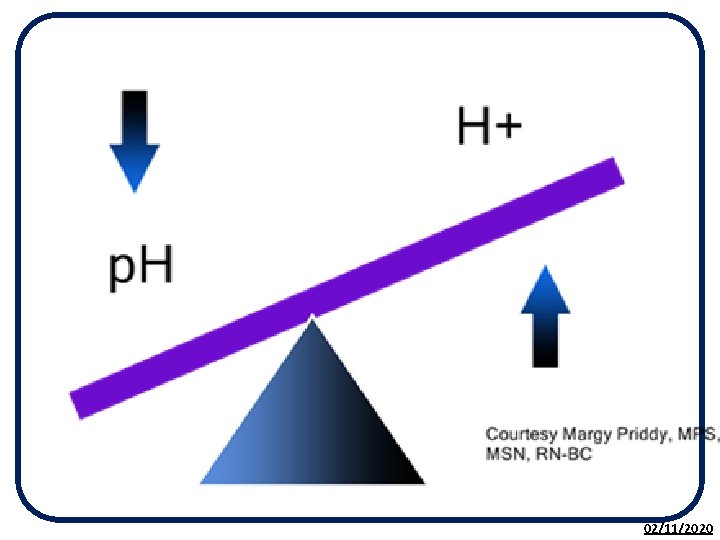
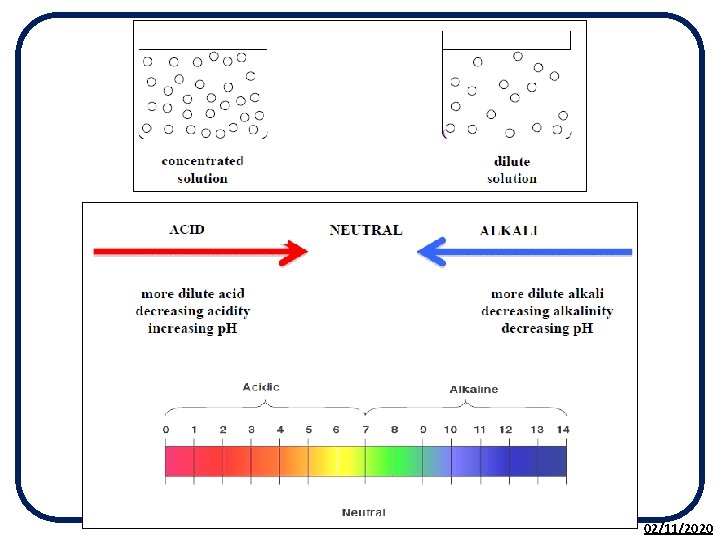
The p. H Scale • The p. H scale is used to measure the acidity or alkalinity of a solution. • p. H is a measure of the H+ (hydrogen) ion concentration in a solution. • It is based on the p. H of pure water being 7. • The p. H scale is a continuous range from less than 0 to above 14. 02/11/2020
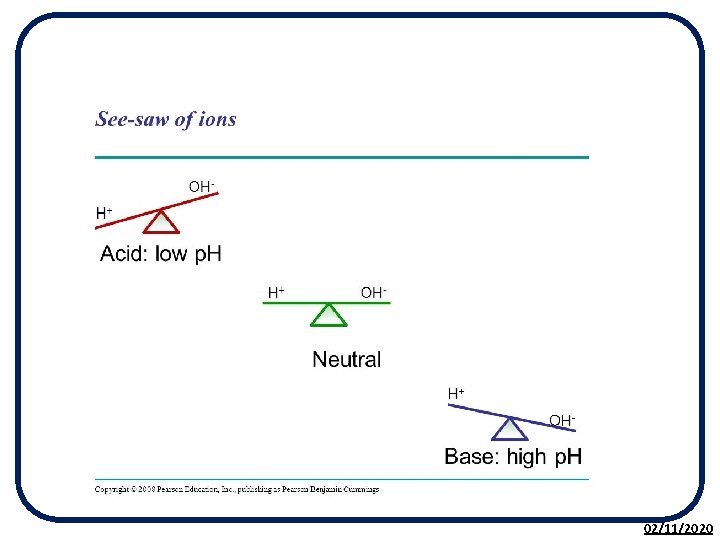
02/11/2020
02/11/2020

Measuring p. H There are three ways we can determine the p. H of a substance: 1. p. H paper 2. Universal indicator 3. Digital p. H meters This will determine if a substance is acidic, alkaline or neutral 02/11/2020

Household Acids 1. Vinegar 2. Lemonade or any fizzy drink 3. Soda water (fizzy water) 02/11/2020

Household Alkalis 1. Baking soda 2. Oven cleaner 3. Dishwashing powder 4. Bleach 02/11/2020
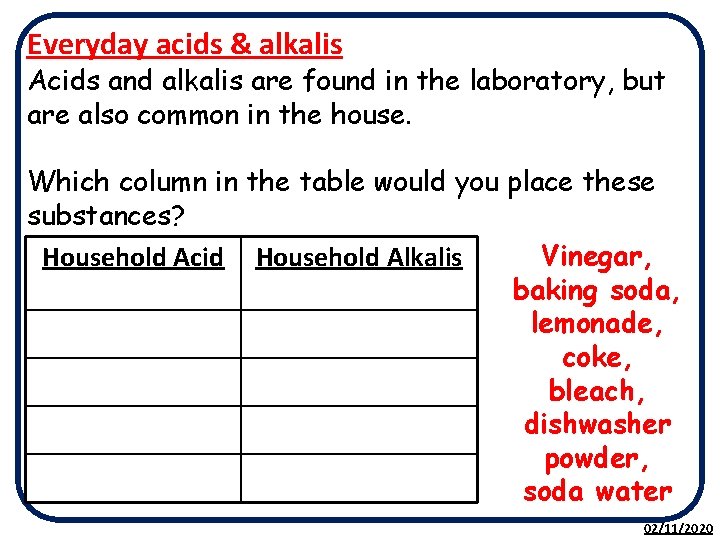
Everyday acids & alkalis Acids and alkalis are found in the laboratory, but are also common in the house. Which column in the table would you place these substances? Vinegar, Household Acid Household Alkalis baking soda, lemonade, coke, bleach, dishwasher powder, soda water 02/11/2020
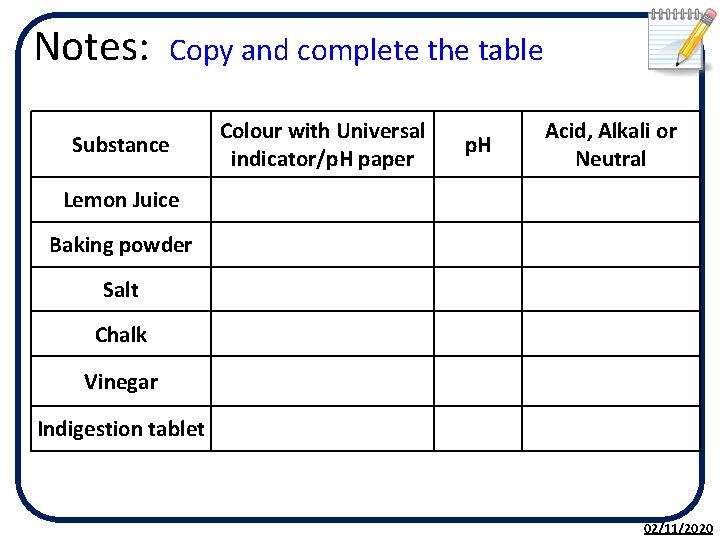
Notes: Copy and complete the table Substance Colour with Universal indicator/p. H paper p. H Acid, Alkali or Neutral Lemon Juice Baking powder Salt Chalk Vinegar Indigestion tablet 02/11/2020
02/11/2020

