RWE is the New Black A Perspective from

RWE is the New Black: A Perspective from Industry Julia Brown, Vice President Government Affairs and Market Access CADTH Symposium April 2015 Gwendolyn Ryals, Look at Me Artwork from The Creative Center

What is Real World Evidence (RWE)? A working definition of RWE… Generated using data from usual care setting in a range of noninterventional (observational) studies, including: § Primary data collections § Analyses of secondary data RWE can be generated using so called “pragmatic” randomized trials that are conducted in more usual care settings that employ broad patient inclusion criteria and that investigate effectiveness (rather than efficacy) of different treatments. 1

Growing demand increasing data availability moving RWE close to tipping point Growing demand – Focus on value and transparency Increasing supply – Easier availability of data/analytics § Societal trends (e. g. , reform, aging population, emerging middle class) increasing utilization § More data from new data sources, e. g. , sensors, selfreported data § Markets becoming increasingly demanding as health care systems seek meaningful innovation, value and transparency § Better insights from structured and unstructured data § Substantial economic / pricing pressure; increased legal, regulatory, and business scrutiny § Governments (e. g. , US, UK, Denmark) playing an active role making health data liquid § Engaged patients – more involved in care, sharing and generating data RWE is growing in importance and relevance at accelerated pace 2
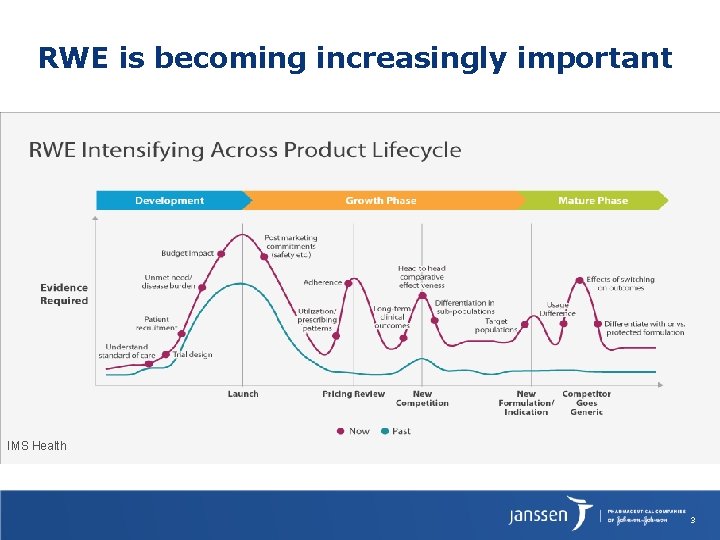
RWE is becoming increasingly important IMS Health 3

