Rutgers GSBS PATH N 5209 The Business of

Rutgers - GSBS PATH N 5209 The Business of Science From Molecules to Medicines Fall 2016 Course Organizers: Nick Ponzio Steve Ritland Shanon Hunt Holly Hilton N 5209 1

Today’s Agenda • Welcome and Introduction (Nick) • A whirlwind tour of drug discovery and development (Shanon) • Business meets Science: The Drug Innovation Industry (Steve) • Course structure and deliverables (Shanon) Project team: Introduction to your target Course Website: http: //njms. rutgers. edu/gsbs/olc/fmtm/ N 5209 2

Introduction This course is about how new drugs get created This course will provide… • An introduction to the Big Picture of How Things Are Actually Done in Biotech/Pharma • Insight into the day-to-day activities of scientists / clinicians working in industry • Practical experience in how molecules move through the drug development cycle ( semester project) • Examples of areas where industry and academia collaborate to solve healthcare problems • Examples of career possibilities related to stages of the drug development cycle for graduates with advanced degrees in biomedical sciences Whether you pursue a career in industry or academics, it’s an N 5209 advantage to understand how the biotech/pharma industry works 3

Course Directors N 5209 4

Will work for Diet Coke! N 5209 5

Expectations and Evaluation Expectations: • Courtesy to our guest speakers and team members • Active participation in group work and Project Teams • Critical Thinking – applying concepts from lectures to the team project that you will develop Evaluation: • No Exams ! (are you kidding me? ) • Several oral team presentations • Project Portfolio • Class assignments and participation (by course directors) N 5209 • Peer feedback on team participation 6

If you think what you are doing in school is only to please your teacher or get a grade, you are missing a chance to not only learn, but also practice what you will be doing in the “real world” after you graduate. So: Be on time Be prepared Speak up and contribute without fear Collaborate on your projects Make professional presentations N 5209 7

M 2 M Biotechnology O I B M 2 M Biotechnology specializes in the full drug discovery and development lifecycle through first US Registration. 2 N 5209 As M 2 MBio employees, you will work on one of 3 teams involved in developing new drugs 8

Field Trips! N 5209 9

Faculty and Student Introductions Nick 1. Your name 2. Your major (or your job) 3. Your prior experience (if any) with Pharma/Biotech 4. Your favorite way to celebrate after your (successful) project presentations! N 5209 10

Course Structure and Organization Shanon N 5209 11

Course Overview When and where you need to be n Session Dates – Wednesday evenings – Sept 7 - Dec 21 (see syllabus) n Session Times – 6: 00 PM - 9: 00 PM (with one break, if you’re really, really lucky) n Classroom Location – GSBS Newark Campus – Medical Sciences Building (MSB) in the Department of Pathology – Room C-555 (main lecture); breakout rooms for team activities n Critical Due Dates – – N 5209 Date of your assigned Debate Session MC Presentation #1: October 19 MC Presentation #2: November 16 Final Project Portfolios: December 21 th (last day of class) 12

Course Overview How you’ll spend your class time Lecture (first ~1 hour of each class) n Each session will begin with a high level Introduction of the topic and its context for your semester project Activities Section (~2 hours for each class) n Activities will apply concepts from that day’s lecture n The activities conducted in class will be part of your Project Portfolio, due at the end of the course. N 5209 6 pm ~ 7: 00 15 mins 7: 15 ~ 9 pm Lecture Break Activities 13