Research Task After discussion with your teacher and others, find suitable sources of information on the use of acids in the food industry such as ethanoic acid, citric acid and benzoic acid. Produce a public information pamphlet to help the public be aware of the benefits of acids in the food and drinks industry. 02/11/2020

Lesson 2 How are Acids made? What is Acid rain? 02/11/2020

Non-metal Oxides 02/11/2020 We are learning to: • understand how acids can be made • What is acid rain and what effect does it have? I can: • Test the p. H of some non-metal oxides • Write word equations and balanced chemical equations for the formation of some non-metal oxides. • Describe the effects of acid rain.

Experiment: Non-Metal Oxides ACT. 5. 1 • Using a straw carefully blow into a test tube containing a small amount (1 cm depth) of water with a few drops of Universal indicator. • Note the colour change. • Your teacher may show you what happens when sulfur is burned and the gas produced is dissolved in water containing Universal indicator. • They may also show what happens when air is sparked causing nitrogen and oxygen to combine. • Note the colour change each time. 02/11/2020
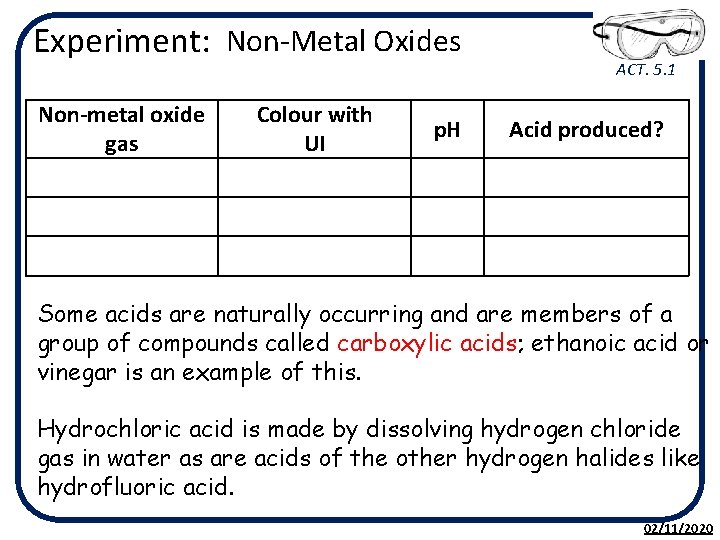
Experiment: Non-Metal Oxides Non-metal oxide gas Colour with UI ACT. 5. 1 p. H Acid produced? Some acids are naturally occurring and are members of a group of compounds called carboxylic acids; ethanoic acid or vinegar is an example of this. Hydrochloric acid is made by dissolving hydrogen chloride gas in water as are acids of the other hydrogen halides like hydrofluoric acid. 02/11/2020

Acid Rain The formation of oxides of carbon, sulfur and nitrogen are associated with burning fossil fuels. When these dissolve in water in the atmosphere they form acid rain. 02/11/2020

Sources of Carbon Dioxide Carbon dioxide is produced when any compound containing carbon burns (reacts with the oxygen in the air). Other sources of carbon dioxide include the manufacture of cement. 02/11/2020

Non-metal Oxide Pollutants SULPHUR DIOXIDE is also produced when some fuels are burned. This then dissolves in atmospheric water and falls as ACID RAIN. 02/11/2020

Other Non-metal Oxide Pollutants N 2 NO 2 NO NO 2 NO NO NO O 2 2 NITROGEN DIOXIDE is produced when the spark plug in a car provides enough energy for nitrogen and oxygen to react. These oxides can dissolve in atmospheric water (clouds) and fall as ACID RAIN. 02/11/2020

Acid rain has many damaging effects: • Corrosion of buildings made of iron and steel • Erosion of buildings made from carbonate rocks • Increases the acidity of soil • Harmful to plant and animal life 02/11/2020

Write word equations and balanced chemical equations for the formation of the following gases: Sulphur dioxide, Nitrogen dioxide and Carbon dioxide. • Sulphur Dioxide 02/11/2020

Write word equations and balanced chemical equations for the formation of the following gases: Sulphur dioxide, Nitrogen dioxide and Carbon dioxide. • Nitrogen dioxide 02/11/2020

Write word equations and balanced chemical equations for the formation of the following gases: Sulphur dioxide, Nitrogen dioxide and Carbon dioxide. • Carbon dioxide 02/11/2020

RULE: Lear n! Oxides of NON-METALS which dissolve in water make ACIDIC SOLUTIONS. If a non-metal oxide does not dissolve in water it will have no effect on the p. H of water. EXCEPTION: Oxide of HYDROGEN is NEUTRAL (water) 02/11/2020
REMEMBER!! Solubilites of selected compounds in water can be found on page 8 of the data booklet! 02/11/2020

Lesson 3 How are Alkalis made? 02/11/2020

Metal Oxides 02/11/2020 We are learning to: . • understand how alkalis can be made I can: • Test the p. H of some metal oxides • Write word equations and balanced chemical equations for the formation of some metal oxides.
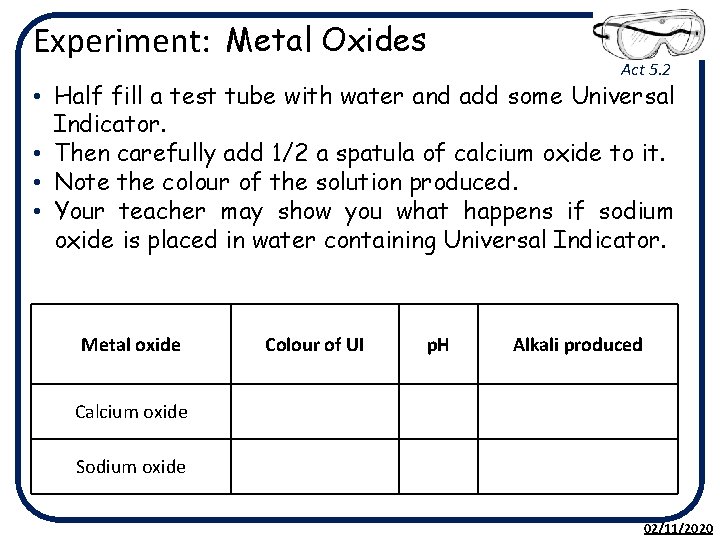
Experiment: Metal Oxides Act 5. 2 • Half fill a test tube with water and add some Universal Indicator. • Then carefully add 1/2 a spatula of calcium oxide to it. • Note the colour of the solution produced. • Your teacher may show you what happens if sodium oxide is placed in water containing Universal Indicator. Metal oxide Colour of UI p. H Alkali produced Calcium oxide Sodium oxide 02/11/2020