Stakeholders responding globally HTA bodies Patients ▪ ▪ ▪ Interest in real-world evidence mixed with questions on data privacy Starting to “create” real-world data via forums Broad variation of use, some systematic (e. g, UK, Italy) others more research focused (e. g. , US) Payors ▪ ▪ Selected payers use RWE formulary decisions (US) or cost/quality analyses (GER) Emerging outcomes based/risk-sharing contracts Regulators Providers/clinicians ▪ ▪ ▪ Mainly focused on pharmacovigilance Adaptive licensing concepts and corresponding use of RWE data Academia ▪ ▪ Broad use of RWE as source of insight to medical research Partnerships with other stakeholders ▪ Usage of internal RWE datasets for cost/quality monitoring and physician incentives Creating registries to generate RWE 4
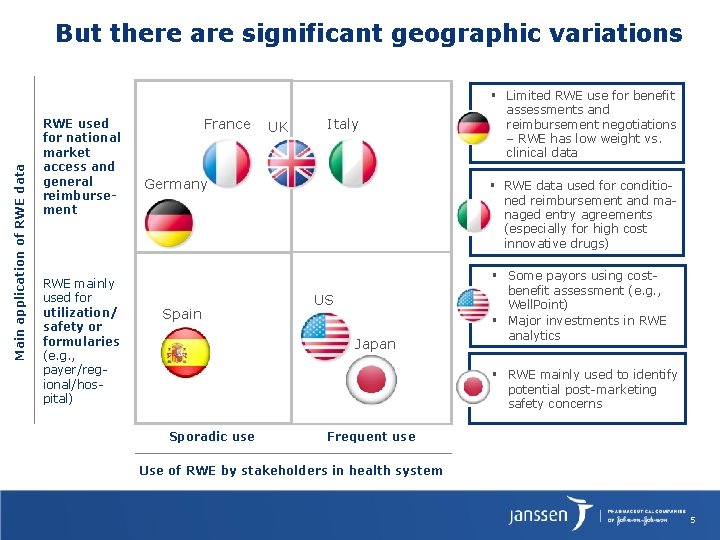
But there are significant geographic variations Main application of RWE data ▪ RWE used for national market access and general reimbursement RWE mainly used for utilization/ safety or formularies (e. g. , payer/regional/hospital) France UK Italy Germany § RWE data used for conditioned reimbursement and managed entry agreements (especially for high cost innovative drugs) ▪ Spain US ▪ Japan ▪ Sporadic use Limited RWE use for benefit assessments and reimbursement negotiations – RWE has low weight vs. clinical data Some payors using costbenefit assessment (e. g. , Well. Point) Major investments in RWE analytics RWE mainly used to identify potential post-marketing safety concerns Frequent use Use of RWE by stakeholders in health system 5
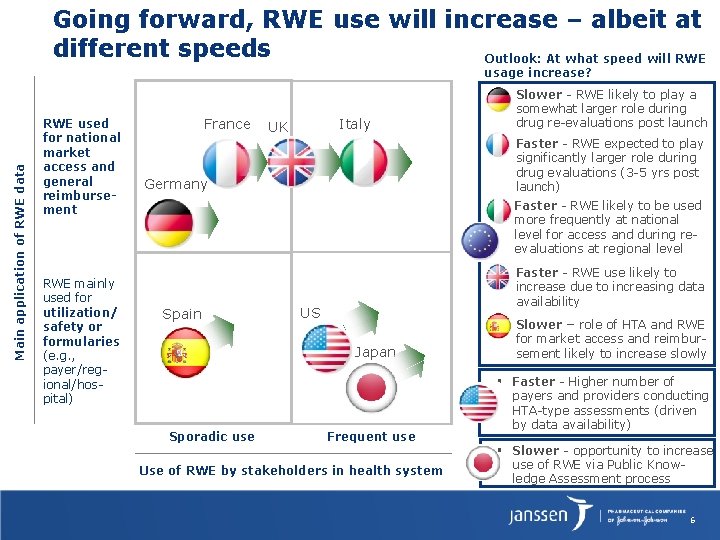
Going forward, RWE use will increase – albeit at different speeds Outlook: At what speed will RWE Main application of RWE data usage increase? RWE used for national market access and general reimbursement RWE mainly used for utilization/ safety or formularies (e. g. , payer/regional/hospital) France ▪ Slower - RWE likely to play a somewhat larger role during drug re-evaluations post launch ▪ Faster - RWE expected to play significantly larger role during drug evaluations (3 -5 yrs post launch) Italy UK Germany Faster - RWE likely to be used more frequently at national level for access and during reevaluations at regional level Spain US ▪ Faster - RWE use likely to increase due to increasing data availability ▪ Slower – role of HTA and RWE for market access and reimbursement likely to increase slowly ▪ Faster - Higher number of payers and providers conducting HTA-type assessments (driven by data availability) ▪ Slower - opportunity to increase use of RWE via Public Knowledge Assessment process Japan Sporadic use Frequent use Use of RWE by stakeholders in health system 6

What do we need to do in Canada to catch up? From… …To Mindset ▪ Ad hoc and reactive; postlaunch and product-centric ▪ Systematic and proactive; earlier and patient-centric Process ▪ Fragmented and serial lacking cohesive approach ▪ Iterative and strategic use of RCT and RWE Capabilities ▪ Low awareness, variable capabilities and no framework for incorporation into decision-making ▪ Broad awareness, collaboration and information sharing Partnerships ▪ One-off collaborations ▪ Coordinated and integrated partnerships Data/infrastructure ▪ Limited access and coordination ▪ Greater access, coordination Funding ▪ Limited funding ▪ Pre-planned and coordinated 7

In Summary § Engagement and collaboration from all stakeholders: – Patients, HTA bodies, payers, healthcare professionals, academia, industry… § Framework for RWE generation and incorporation into HTA reviews, public policy and formulary decision-making § Generation of data that balances commercial and public interests § Appropriate allocation of resources and willingness to risk-share § Prioritization of patient outcomes over short-term costcontainment 8
- Slides: 9