Rules for nomenclature in inorganic coordination chemistry Created


![[Co(NH 2 CH 2 NH 2)2 Cl 2]Cl (ethylenediamine, en) dichlorobis(ethylenediamine)Cobalt(III) chloride [Co(NH 3)4 [Co(NH 2 CH 2 NH 2)2 Cl 2]Cl (ethylenediamine, en) dichlorobis(ethylenediamine)Cobalt(III) chloride [Co(NH 3)4](https://slidetodoc.com/presentation_image_h2/4ca6c535b135cf7c29e4df2d41b0b00d/image-4.jpg)
![Na 2[Ti. F 6] Sodium hexafluorotitanate(IV) Na 2 Fe(CO)4 Collman’s reagent : p Sodium Na 2[Ti. F 6] Sodium hexafluorotitanate(IV) Na 2 Fe(CO)4 Collman’s reagent : p Sodium](https://slidetodoc.com/presentation_image_h2/4ca6c535b135cf7c29e4df2d41b0b00d/image-5.jpg)

![cis-[Co(NH 3)4 Cl 2]+ vs trans-[Co(NH 3)4 Cl 2]+ fac-[Co(NH 3)3(H 2 O)3]3+ vs cis-[Co(NH 3)4 Cl 2]+ vs trans-[Co(NH 3)4 Cl 2]+ fac-[Co(NH 3)3(H 2 O)3]3+ vs](https://slidetodoc.com/presentation_image_h2/4ca6c535b135cf7c29e4df2d41b0b00d/image-8.jpg)
- Slides: 9

Rules for nomenclature in inorganic coordination chemistry Created by Adam R. Johnson, Harvey Mudd College (adam_johnson@hmc. edu) and posted on VIPEr in July 2020. Copyright Adam R. Johnson, 2020. This work is licensed under the Creative Commons Attribution-Non. Commercial. Share. Alike License. To view a copy of this license visit http: //creativecommons. org/licenses/by-nc-sa/4. 0/

Nomenclature rules 1. Name cation first, then anion 2. In a formula a) Inner coordination sphere [in brackets] b) Write metal first than ligands 3. In a name a) Write ligands first, then metal b) Indicate # of ligands with prefix (below) c) Ligands in alphabetical order not counting prefix d) “complicated” ligands enclosed (in parantheses) e) Water is “aqua” and ammonia is “ammine” f) Anionic ligands have “-o” suffix (chloro) 4. Describe metal OS in 2 ways: M(III) or [MLn]n+ 5. If complex ion is negative, use “-ate” ending (ferrate) bis-, ditris-, tritetrakis-, tetrapentakis-, pentahexakis-, hexa- bis(ethylenediamine), diaqua tris(dimethylamido), trichloro tetracyano, tetrakis(triphenylphosphine)

Nomenclature rules Common ligands: NH 3: ammine Water: aquo Cl/Br: chloro/bromo En: ethylenediamine Ox: oxalate (-O 2 CCO 2 -) N 3 -: azido Py: pyridine CO: carbonyl CH 3 -: methyl Acetylacetonato - mu, bridging - eta, hapto, denticity - kappa, connectivity Lets try some practice problems
![CoNH 2 CH 2 NH 22 Cl 2Cl ethylenediamine en dichlorobisethylenediamineCobaltIII chloride CoNH 34 [Co(NH 2 CH 2 NH 2)2 Cl 2]Cl (ethylenediamine, en) dichlorobis(ethylenediamine)Cobalt(III) chloride [Co(NH 3)4](https://slidetodoc.com/presentation_image_h2/4ca6c535b135cf7c29e4df2d41b0b00d/image-4.jpg)
[Co(NH 2 CH 2 NH 2)2 Cl 2]Cl (ethylenediamine, en) dichlorobis(ethylenediamine)Cobalt(III) chloride [Co(NH 3)4 Cl 2]Cl tetraamminedichlorocobalt(III) chloride Postassium salt of [Pt(ox)2]2 - (oxalate) Potassium bisoxalatoplatinate(II)
![Na 2Ti F 6 Sodium hexafluorotitanateIV Na 2 FeCO4 Collmans reagent p Sodium Na 2[Ti. F 6] Sodium hexafluorotitanate(IV) Na 2 Fe(CO)4 Collman’s reagent : p Sodium](https://slidetodoc.com/presentation_image_h2/4ca6c535b135cf7c29e4df2d41b0b00d/image-5.jpg)
Na 2[Ti. F 6] Sodium hexafluorotitanate(IV) Na 2 Fe(CO)4 Collman’s reagent : p Sodium tetracarbonylferrate(-II) Mn(Br)(CO)2(PPh 3)3 Bromodicarbonyl tris(triphenylphosphine) manganese