Notes: When metal oxides dissolve in water they form alkalis. Ca. O(s) + H 2 O(l) ® Ca 2+(aq) + 2 OH-(aq) Write a balanced chemical equation for the reaction of sodium oxide with water: 02/11/2020
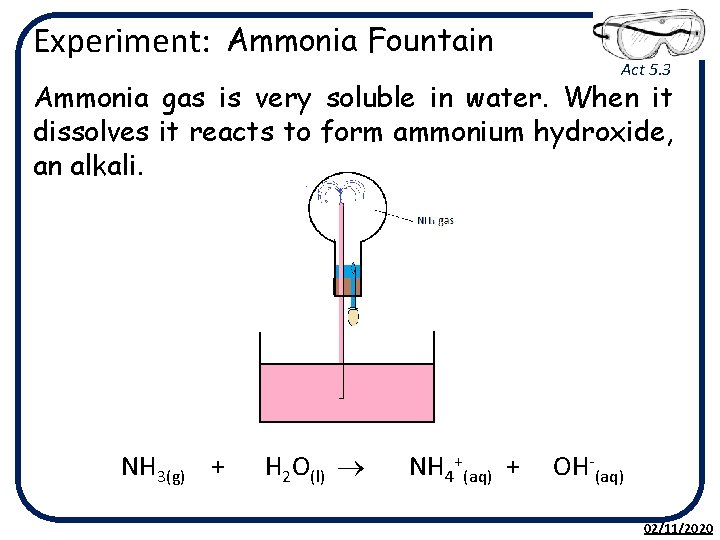
Experiment: Ammonia Fountain Act 5. 3 Ammonia gas is very soluble in water. When it dissolves it reacts to form ammonium hydroxide, an alkali. NH 3(g) + H 2 O(l) ® NH 4+(aq) + OH-(aq) 02/11/2020

Experiment: Insoluble Oxides Act 5. 4 • Half fill a test tube with water and add some Universal Indicator. • Then carefully add 1/2 a spatula of silicon dioxide to it. • Note the colour of the solution produced. • Repeat the experiment but add 1/2 a spatula of copper(II) oxide. • You will need to leave the copper oxide to settle to see the colour of the Universal indicator. Oxide Colour of Universal indicator p. H Silicon dioxide Copper(II) oxide 02/11/2020

RULE: Lear n! Oxides of METALS which dissolve in water make ALKALINE SOLUTIONS. If a metal oxide does not dissolve in water it will have no effect on the p. H of water. REMEMBER: Solubilites of selected compounds in water can be found on page 8 of the data booklet! 02/11/2020
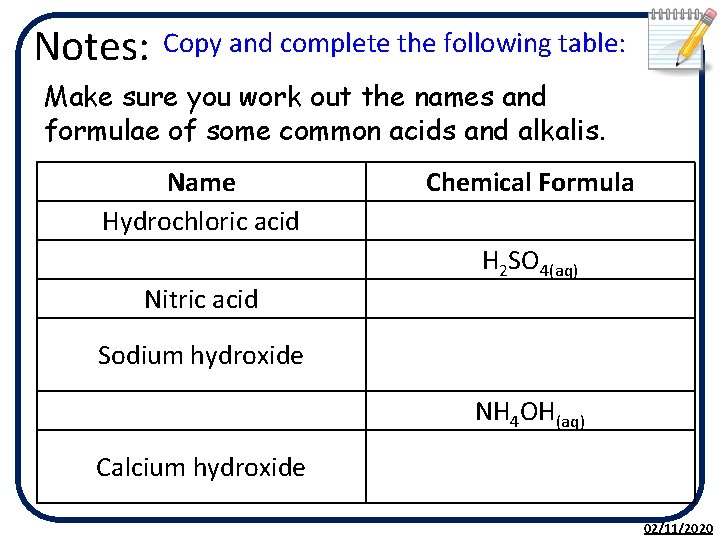
Notes: Copy and complete the following table: Make sure you work out the names and formulae of some common acids and alkalis. Name Hydrochloric acid Nitric acid Chemical Formula H 2 SO 4(aq) Sodium hydroxide NH 4 OH(aq) Calcium hydroxide 02/11/2020

Practice Question Do Quick Test 1 on page 8 of the Kitchen Chemistry workbook 02/11/2020

Practice Question (You may wish to use your data booklet) 1. Which of the following oxides would dissolve in water to produce an acidic solution? A. sulfur dioxide B. iron(III) oxide C. copper(II) oxide D. sodium oxide 2. Which of the following oxides would dissolve in water to produce an alkaline solution? A. sulfur dioxide B. iron(III) oxide C. copper(II) oxide D. sodium oxide 3. Which of the following oxides would produce a neutral solution in water? A. NO 2 B. Si. O 2 C. Li 2 O D. SO 2 02/11/2020

Practice Question 4. What colour would Universal indicator turn in a solution made by dissolve lithium oxide in water? A. Green B. Purple C. Red D. Orange 5. What colour would Universal indicator turn in a solution made by dissolve carbon dioxide in water? A. Green B. Purple C. Blue D. Orange 02/11/2020

Lesson 4 Bonding in Acids 02/11/2020

Lesson Starter: Write the chemical formula for • • • Hydrochloric acid Sulphuric acid Sodium hydroxide State whether the following give acidic or alkaline solutions. • • • nitrogen dioxide sodium oxide copper oxide 02/11/2020

Bonding in Acids 02/11/2020 We are learning to: • explain why acids conduct electricity and why hydrogen always forms at the negative electrode. I can: • explain why acids conduct electricity and why hydrogen always forms at the negative electrode.
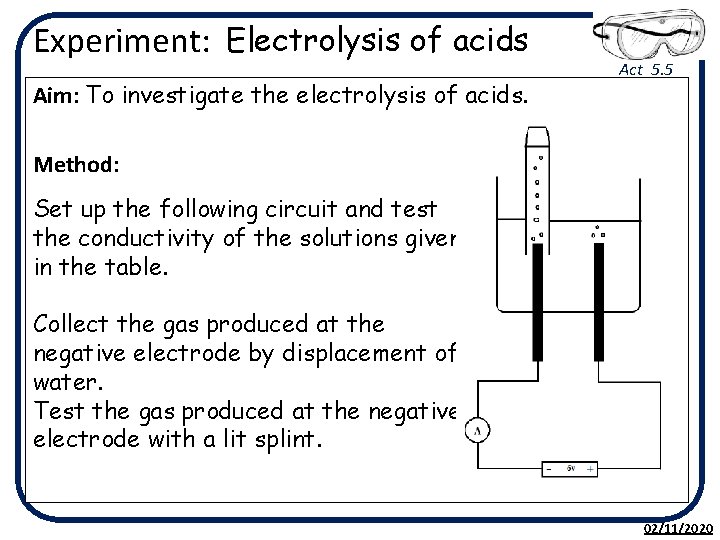
Experiment: Electrolysis of acids Aim: To investigate the electrolysis of acids. Act 5. 5 Method: Set up the following circuit and test the conductivity of the solutions given in the table. Collect the gas produced at the negative electrode by displacement of water. Test the gas produced at the negative electrode with a lit splint. 02/11/2020
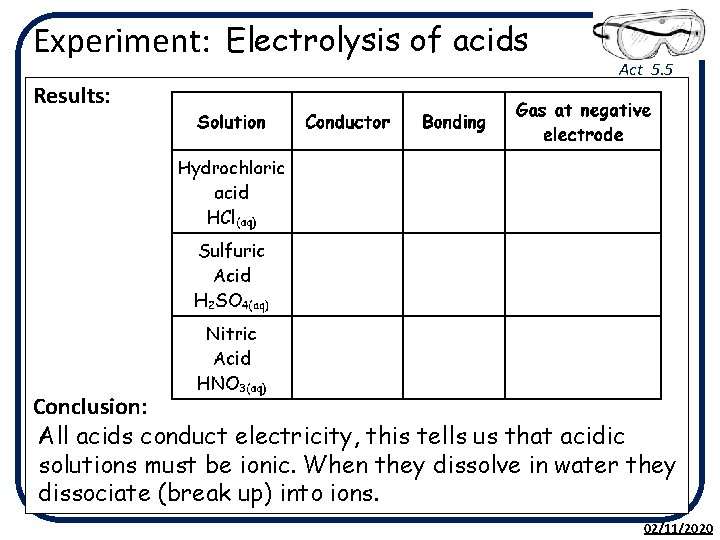
Experiment: Electrolysis of acids Act 5. 5 Results: Conclusion: All acids conduct electricity, this tells us that acidic solutions must be ionic. When they dissolve in water they dissociate (break up) into ions. 02/11/2020

