RUBYI Study cohort 2 ombitasvirparitaprevirritonavir dasabuvir RBV for

RUBY-I Study, cohort 2: ombitasvir/paritaprevir/ritonavir + dasabuvir + RBV for HCV genotype 1 with renal impairment § Design ≥ 18 years Chronic HCV infection Genotype 1 Treatment-naïve or PEG-IFN + RBV-experienced HCV RNA > 1, 000 IU/ml Chronic kidney disease with e. GFR (MDRD) < 30 ml/min/1. 73 m 2 (dialysis permitted) Compensated cirrhosis allowed ** No HBV or HIV co-infection Open label Genotype 1 a N = 28 F 0 -F 3 Genotype 1 a N = 9 F 4 Genotype 1 b N = 11 F 0 -F 4 W 12 W 24 OBV/PTV/r DSV + RBV OBV/PTV/r + DSV ** Liver biopsy (Metavir ≤ F 3, Ishak score ≤ 4) or Fibroscan < 12. 5 k. Pa or Fibro. Test ≤ 0. 72 + APRI ≤ 2 § Treatment regimens – Co-formulated ombitasvir (OBV)/paritaprevir (PTV)/rironavir (r): 25/150/100 mg qd = 2 tablets – Dasabuvir (DSV): 250 mg bid ; RBV 200 mg qd (genotype 1 a) § Objective – SVR 12 (HCV RNA < 25 IU/ml) by intent-to-treat with 2 -sided 95% CI RUBY-I, cohort 2 Vierling JM. AASLD 2016, Abs. 886
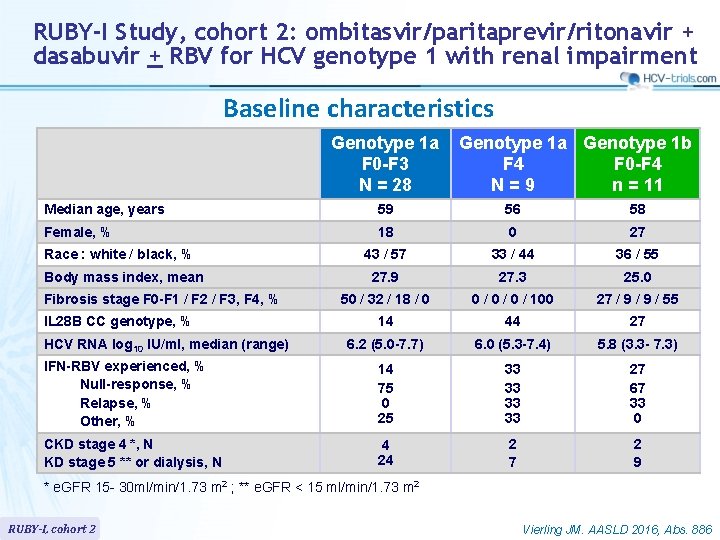
RUBY-I Study, cohort 2: ombitasvir/paritaprevir/ritonavir + dasabuvir + RBV for HCV genotype 1 with renal impairment Baseline characteristics Genotype 1 a F 0 -F 3 N = 28 Genotype 1 a Genotype 1 b F 4 F 0 -F 4 N=9 n = 11 Median age, years 59 56 58 Female, % 18 0 27 43 / 57 33 / 44 36 / 55 27. 9 27. 3 25. 0 50 / 32 / 18 / 0 0 / 0 / 100 27 / 9 / 55 14 44 27 6. 2 (5. 0 -7. 7) 6. 0 (5. 3 -7. 4) 5. 8 (3. 3 - 7. 3) IFN-RBV experienced, % Null-response, % Relapse, % Other, % 14 75 0 25 33 33 27 67 33 0 CKD stage 4 *, N KD stage 5 ** or dialysis, N 4 24 2 7 2 9 Race : white / black, % Body mass index, mean Fibrosis stage F 0 -F 1 / F 2 / F 3, F 4, % IL 28 B CC genotype, % HCV RNA log 10 IU/ml, median (range) * e. GFR 15 - 30 ml/min/1. 73 m 2 ; ** e. GFR < 15 ml/min/1. 73 m 2 RUBY-I, cohort 2 Vierling JM. AASLD 2016, Abs. 886
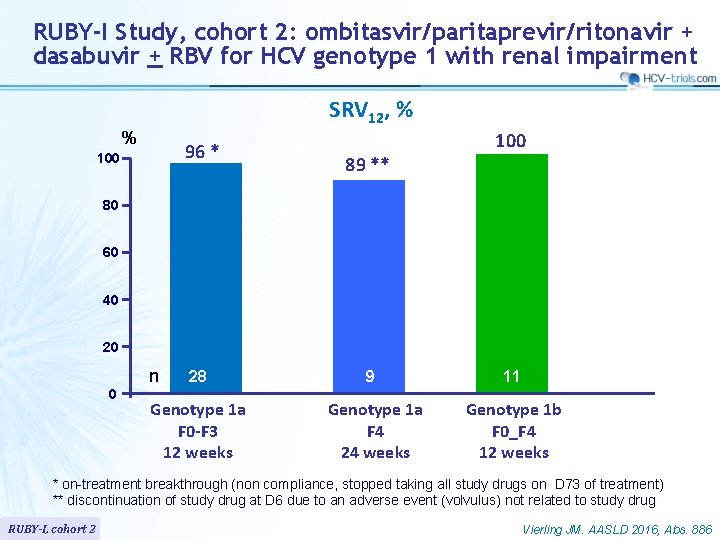