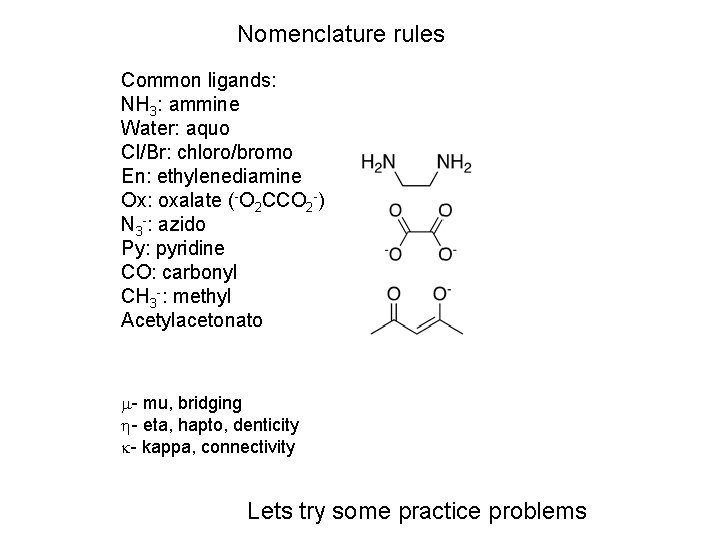
Greek prefixes: eta (h) and mu (m) Ti( 5 -C 5 H 5)2 Cl 2 Contrast ( 1 -C 5 H 5) (NH 3)5 Ru( 2 -pyrazine)Ru(NH 3)5]5+ (pz = ) Also briding halides, oxo, hydroxo

Isomers Stereoisomerism cis/trans fac/mer /
![cisCoNH 34 Cl 2 vs transCoNH 34 Cl 2 facCoNH 33H 2 O33 vs cis-[Co(NH 3)4 Cl 2]+ vs trans-[Co(NH 3)4 Cl 2]+ fac-[Co(NH 3)3(H 2 O)3]3+ vs](https://slidetodoc.com/presentation_image_h2/4ca6c535b135cf7c29e4df2d41b0b00d/image-8.jpg)
cis-[Co(NH 3)4 Cl 2]+ vs trans-[Co(NH 3)4 Cl 2]+ fac-[Co(NH 3)3(H 2 O)3]3+ vs mer-Co(NH 3)3(H 2 O)3]3+

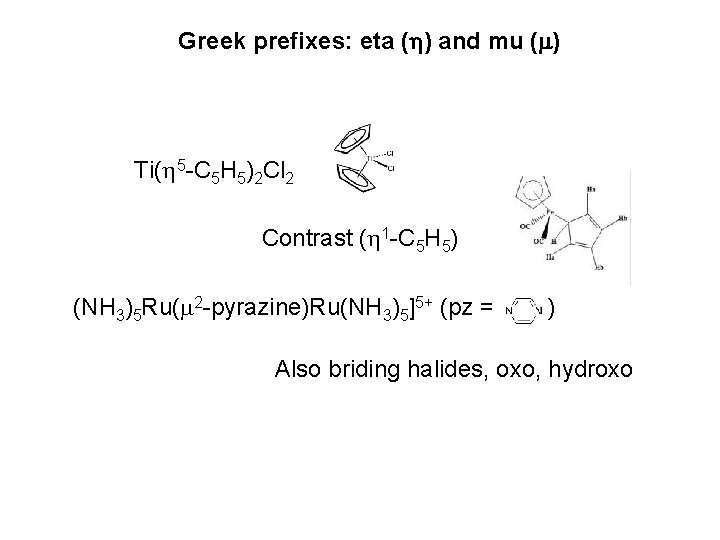
Cobalt(III) tris-ethylenediamine Right handed Left handed