Notes: • This means that the hydrogen in an acid must exist as positive hydrogen ions, H+(aq). • At the negative electrode, the hydrogen ions, 2 H+(aq) gain electrons to form uncharged hydrogen molecules. 2 H+(aq) + 2 e- H 2(g) • It is the presence of aqueous hydrogen ions, H+(aq) that makes a solution an acid. Acidic solutions contains more hydrogen ions than hydroxide ions. 02/11/2020
02/11/2020

Lesson 5 Dilution of Acids 02/11/2020

Rainbow burette • Your teacher may demonstrate the p. Hscale using a mix of acid, alkali and universal indicator 02/11/2020

Experiment: p H Rainbow 02/11/2020

Dilution 02/11/2020 We are learning to: • Dilute acids and alter p. H I can: • Complete the practical safely producing a range of colours representing the p. H scale

Experiment: Diluting Acids Act 5. 6 1. Carefully measure 10 cm 3 of 0. 1 moll-1 hydrochloric acid into a test tube. 2. Carefully measure 1 cm 3 of the acid from this test tube into a new test tube, then add exactly 9 cm 3 of water to it. 3. Carefully measure 1 cm 3 of the acid from this test tube into a new test tube, then add exactly 9 cm 3 of water to it. 4. Repeat another four times. You should now have 7 test tubes of acid each 10 x weaker than the one next to it. 5. Add 10 drops of Universal indicator to each and mix thoroughly. 02/11/2020

Diluting Acids Dilute p. H 1 High H+ concentration p. H 7 Low H+ concentration As an acid is diluted, its p. H increases towards p. H 7. This is due to the DECREASE in H+ ion concentration. 02/11/2020

Experiment: Diluting Alkalis Act 5. 6 1. Carefully measure 10 cm 3 of 0. 1 moll-1 Sodium hydroxide into a test tube. 2. Carefully measure 1 cm 3 of the alkali from this test tube into a new test tube, then add exactly 9 cm 3 of water to it. 3. Carefully measure 1 cm 3 of the alkali from this test tube into a new test tube, then add exactly 9 cm 3 of water to it. 4. Repeat another four times. You should now have 7 test tubes of alkali each 10 x weaker than the one next to it. 5. Add 10 drops of Universal indicator to each and mix thoroughly. 02/11/2020

Diluting Alkalis Dilute p. H 7 Low OHconcentration p. H 14 High OH- concentration As an alkali is diluted, its p. H decreases towards p. H 7. This is due to the DECREASE in OH- ion concentration. 02/11/2020

The p. H Scale Putting both experiments together we get the full p. H scale. 02/11/2020
02/11/2020

Water The chemical name for water is hydrogen oxide What would you expect the bonding to be in water? Covalent bonding should not conduct electricity. Water DOES conduct electricity For water to conduct, there MUST be charged particles The charged particles present are IONS 02/11/2020
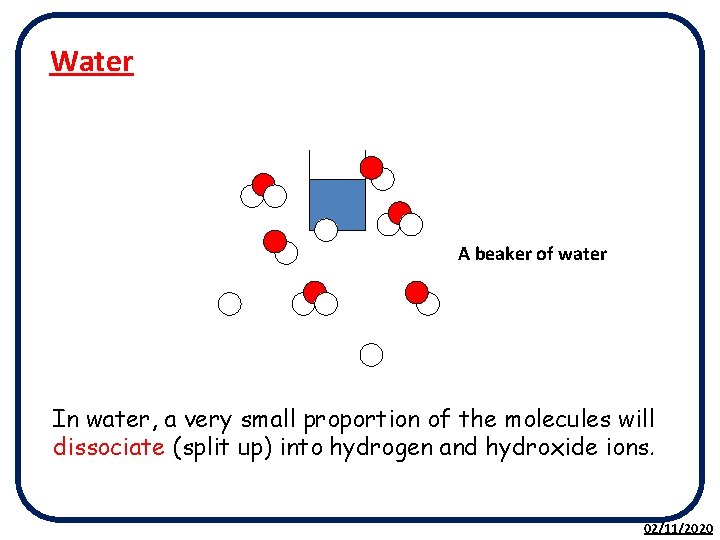
Water A beaker of water In water, a very small proportion of the molecules will dissociate (split up) into hydrogen and hydroxide ions. 02/11/2020

Water molecules dissociate (split up) into ions: H 2 O(l) H+(aq) + OH-(aq) However this process is REVERSIBLE. At the same time as ions are forming, ions are reforming into water molecules H+(aq) + OH-(aq) H 2 O(l) The rate at which the water molecules dissociate is equal to the rate at which ions react to form molecules, therefore the concentration of ions and molecules is CONSTANT. 02/11/2020

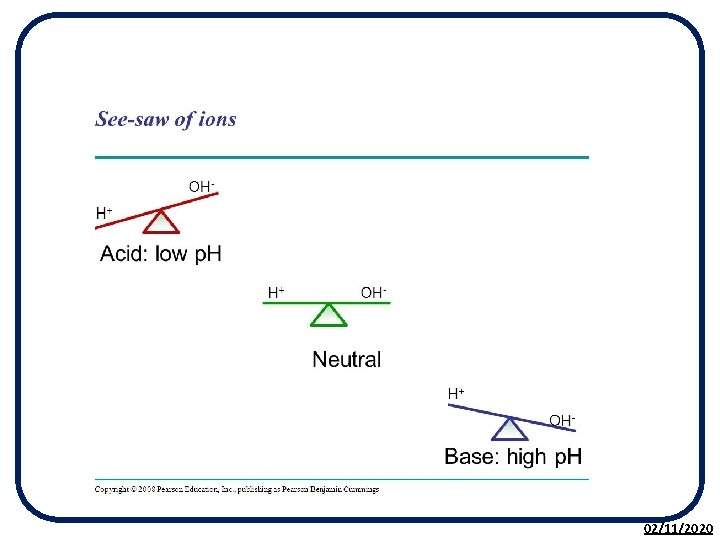
H+(aq) and OH-(aq) Ions The p. H of a solution can be described by the concentration of H+ and OH- ions: NEUTRAL solutions contain an EQUAL concentration of H+(aq) and OH-(aq) ions. ACIDIC solutions contain a GREATER concentration of H(aq) than OH(aq) ions. ALKALINE solutions contain a GREATER concentration of OH(aq) than H(aq) ions. 02/11/2020
02/11/2020