Game Rules Lectures Our guest speakers are industry professionals who volunteer their time to teach this class. 1. Please come to class on time. 2. Please do not do emails, texts, Amazon shopping, etc. during lectures. Computers or notepads are fine for taking notes, and they will be very useful during project time (the second half of class), so please bring them. 3. Please feel free to ask questions/interact with lecturers. This is a good opportunity for you to get to know experts in their fields who do drug development every day. N 5209 14
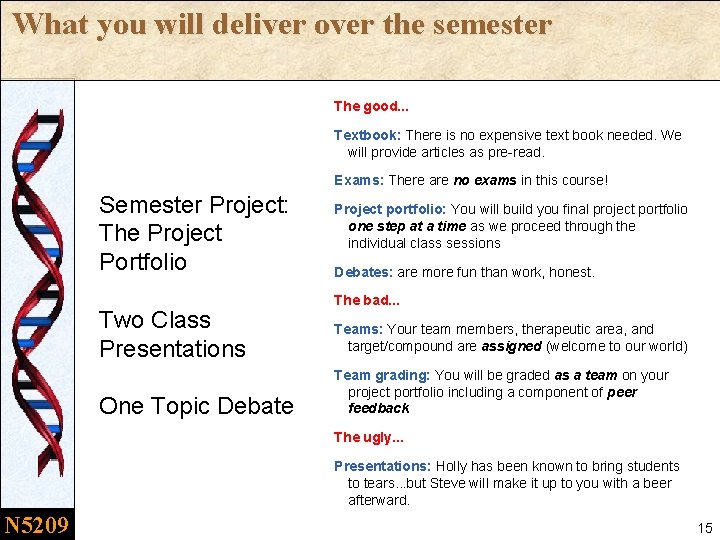
What you will deliver over the semester The good. . . Textbook: There is no expensive text book needed. We will provide articles as pre-read. Exams: There are no exams in this course! Semester Project: The Project Portfolio Two Class Presentations One Topic Debate Project portfolio: You will build you final project portfolio one step at a time as we proceed through the individual class sessions Debates: are more fun than work, honest. The bad. . . Teams: Your team members, therapeutic area, and target/compound are assigned (welcome to our world) Team grading: You will be graded as a team on your project portfolio including a component of peer feedback The ugly. . . Presentations: Holly has been known to bring students to tears. . . but Steve will make it up to you with a beer afterward. N 5209 15

Semester Project Course Overview Project Portfolios n n The Project Portfolio (PP) is a collection of deliverables that you will prepare over the course of the semester Each group will be required to submit a project portfolio at the end of the semester PPs will count for 30% of your semester grade Due Date: December 21 Project Portfolios include the following: 1. 2. 3. 4. 5. 6. 7. 8. 9. N 5209 Executive Summary Overview of Disease Pathogenesis, Epidemiology, and Current Treatment Target Rationale Investigator’s Brochure (Non-clinical Section) Phase 1 Protocol Synopsis Summary Investigator’s Brochure (Clinical Section) Target Product Profile (Start of Phase 3) Gantt Chart Lifecycle Strategic Plan 16

Group Presentations Course Overview Management Committee Requests for Decision n N 5209 Each group will develop and deliver two Management Committee Presentations (20 min. each) This is a “decision point” presentation, with a request to management Teams must select 2 -3 presenters for each of the presentations (all team members must present at least once) n Week 7: MC Presentation #1 – Entry into Development Portfolio – Therapeutic Area – Target, Mo. A, Scientific Rationale – Pharmacology – 20% of your final grade n Week 11: MC Presentation #2 – Proof of Concept, Entry into Phase 3 – Opportunity assessment, market potential – CMC strategy to FMI – Clinical Proof of Concept (Ph 1 & 2 data, efficacy & safety) – 20% of your final grade 17

Course Overview Class Debates; Assignments and Participation Debates: • 4 – 6 people will be assigned to each debate, assignments to follow • Students will need to prepare for their role ahead of class • More details on the debates will be provided as we get closer • Debates will count for 10% of your final grade Week 8 (Oct 26) Ethics of Ph 1 studies: How much risk? Week 12 (Nov 30) Expedited Approvals: Success or Failure? Week 14 (Dec 14) Drug Pricing and Access: What is fair? Class Assignments and Participation: • N 5209 These count for 10% of your final grade 18

Peer Evaluation - Teams Course Overview Feedback from your teammates will determine 10% of your final grade N 5209 19

