Russian Academy Of Science B P Konstantinov Petersburg

Russian Academy Of Science B. P. Konstantinov Petersburg Nuclear Physics Institute Neutron Research Division Hydrogen Isotope Separation Laboratory Head of the laboratory: Ivan A. Alekseev HISL total staff: 17 employees 9/2/2021 The 2 nd International Workshop on the “MUON“ Project 1

Activity of the Laboratory n n n 9/2/2021 Full scale studies of various hydrogen isotope separation methods for the development of heavy and light water detritiation technology The scientific guidance and design supervision for the construction of the reactor PIK heavy water Detritiation and Deprotization Plant Computer simulation of the studying processes Provision and new procedures development of water and hydrogen gas isotope analysis Production of high quality heavy water and gaseous deuterium Pilot-scale production deuterium depleted water (up to 3 orders of magnitude below natural) The 2 nd International Workshop on the “MUON“ Project 2

Semi-industrial Facilities n Cryogenic distillation (CD) • The single column of 5. 5 m height with the interchangeable mass exchange sections of diameter up to 300 mm (the vacuum jacket of 1200 mm diameter) • Helium supply system (4 k. W at 20 K); Reflux flow of HD-D 2 -DT mixtures in the column - up to 10 k. Moles/h • Provided with the systems of deuterium production, gas compression, cleaning and storage n Water distillation (WD) • 4 distillation columns of 80 mm diameter and 10 m height with very high efficiency (HETP = 2. 5 cm) n Combine electrolysis and catalytic exchange (“EVIO”) • 2 exchange columns of 100 mm inner diameter and height of 6. 9 m filled with hydrophobic catalyst and packing • Electrolysis cells with capacity of 5 m 3 H 2 (D 2) per hour 9/2/2021 The 2 nd International Workshop on the “MUON“ Project 3

Separation Factor Is the Magnitude of an Elementary Separation Effect Table. The equilibrium separation factors hydrogen-water isotope exchange’s for tritium T and deuterium D at 60 С Deuterium concentration, at. % 0. 015 50 60 99. 9 T 4. 9 3. 1 2. 8 1. 5 D 3. 1 2. 8 2. 7 § Separation factor for isotopic exchange between water and hydrogen depends on what molecules are to be separated § Relatively high values of separation factor at ordinary conditions make the process of water-hydrogen isotope exchange attractive for the different tasks of hydrogen isotope separation. For comparison: separation factor for water distillation H-D=1. 026 at atmospheric pressure 9/2/2021 The 2 nd International Workshop on the “MUON“ Project 4

Demonstration Very High Detritiation Factor With “EVIO” Facility § Repeated contact over catalyst between water vapour and gaseous hydrogen and counter current between liquid water and vapour-gas mixture underlies the CECE process and leads to the effect of separation multiplying § “EVIO” facility is under operation now. Heavy water purification from tritium is successfully being carried out with the following characteristics: § The achieved separation degree (SD) is more than 2*103 § The top product output capacity is about 8 litres per day § Today’s results receiving on the column of height only 7 m do not have any similar ones in the world 9/2/2021 The 2 nd International Workshop on the “MUON“ Project 5

The Task of Pilot-scale Production of Deuterium Depleted Water at PNPI n n n Gaseous hydrogen containing deuterium in an amount about 0. 1 ppm is needed Natural water contains about 150 ppm of deuterium. Thus needed separation degree (SD) is 1. 5*103. The total required amount of the product does not exceed 10 litres. This is much less intensive task compared to the separation possibilities of our facilities: n n 9/2/2021 “EVIO” lets to process diluted heavy water waste contaminated by tritium and produce high quality heavy water. The achieved SD for D-H in a wide range of deuterium concentration is about 1*105 The achievable SD for the task of heavy water conditioning is about 5*103 in any of the existing WD column The 2 nd International Workshop on the “MUON“ Project 6