Lesson 6 Properties of Acids 02/11/2020

Lesson Starter: . . . What can you say about the concentrations of H+ and OH- ions in water? In an Acidic solution? In an Alkaline solution 02/11/2020

02/11/2020 We are learning to: • Describe the corrosive properties of common acids in the lab I can: • Complete the practical work involving acids

Properties of acids Acids are corrosive, that means that when they react with something solid they will break up that substance causing the solid to be damaged. All acids can be corrosive if they are concentrated enough. 02/11/2020

activity • ACTIVITY 5. 7 Corrosive Acids • 1. Put a piece of limestone or marble in a small beaker and cover it with 1 moll-1 hydrochloric acid, watch what happens at the surface. What happens to the size of it? • 2. Put a piece of magnesium metal into a small beaker and cover it with 1 moll-1 hydrochloric acid, watch what happens to it. • 3. Place a piece of meat in a small beaker and cover it with 1 moll-1 hydrochloric acid and leave it over night. What happens? • 4. If you have a camera you could take a series of photos of each of these experiments to show the changes that occur as the acid reacts with the surface of the solid over time. 02/11/2020
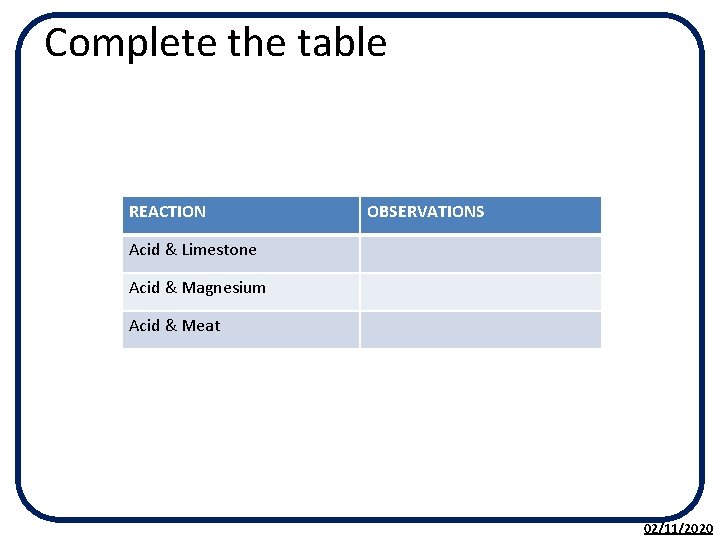
Complete the table REACTION OBSERVATIONS Acid & Limestone Acid & Magnesium Acid & Meat 02/11/2020

Notes: • Our stomachs contain acids which kill dangerous bacteria and also help to break down proteins such as meat. Bacteria in our mouths feed on sugars and produce acids which can erode our teeth. • Even dilute acids can cause harm. Acid rain is formed when carbon dioxide, sulphur dioxide and oxides of nitrogen, formed by the burning of fossil fuels in cars and lorries, dissolve in rain water, causing millions of pounds worth of damage every year. 02/11/2020

Research task- effects of acid rain RESEARCH TASK After discussion with your teacher and others, find suitable sources of information on the cause and effects of acid rain. On buildings On fish On iron structures On trees 02/11/2020

Lesson 7 Neutralisation reactions Metal oxides 02/11/2020

02/11/2020 We are learning to: • I can list the products of a neutralisation reaction I can: Successfully conduct a neutralisation reaction

Notes: • In order to reduce the amount of damage caused by an acid spill, fire fighters need to try and reduce the concentration of the acid. They can do this in two ways: • �Diluting the acid with lots of water • �Neutralising the acid. • Diluting an acid will make the acid less corrosive as its concentration decreases and the p. H rises towards 7. You have already seen this in Activity 5. 5. 02/11/2020

Notes: • When an acid is neutralised by an alkali a reaction occurs between the two ions forming water. • H+(aq) + OH-(aq)→ H 2 O(l) • This means that the amount of hydrogen ions decreases until the number of hydrogen ions is the same as in water. • There a number of types of chemical that can neutralise an acid and these are generally called bases. There are three types of base that we shall look at: • �Metal oxides • �Metal carbonates • �Metal hydroxides(alkalis) 02/11/2020

Notes: • When an acid is neutralised by a base, a salt plus water is always formed. • Salts are formed by the metal ion from the base, either a metal hydroxide, metal oxide or metal carbonate and the non-metal ion from the acid. 02/11/2020
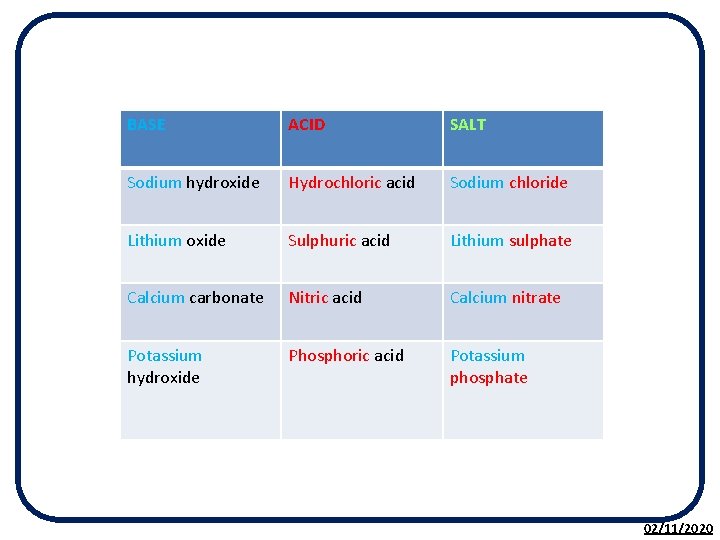
BASE ACID SALT Sodium hydroxide Hydrochloric acid Sodium chloride Lithium oxide Sulphuric acid Lithium sulphate Calcium carbonate Nitric acid Calcium nitrate Potassium hydroxide Phosphoric acid Potassium phosphate 02/11/2020

Experiment: Neutralisation • Metal Oxides • We have already seen that some metal oxides can dissolve in water to form alkalis. So can a metal oxide directly neutralise an acid? • ACTIVITY 5. 11 Neutralisation With a Metal Oxide • 1. Pour approximately 40 cm 3 of 1 moll-1 sulphuric acid into a beaker. • 2. Test its p. H using a strip of p. H paper. • 3. Add a spatula of either copper oxide or calcium oxide powder and stir until dissolved. You may need to gently warm the acid with a gentle blue flame to achieve this. • 4. Test its p. H using a strip of p. H paper, note the colour. • 5. Repeat until the p. H paper shows that the acid has been neutralised and there is an excess of unreacted metal oxide. • 6. Carefully filter of the excess powder and pour the filtrate into an evaporating basin. • 7. Gently evaporate off 2/3 of the water and leave for crystals of the salt to form overnight. 02/11/2020