Session Evaluation Course Overview Your feedback helps lecturers and course design N 5209 20
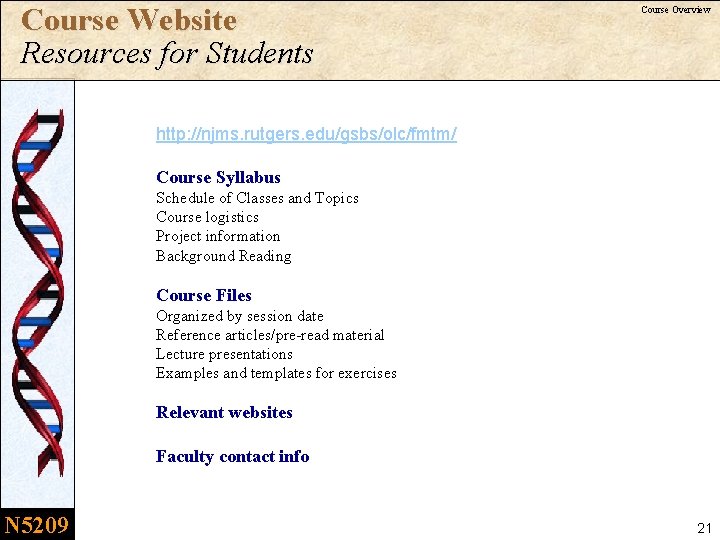
Course Website Resources for Students Course Overview http: //njms. rutgers. edu/gsbs/olc/fmtm/ Course Syllabus Schedule of Classes and Topics Course logistics Project information Background Reading Course Files Organized by session date Reference articles/pre-read material Lecture presentations Examples and templates for exercises Relevant websites Faculty contact info N 5209 21
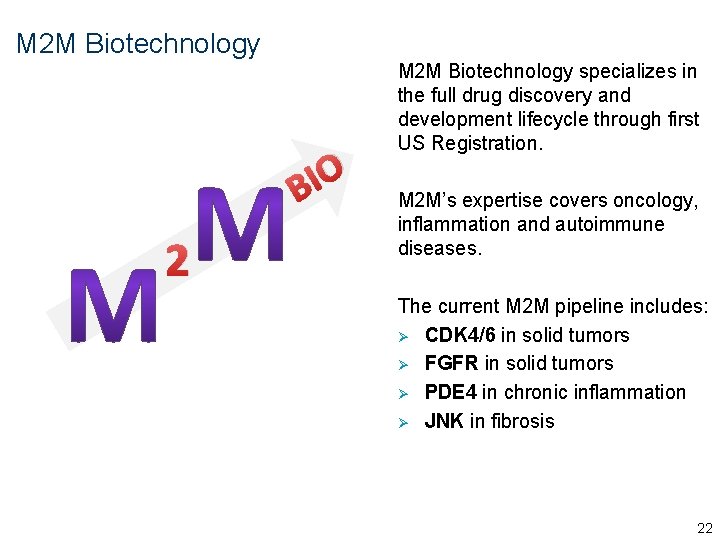
M 2 M Biotechnology O I B 2 M 2 M Biotechnology specializes in the full drug discovery and development lifecycle through first US Registration. M 2 M’s expertise covers oncology, inflammation and autoimmune diseases. The current M 2 M pipeline includes: Ø CDK 4/6 in solid tumors Ø FGFR in solid tumors Ø PDE 4 in chronic inflammation Ø JNK in fibrosis 22

Project Team and Debate Assignments N 5209 23

Assignment for Tonight Team Introductions n Collect your group into a breakout room n Introduce yourselves and your background Research and discuss your target for the semester project n Start with the articles we provided n Understand the target, the pathway, and potential therapeutic applications n What other companies are developing agents with this target, and what diseases are they in? N 5209 24