Possible Ways and Weak Points of the Task Solution n Any of the above mentioned methods and existing facilities can be used in principle for the production of super-light water (0. 1 ppm of D) But according to the required small scale production only two methods were dealt with: n A new laboratory scale installation based on Combine Electrolysis and Catalytic Exchange (CECE) process and n One of the existing Water Distillation column There are two weak points of this separation task solution. They mainly concern earlier used (for heavy water production) equipment: n The first is trouble with washing of dead spaces and n The second is lack of operative analysis that would allow the tracing of the washing process 9/2/2021 The 2 nd International Workshop on the “MUON“ Project 7
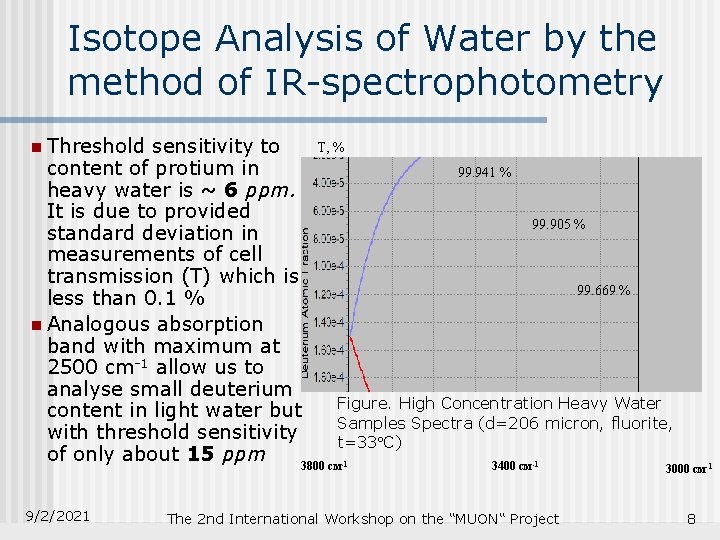
Isotope Analysis of Water by the method of IR-spectrophotometry n Threshold Т, % sensitivity to content of protium in 99. 941 % heavy water is ~ 6 ppm. It is due to provided 99. 905 % standard deviation in measurements of cell transmission (T) which is 99. 669 % less than 0. 1 % n Analogous absorption band with maximum at 2500 cm-1 allow us to analyse small deuterium Figure. High Concentration Heavy Water content in light water but Samples Spectra (d=206 micron, fluorite, with threshold sensitivity t=33 C) of only about 15 ppm 3800 см 3400 см -1 9/2/2021 -1 The 2 nd International Workshop on the “MUON“ Project 3000 см-1 8
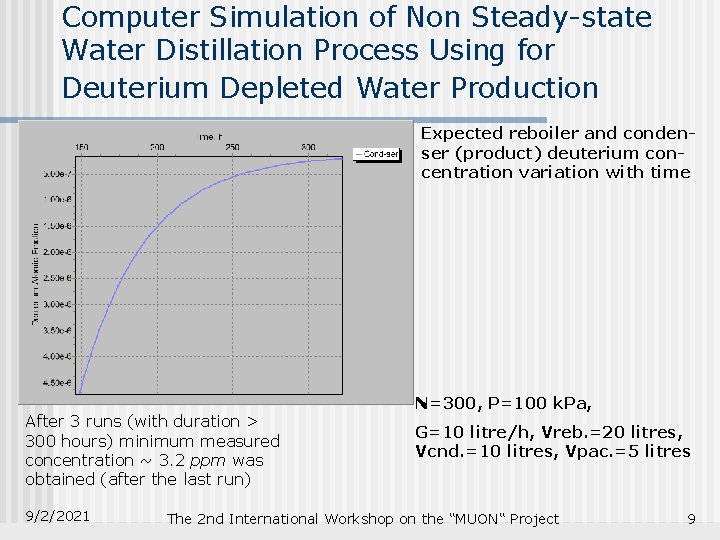
Computer Simulation of Non Steady-state Water Distillation Process Using for Deuterium Depleted Water Production Expected reboiler and condenser (product) deuterium concentration variation with time After 3 runs (with duration > 300 hours) minimum measured concentration ~ 3. 2 ppm was obtained (after the last run) 9/2/2021 N=300, P=100 k. Pa, G=10 litre/h, Vreb. =20 litres, Vcnd. =10 litres, Vpac. =5 litres The 2 nd International Workshop on the “MUON“ Project 9

9/2/2021 The 2 nd International Workshop on the “MUON“ Project 10

Laboratory Scale CECE Process for Deuterium Depleted Water Production or Conditioning n n n Burner Electrolysis cells capacity is Product as gas 0. 5 m 3 H 2 per hour Condenser or as water Catalytic Burner capacity is the Water dosing 3 same - 0. 5 m H 2 per hour pump 1 litre of hydrophobic Catalyst - 0. 8 % wt. Pt deposited on porous styrene-divinylbenzene copolymer Isotope exchange Thermostat 2. 5 litres of Packing - stainless apparatus steel spiral-prismatic with size 2. 2*0. 2 mm Including such an installation in the structure of Gas Electrolysis Circulation System would Waste Cells provide a guarantee of low water level deuterium admixture 9/2/2021 The 2 nd International Workshop on the “MUON“ Project 11
- Slides: 11