Can you complete the word equation for this reaction? Copper Hydroxide + Nitric Acid→ Copper Nitrate + Water 02/11/2020

RULE: Lear n! Acid + Metal oxide → Salt + Water REMEMBER: this is a neutralisation reaction!! 02/11/2020

LESSON 8 Neutralisation reactions Metal Carbonates 02/11/2020

02/11/2020 We are learning to: • Conduct a neutralisation reaction using a metal carbonate I can: • Describe what the products are when an acid neutralises a metal carbonate

Experiment: method 02/11/2020

Notes: • Metal Carbonates • Copper(II) Carbonate (Cu. CO 3) powder can neutralise an acid such as Sulphuric acid (H 2 SO 4). 02/11/2020

Experiment: Metal Hydroxide • Metal hydroxide – alkalis • ACTIVITY 5. 8 Neutralisation With an Alkali • 1. Using a measuring cylinder, measure and pour 20 cm 3 of 0. 1 moll 1 hydrochloric acid into a beaker. • 2. Add a few drops of Universal indicator. • 3. Using a different measuring cylinder, measure and pour 20 cm 3 of 0. 1 moll-1 sodium hydroxide solution into the same beaker. • 4. Depending on the colour add drop wise, using a plastic pipette, just enough acid or alkali to get the solution neutral, count the number of drops. • 5. Repeat the experiment but do not add the indicator, remember to add the same number of drops of the acid or alkali. • 6. You should now have a neutral solution. • 7. Pour this into an evaporating basin and gently evaporate off the water. • 8. You should be left with a white solid, called a salt. 02/11/2020

Notes: • ACID + METAL OXIDE→ SALT + WATER • ACID + METAL CARBONATE→ SALT + WATER + CARBON DIOXIDE • ACID + ALKALI(BASE)→ SALT + WATER 02/11/2020

DISCUSS After discussion with your teacher and others, design an experiment that would prove that your chemical equation for the neutralisation of sulphuric acid with copper carbonate is correct. 02/11/2020

Notes: • In the space below, write a set of instructions to follow. 02/11/2020

Hint!!! How can we collect the gas? How do we test the gas? 02/11/2020

• RULE Lear n! • Acids can be neutralised by Metal Hydroxides, Metal Oxides or Metal Carbonates. A salt and water is always produced but with a metal carbonate, carbon dioxide gas is also formed. REMEMBER: this is a neutralisation reaction!! 02/11/2020

LESSON 9 TITRATIONS 02/11/2020

Lesson Starter: . What is a Titration? A Chemical technique where a solution of known concentration is used to determine the concentration of an unknown solution 02/11/2020

So what? • Real life examples • • • MEDICINES – PHARMACY FOOD VALUES and NUTRITION SCIENCE WATER TESTING SPORT SCIENCE 02/11/2020

Drug testing in sport 02/11/2020

Notes: • This all comes under the title of Chemical Analysis which is an important technique which you will come across as you progress on to National 5 Chemistry 02/11/2020

Experiment: Titration with an alkali KC 5. 10 • Your teacher will show you what a burette and pipette are and how to use them properly. • 1. Using a pipette measure 10 cm 3 of 0. 1 moll-1 hydrochloric acid into a conical flask. • 2. Add a few drops of screened phenolphthalein this will change colour when the acid is neutralised. • 3. Using a burette containing 0. 1 moll-1 sodium hydroxide solution add 1 cm 3 at a time, swirling the flask until the colour changes from colourless to pink. • 4. Note the volume of alkali added. • 5. Repeat the experiment but add the last few cm 3 one drop at a time so that the addition of one drop changes the indicator colour to green. Note the volume added again. • 6. Repeat until you get two volumes within 0. 2 cm 3 of each other, this is called being concordant. 02/11/2020
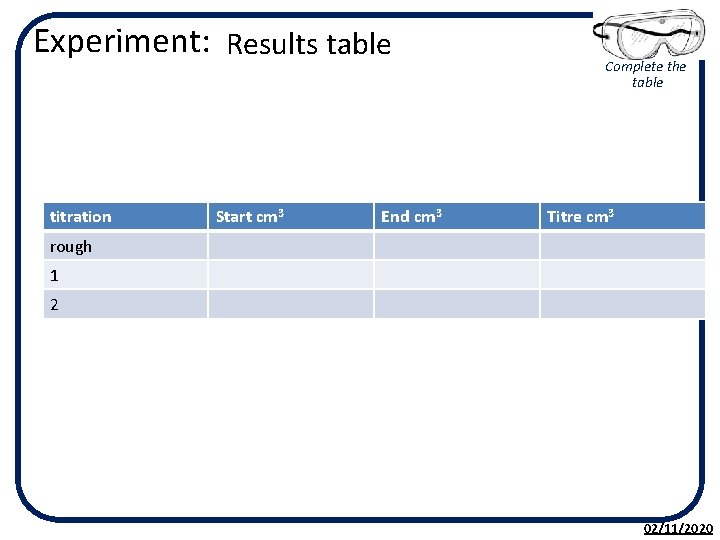
Experiment: Results table titration Start cm 3 End cm 3 Complete the table Titre cm 3 rough 1 2 02/11/2020

Notes: • Titration Calculations • Titrations can be used to find the unknown concentration of an acid. We have seen with our ionic equations for neutralisation reactions that its only the hydrogen and hydroxide ions that actually react. We also know that one H+ reacts with one OH– ion. 02/11/2020

Notes: • So at the exact neutralisation point the number of moles of H+ ions neutralised will be equal to the number of moles of OH- ions added. • n H+ ions = n OH- ions where n = number of moles • We know for solutions n= cv • So PVC (acid) = PVC (alkali) where P = number of H+ or OH- ions in formula • V = volume of acid or alkali • C = concentration of acid or alkali 02/11/2020

Notes: • For example: • What is the concentration of acid when 25 cm 3 of Sulphuric acid (H 2 SO 4) is neutralised by 25 cm 3 of 1. 0 moll-1 Sodium Hydroxide (Na. OH) solution? • PVC (acid) = PVC (alkali) • 2 x 25 x. C = 1 x 25 x 1 • 2 x 25 • C = 0. 5 moll-1 02/11/2020

Glossary l acid – A chemical that has a p. H less than 7. l alkali – A chemical that has a p. H greater than 7 and dissolves in water. l indicator – A special chemical that changes into different colours in acid and alkali solutions. l neutral – A chemical with a p. H of 7 that is not an acid or an alkali. l neutralization – The chemical reaction between an acid an alkali. l p. H scale – The range of values that shows how strong or weak an acid or alkali is. l universal indicator – The indicator that shows the p. H value of a chemical. 02/11/2020

Anagrams 02/11/2020
Word search 02/11/2020

Multiple-choice quiz 02/11/2020

Starter 1) What mass of carbon dioxide will be produced on burning 10 g of propane? C 3 H 8 + 5 O 2 3 CO 2 + 4 H 2 O 2) What mass of hydrogen is required if 40 g of nitrogen is added to the reaction vessel? N 2 + 3 H 2 2 NH 3 02/11/2020