RUBY-I Study, cohort 2: ombitasvir/paritaprevir/ritonavir + dasabuvir + RBV for HCV genotype 1 with renal impairment SRV 12, % % 96 * 100 89 ** 100 80 60 40 20 n 0 28 9 11 Genotype 1 a F 0 -F 3 12 weeks Genotype 1 a F 4 24 weeks Genotype 1 b F 0_F 4 12 weeks * on-treatment breakthrough (non compliance, stopped taking all study drugs on D 73 of treatment) ** discontinuation of study drug at D 6 due to an adverse event (volvulus) not related to study drug RUBY-I, cohort 2 Vierling JM. AASLD 2016, Abs. 886
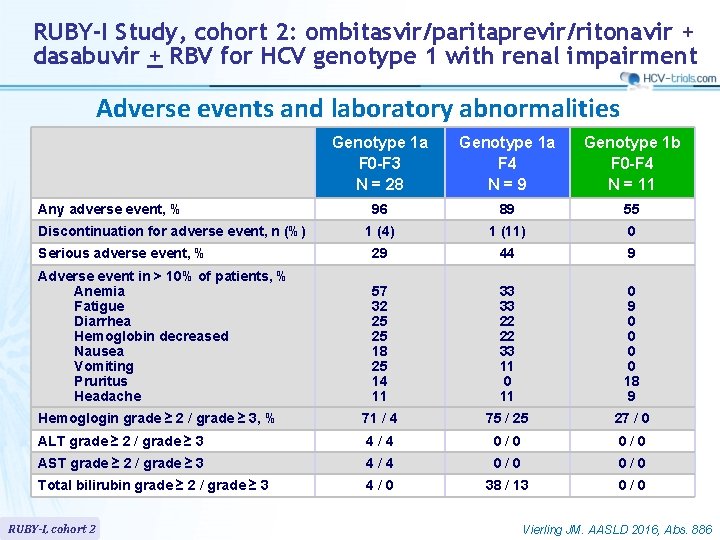
RUBY-I Study, cohort 2: ombitasvir/paritaprevir/ritonavir + dasabuvir + RBV for HCV genotype 1 with renal impairment Adverse events and laboratory abnormalities Genotype 1 a F 0 -F 3 N = 28 Genotype 1 a F 4 N=9 Genotype 1 b F 0 -F 4 N = 11 96 89 55 1 (4) 1 (11) 0 Serious adverse event, % 29 44 9 Adverse event in > 10% of patients, % Anemia Fatigue Diarrhea Hemoglobin decreased Nausea Vomiting Pruritus Headache 57 32 25 25 18 25 14 11 33 33 22 22 33 11 0 9 0 0 18 9 Hemoglogin grade ≥ 2 / grade ≥ 3, % 71 / 4 75 / 25 27 / 0 ALT grade ≥ 2 / grade ≥ 3 4/4 0/0 Any adverse event, % Discontinuation for adverse event, n (%) AST grade ≥ 2 / grade ≥ 3 4/4 0/0 * e. GFR 15 - 30 ml/min/1. 73 m 2 ; ** e. GFR < 15 ml/min/1. 73 m 2 or hemodialysis Total bilirubin grade ≥ 2 / grade ≥ 3 4/0 38 / 13 RUBY-I, cohort 2 0/0 Vierling JM. AASLD 2016, Abs. 886

RUBY-I Study, cohort 2: ombitasvir/paritaprevir/ritonavir + dasabuvir + RBV for HCV genotype 1 with renal impairment § Summary – OBV/PTV/r + DSV ± RBV resulted in an SVR 12 rate of 96% in patients with CKD stages 4 or 5 in cohort 2 of the RUBY-I study – The regimen was generally well tolerated for this group of patients with severe underlying comorbidities, with • 1 possibly DAA-related serious adverse event of diarrhea • and 1 discontinuation due to an adverse event unrelated to treatment – A large proportion of patients on RBV required RBV dose modification for anemia – Most adverse events were mild or moderate in severity – These results of efficacy and safety support the use of this regimen in patients with advanced renal disease, for whom treatment options are limited RUBY-I, cohort 2 Vierling JM. AASLD 2016, Abs. 886
- Slides: 5