Q&A N 5209 25

A Whirlwind Overview of DD (Shanon) N 5209 26
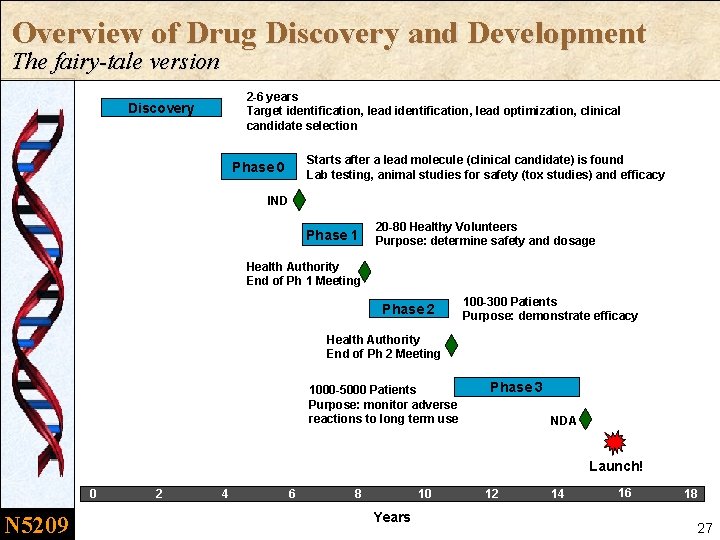
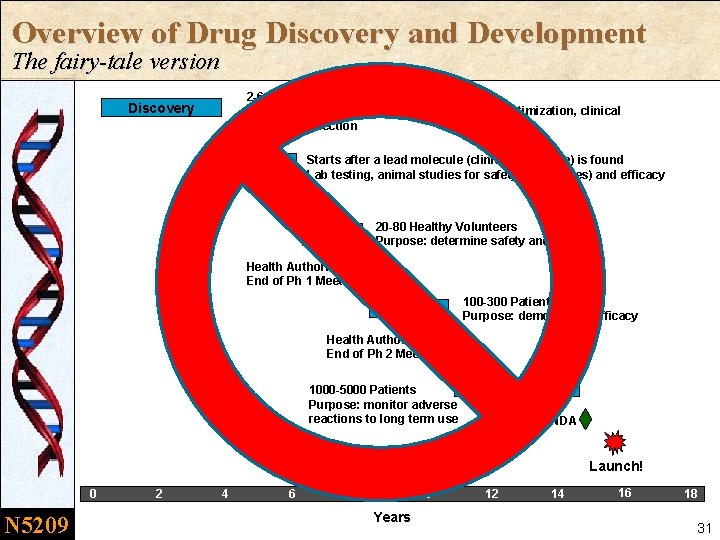
Overview of Drug Discovery and Development The fairy-tale version 2 -6 years Target identification, lead optimization, clinical candidate selection Discovery Starts after a lead molecule (clinical candidate) is found Lab testing, animal studies for safety (tox studies) and efficacy Phase 0 IND Phase 1 20 -80 Healthy Volunteers Purpose: determine safety and dosage Health Authority End of Ph 1 Meeting Phase 2 100 -300 Patients Purpose: demonstrate efficacy Health Authority End of Ph 2 Meeting 1000 -5000 Patients Purpose: monitor adverse reactions to long term use Phase 3 NDA Launch! 0 N 5209 2 4 6 8 10 Years 12 14 16 18 27

Q: Is it really this simple? A: No way. n It is not a stepwise linear process. – The compound could be sent backward to further explore a safety issue or even skip phases – Some phases might be duplicated (e. g, combinations) n There is considerable overlap of activities between Phases. – Toxicology animal studies continue to the NDA – Phase 1 or Phase 2 clinical trials can be running concurrently with Phase 3 trials – Phase 1/2 studies. . . Phase 2/3 studies. . n There are other aspects of drug development beyond the Clinical Plan – Health Authorities beyond the FDA, like MHRA (UK), Health Canada, Ministry of Health (China), PMDA (Japan) – Parallel development processes: drug manufacturing (CMC), companion diagnostics – New competitor activities or changes in market demand could change the development strategy (Business teams) N 5209 28