Starter 1. The grid below contains various statements which apply to solutions. (a)Which 3 boxes apply to an acid? (b)Which 2 boxes apply to pure water? (c) Which 3 boxes apply to an alkali? A It contains more H+ ions than B It has a p. H =7 pure water C It has a p. H less than 7 D It contains ions E It contains more OH- ions F It has a p. H greater than 7 than pure water 02/11/2020

Starter 1. For the following neutralisation reactions, Complete the word equation. Write a chemical equation. Then balance the chemical equation. Finally circle the salt in each word equation. A) Potassium hydroxide + hydrochloric acid B) Calcium hydroxide + hydrochloric acid C) Sodium hydroxide + nitric acid D) Potassium hydroxide + sulphuric acid E) Calcium hydroxide + sulphuric acid F) Ammonium hydroxide + hydrochloric acid 2. Using one of the examples above, explain what is meant by a “salt”. 02/11/2020

Starter For the following neutralisation reactions, Complete the word equation. Write a chemical equation. Then balance the chemical equation. Finally circle the salt in each word equation. A) Hydrochloric acid + calcium oxide B) Nitric acid + copper (ll) oxide C) Sulphuric acid + zinc (II) oxide Re - write one of the above equations, showing ions and cancel out the spectator ions. Re - write the equation showing the ions which react. 02/11/2020

Past Paper Question 2016 02/11/2020
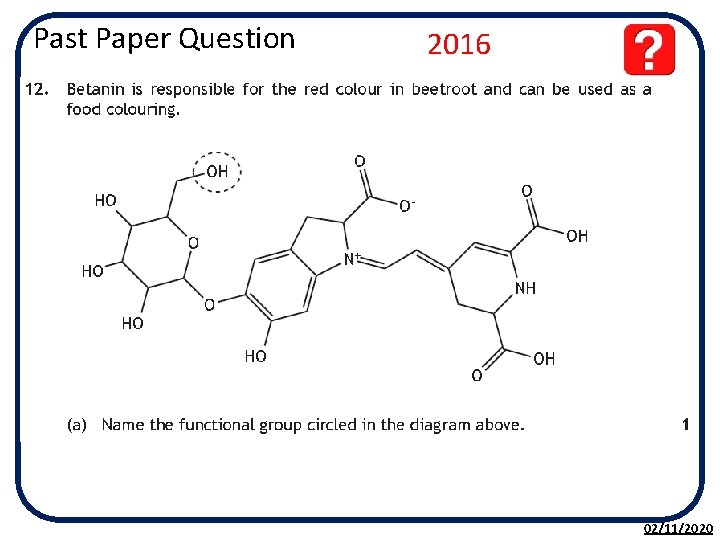
Past Paper Question 2016 02/11/2020
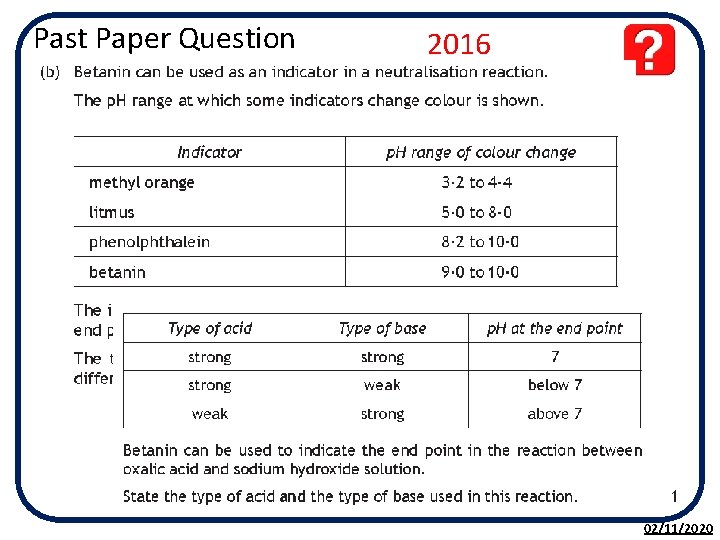
Past Paper Question 2016 02/11/2020
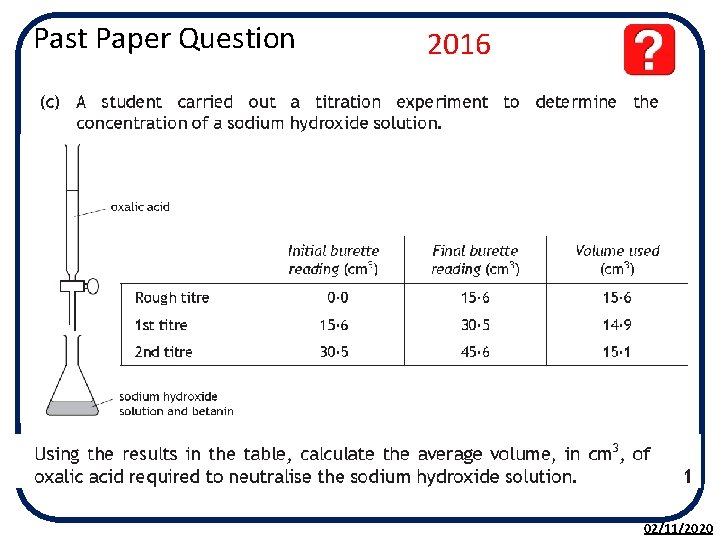
Past Paper Question 2016 02/11/2020

Past Paper Question 2016 02/11/2020
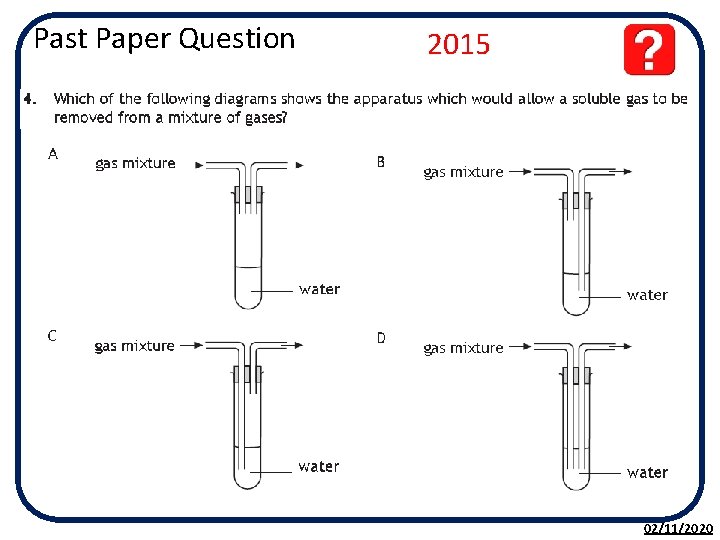
Past Paper Question 2015 02/11/2020

