S 1316 Data Submission Roxanne Topacio CCRP SWOG

S 1316 Data Submission Roxanne Topacio, CCRP SWOG Data Operations Center Seattle, WA October 24, 2014

Overview • General form submission timelines • Special form submission timelines • Submitting data in Rave October 24, 2014 S 1316 Training 2

S 1316 General Form Submission: Where are these guidelines found? • S 1316 Protocol – Section 7. 5 Study Schedule for All Patients – Section 9. 0 Study Calendar – Section 14. 4 Data Submission Overview and Time points • Handouts for S 1316 Study Forms and Procedures packet – S 1316 Study Flow (handout pgs. 2 -4) – S 1316 Forms & Documents Overview (handout pgs. 23 -25) October 24, 2014 S 1316 Training 3

S 1316 General Form Submission: Patient Contact Schedule • Registration is Day 0 Use to plan for weekly follow-up through Week 53 • Weeks 1 – 13 Site calls patient for weekly assessments • Weeks 17 – 53 Site calls patient every 4 weeks for assessments October 24, 2014 S 1316 Training 4

S 1316 General Form Submission: standard forms • Baseline data – Submit within 7 days of registration • Weekly follow-up data (Weeks 1 -13) – Submit within 7 days of assessment ** If the patient is HOSPITALIZED, then also collect • Hospitalization data (Weeks 1 -13) – Submit within 14 days after discharge October 24, 2014 S 1316 Training 5

S 1316 General Form Submission: more standard forms • Hospitalization Days Record – Submit ONE time only – Submit within 14 days after Week 13 • Follow-up data every 4 weeks (Weeks 17 -53) – Submit within 14 days of assessment October 24, 2014 S 1316 Training 6

Two ‘special’ forms with non-standard timing • S 1316 Dietary Recall Contact Form – Email to the Arizona Diet, Behavior and Quality of Life Assessment Lab within 24 hours of registration • S 1316 Off Protocol Notice – Submit within 3 days of removal from follow-up (Section 7. 6) October 24, 2014 S 1316 Training 7

Rave: How data is submitted • Nearly all study data submitted via Rave EDC • Exceptions – Registration data submitted via OPEN – S 1316 Dietary Recall Contact Form submitted to the Arizona Diet, Behavior and QOL Assessment Lab October 24, 2014 S 1316 Training 8

Rave: How to Access • Rave link in the SWOG CRA Workbench • Rave link in CTSU. org • https: //login. imedidata. com/selectlogin October 24, 2014 S 1316 Training 9

Rave: Things to know • Rave forms look different than the paper forms in the forms packet • Rave can show only relevant folders, forms, or parts of a form • Some Rave forms appear conditionally October 24, 2014 S 1316 Training 10
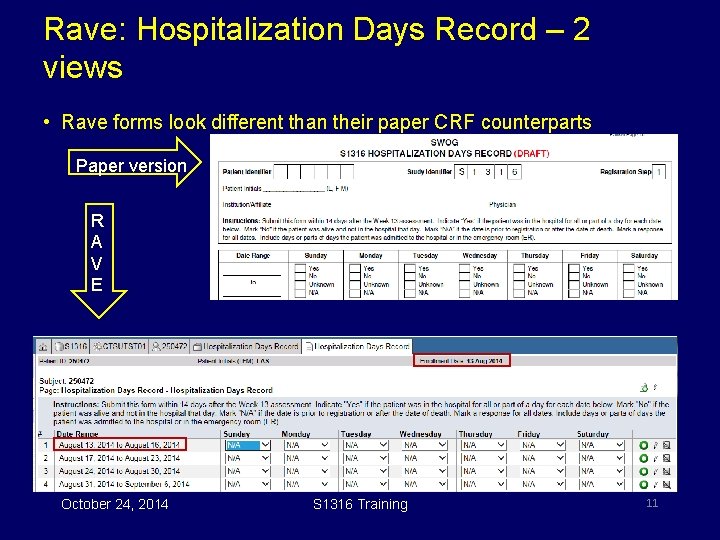
Rave: Hospitalization Days Record – 2 views • Rave forms look different than their paper CRF counterparts Paper version R A V E October 24, 2014 S 1316 Training 11
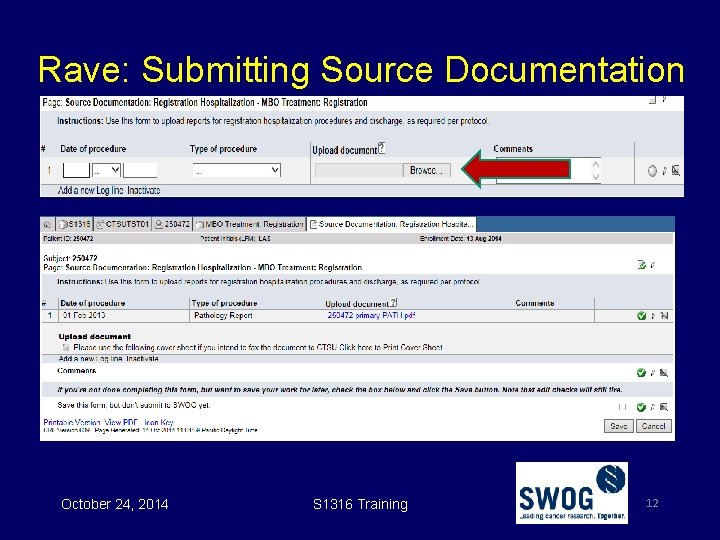
Rave: Submitting Source Documentation October 24, 2014 S 1316 Training 12

Summary • The protocol is your source for what to submit and when (Sections 7, 9 and 14) • Two forms require quick submission Dietary Recall Contact form - Arizona Lab within 24 hours of registration Off Protocol Notice - Submit online within 3 days (Section 7. 6) • Data submitted through Rave October 24, 2014 S 1316 Training 13

Rave: Help with Access? Rave Technical Support • CTSU Help Desk - 1 -888 -823 -5923 9: 30 am-8: 30 pm ET - ctsucontact@westat. com October 24, 2014 S 1316 Training 14

Questions on Data Submission Contact SWOG Data Operations Center • By email: cancercontrolquestion@crab. org • By phone: (206) 652 -2267 6: 30 AM – 4: 00 PM PT October 24, 2014 S 1316 Training 15
- Slides: 15