LEE 011 Clinical Development Overview 2013 2015 2014 2016 LEE 011 X 2101 FIH single agent FPFV May LEE 011 X 2102 Ph 1 s. a. FPFV June Pediatric MRT/NB LEE 011 X 1101 Japan s. a. solid tumors LEE 011 s/a in teratoma FPFV June PHASE 1/2 MEK 162 X 2114 Ph 1 b/2 combo MELANOMA FPFV STUDIES June LEE 011 X 2105 Ph 1 b/2 combo FPFV Sept PHASE 1/2 BC STUDIES LEE + MEK 162 N-RAS melanoma LEE + LGX 818 BRAF melanoma LGX 818 X 2102 Phase 2 rational combo study BRAF melanoma MEK 162 X 2110 Phase 2 triple combo LGX+MEK+LEE BRAF melanoma LEE 011 X 2106 Ph 1 b/2 combo LEE + RAD 011 and exemestane in BC LEE 011 X 2107 Ph 1 b/2 combo LEE + Let ± BYL in ER+ BC LEE 011 X 2108 Ph 1 b/2 combo LEE + Fulv + BYL/BKM in ER+ BC MONALEESA-1 Ph 2 pre-surgical PD dose opt MONALEESA-3 MONALEESA-5 PHASE 3 STUDIES MONALEESA-2 Ph 2 Neoadjuvant ER+ HER 2 - Premenopausal Women Phase 1/2, HER 2+ m. BC, Trastuzumab-Resistant Patients Phase 3: ER+ HER 2 - untreated m. BC MONALEESA-4 Ph 3: ER+ HER 2 - m. BC after NSAI MONALEESA-6 NSCLC 29 N 5209 Phase 3 HR+ HER 2+ Untreated m. BC LEE 011 + LDK 378 in pts with NSCLC | LEE 011 | LEE F 2 F 2013 | S Hunt | LEE at 40 K | Confidential
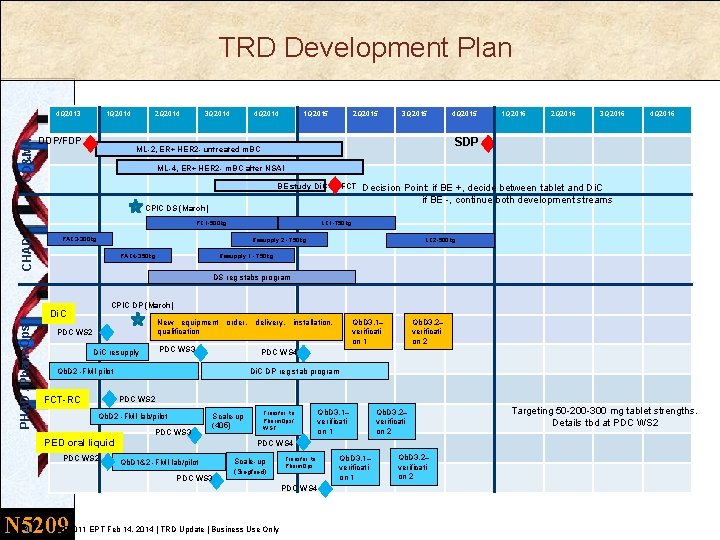
TRD Development Plan CD&MA 4 Q 2013 1 Q 2014 DDP/FDP 2 Q 2014 3 Q 2014 4 Q 2014 1 Q 2015 2 Q 2015 4 Q 2015 1 Q 2016 2 Q 2016 3 Q 2016 4 Q 2016 SDP ML-2, ER+ HER 2 - untreated m. BC ML-4, ER+ HER 2 - m. BC after NSAI BE study Di. C vs FCT CPIC DS (March) PC 1 -500 kg CHAD 3 Q 2015 Decision Point: if BE +, decide between tablet and Di. C if BE -, continue both development streams LC 1 -750 kg PAC 3 -300 kg Resupply 2 - 750 kg PAC 4 -350 kg LC 2 -500 kg Resupply 