Past Paper Question 2015 02/11/2020

Past Paper Question 2015 02/11/2020

Past Paper Question 2015 02/11/2020
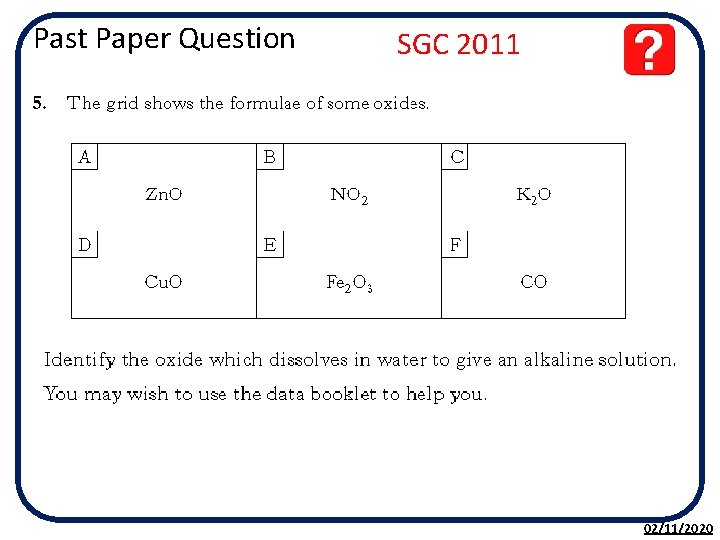
Past Paper Question SGC 2011 02/11/2020
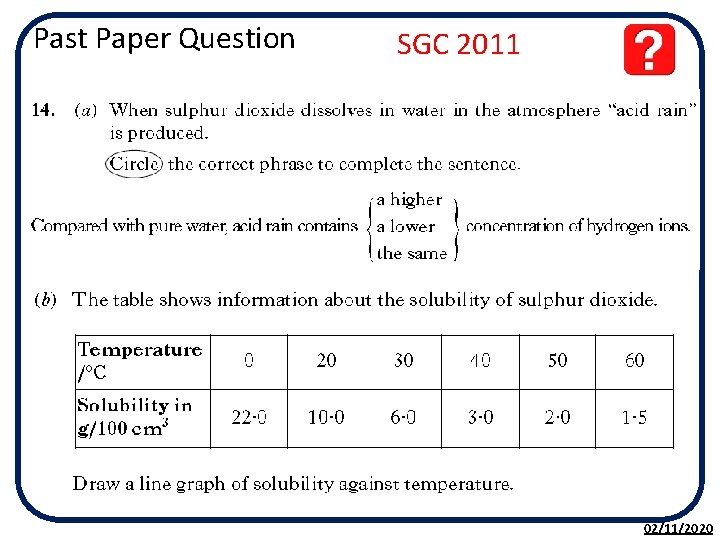
Past Paper Question SGC 2011 02/11/2020

Past Paper Question SGC 2011 02/11/2020

Past Paper Question SGC 2011 02/11/2020

Past Paper Question SGC 2011 02/11/2020

Past Paper Question SGC 2012 02/11/2020

Past Paper Question SGC 2012 02/11/2020
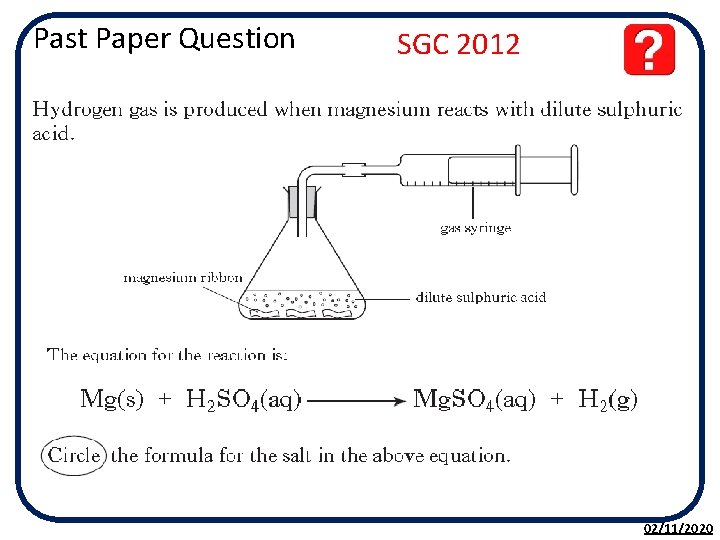
Past Paper Question SGC 2012 02/11/2020

Past Paper Question SGC 2012 02/11/2020

Past Paper Question SGC 2012 02/11/2020

Past Paper Question SGC 2012 02/11/2020

Past Paper Question SGC 2010 02/11/2020
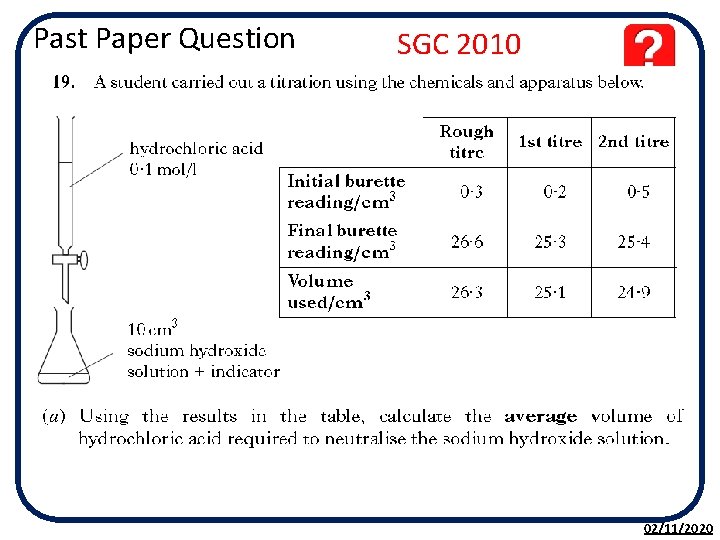
Past Paper Question SGC 2010 02/11/2020
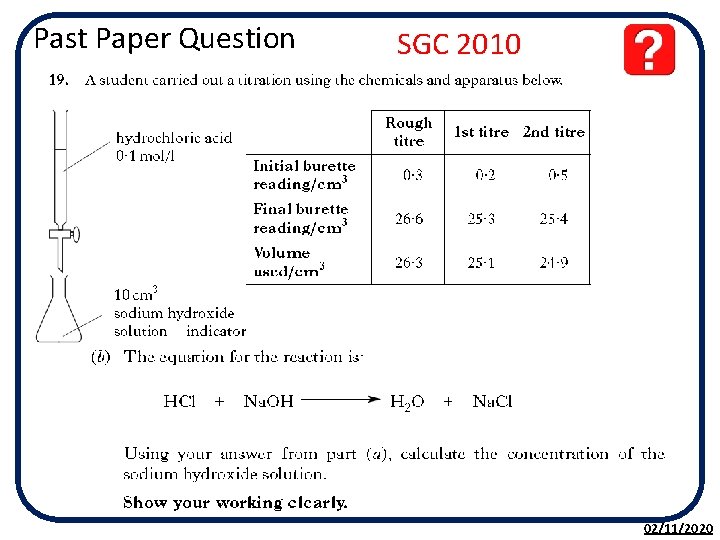
Past Paper Question SGC 2010 02/11/2020

Past Paper Question SGC 2012 02/11/2020
- Slides: 130