1 - 750 kg DS reg stabs program CPIC DP (March) PHAD / Pharm. Ops Di. C New equipment order, delivery, installation, qualification PDC WS 2 PDC WS 3 Di. C resupply Di. C DP reg stab program PDC WS 2 Qb. D 2 -FMI lab/pilot PDC WS 3 Scale-up (405) PED oral liquid PDC WS 2 Transfer to Pharm. Ops/ WST Qb. D 3. 1– verificati on 1 Qb. D 1&2 -FMI lab/pilot Scale-up (Siegfried) Transfer to Pharm. Ops PDC WS 4 30 Qb. D 3. 2– verificati on 2 PDC WS 4 PDC WS 3 N 5209 Qb. D 3. 2– verificati on 2 PDC WS 4 Qb. D 2 -FMI pilot FCT-RC Qb. D 3. 1– verificati on 1 | LEE 011 EPT Feb 14, 2014 | TRD Update | Business Use Only Qb. D 3. 1– verificati on 1 Qb. D 3. 2– verificati on 2 Targeting 50 -200 -300 mg tablet strengths. Details tbd at PDC WS 2
Overview of Drug Discovery and Development The fairy-tale version 2 -6 years Target identification, lead optimization, clinical candidate selection Discovery Starts after a lead molecule (clinical candidate) is found Lab testing, animal studies for safety (tox studies) and efficacy Phase 0 IND Phase 1 20 -80 Healthy Volunteers Purpose: determine safety and dosage Health Authority End of Ph 1 Meeting Phase 2 100 -300 Patients Purpose: demonstrate efficacy Health Authority End of Ph 2 Meeting 1000 -5000 Patients Purpose: monitor adverse reactions to long term use Phase 3 NDA Launch! 0 N 5209 2 4 6 8 10 Years 12 14 16 18 31
All said and done, Drug Development can rival the US Immigration Process N 5209 32
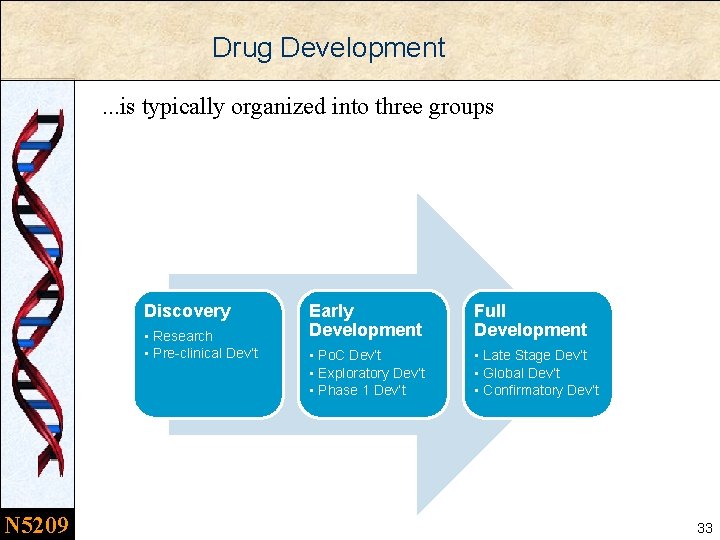
Drug Development. . . is typically organized into three groups Discovery • Research • Pre-clinical Dev’t N 5209 Early Development Full Development • Po. C Dev’t • Exploratory Dev’t • Phase 1 Dev’t • Late Stage Dev’t • Global Dev’t • Confirmatory Dev’t 33
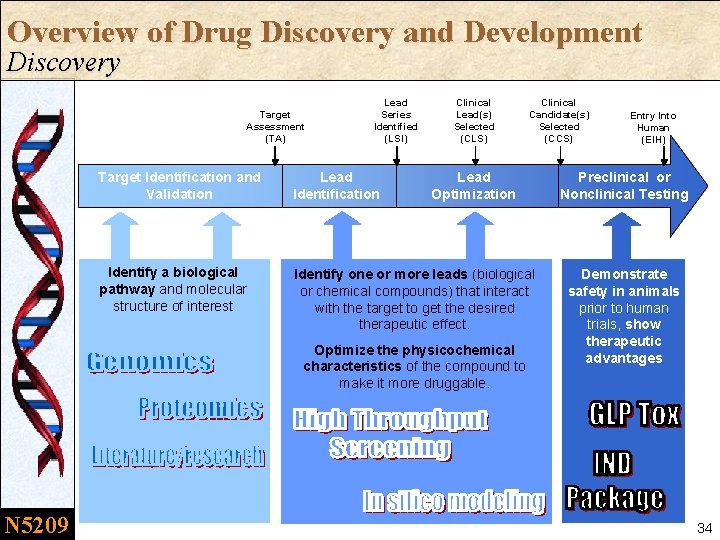
Overview of Drug Discovery and Development Discovery Target Assessment (TA) Lead Series Identified (LSI) Clinical Lead(s) Selected (CLS) Target Identification and Validation Lead Identification Lead Optimization Identify a biological pathway and molecular structure of interest Identify one or more leads (biological or chemical compounds) that interact with the target to get the desired therapeutic effect. Optimize the physicochemical characteristics of the compound to make it more druggable. N 5209 Clinical Candidate(s) Selected (CCS) Entry Into Human (EIH) Preclinical or Nonclinical Testing Demonstrate safety in animals prior to human trials, show therapeutic advantages 34
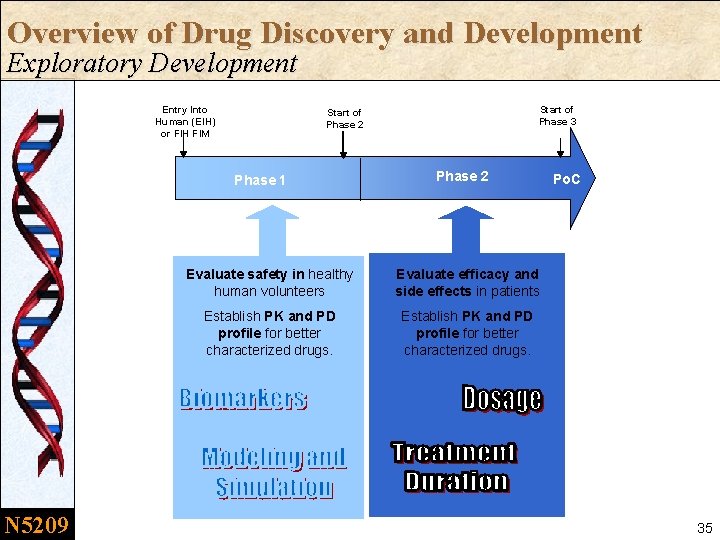
Overview of Drug Discovery and Development Exploratory Development Entry Into Human (EIH) or FIH FIM Phase 1 N 5209 Start of Phase 3 Start of Phase 2 Evaluate safety in healthy human volunteers Evaluate efficacy and side effects in patients Establish PK and PD profile for better characterized drugs. Po. C 35
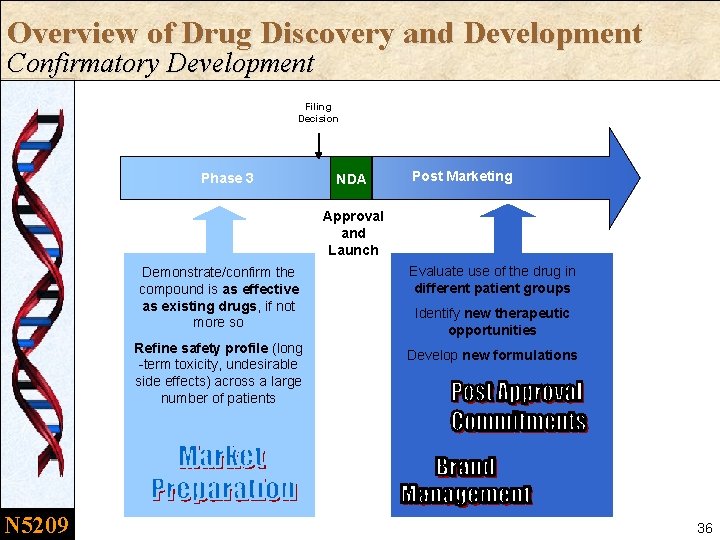
Overview of Drug Discovery and Development Confirmatory Development Filing Decision Phase 3 NDA Post Marketing Approval and Launch Demonstrate/confirm the compound is as effective as existing drugs, if not more so Refine safety profile (long -term toxicity, undesirable side effects) across a large number of patients N 5209 Evaluate use of the drug in different patient groups Identify new therapeutic opportunities Develop new formulations 36
- Slides: 36