RTP TV An 8 Part Live CME Webcast

RTP TV: An 8 -Part Live CME Webcast Series Part VI – Triple-Negative Breast Cancer: Current Clinical Management Tuesday, July 19, 2011 7: 30 PM - 8: 30 PM ET

Neil Love, MD Research To Practice Miami, Florida Harold J Burstein, MD, Ph. D Associate Professor of Medicine, Harvard Medical School Breast Oncology Center Dana-Farber Cancer Institute Boston, Massachusetts Charles E Geyer Jr, MD Director of Medical Affairs National Surgical Adjuvant Breast and Bowel Project Vice-Chair, Department of Human Oncology Allegheny General Hospital Pittsburgh, Pennsylvania

Disclosures for Moderator Neil Love, MD Dr Love is president and CEO of Research To Practice, which receives funds in the form of educational grants to develop CME activities from the following commercial interests: Allos Therapeutics, Amgen Inc, Astellas Pharma Global Development Inc, Astra. Zeneca Pharmaceuticals LP, Aureon Laboratories Inc, Bayer Health. Care Pharmaceuticals/Onyx Pharmaceuticals Inc, Biogen Idec, Boehringer Ingelheim Pharmaceuticals Inc, Bristol. Myers Squibb Company, Celgene Corporation, Cephalon Inc, Daiichi Sankyo Inc, Dendreon Corporation, Eisai Inc, EMD Serono Inc, Genentech Bio. Oncology, Genomic Health Inc, Im. Clone Systems, a wholly owned subsidiary of Eli Lilly and Company, Lilly USA LLC, Millennium: The Takeda Oncology Company, Mundipharma International Limited, Myriad Genetics Inc, Novartis Pharmaceuticals Corporation, OSI Oncology, Sanofi and Seattle Genetics.

Disclosures for Harold J Burstein, MD, Ph. D No financial interests or affiliations to disclose

Disclosures for Charles E Geyer Jr, MD No financial interests or affiliations to disclose

Agenda — Triple-Negative Breast Cancer: Current Clinical Management • Module 1: Metastatic TNBC – Case A: Dr Burstein – ASCO 2011 review – Case B: Dr Geyer • Rapid-Fire Questions 1 • Module 2: Management of Patients Who Are BRCA-Positive – Case C: Dr Burstein • Rapid-Fire Questions 2 • Module 3: Adjuvant/Neoadjuvant Therapy for TNBC – Case D: Dr Geyer – Case E: Submitted by Dr William Adler • Questions and answers

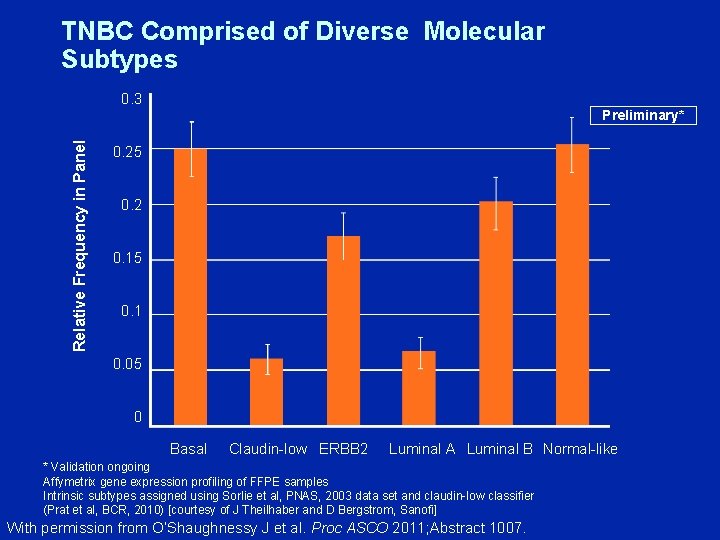
TNBC Comprised of Diverse Molecular Subtypes 0. 3 Relative Frequency in Panel Preliminary* 0. 25 0. 2 0. 15 0. 1 0. 05 0 Basal Claudin-low ERBB 2 Luminal A Luminal B Normal-like * Validation ongoing Affymetrix gene expression profiling of FFPE samples Intrinsic subtypes assigned using Sorlie et al, PNAS, 2003 data set and claudin-low classifier (Prat et al, BCR, 2010) [courtesy of J Theilhaber and D Bergstrom, Sanofi] With permission from O’Shaughnessy J et al. Proc ASCO 2011; Abstract 1007.
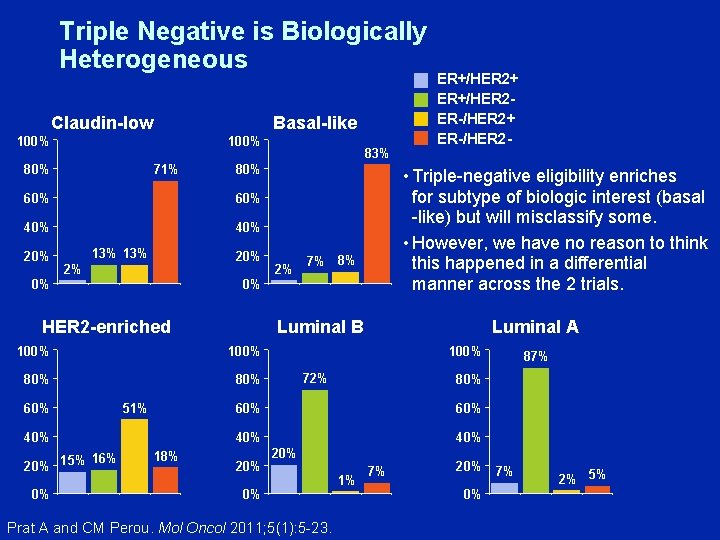
Triple Negative is Biologically Heterogeneous Claudin-low Basal-like 100% 80% 71% 80% 60% 40% 20% 13% 20% 2% 0% 100% 80% 51% 40% 0% 7% 8% Luminal B 100% 16% 20% 15% 2% • Triple-negative eligibility enriches for subtype of biologic interest (basal -like) but will misclassify some. • However, we have no reason to think this happened in a differential manner across the 2 trials. 0% HER 2 -enriched 60% 83% 18% ER+/HER 2+ ER+/HER 2 ER-/HER 2+ ER-/HER 2 - Luminal A 100% 72% 80% 60% 40% 20% 0% Prat A and CM Perou. Mol Oncol 2011; 5(1): 5 -23. 87% 1% 7% 20% 0% 7% 2% 5%

Dr Burstein (Case A) • 2/2010: 73 yo healthy, married woman with mammogram changes confirmed by ultrasound – Core biopsy: Poorly differentiated, triple-negative IDC – Exam: Palpable ALN • FNA: Positive for breast cancer • Days later: Red rash develops on underside of breast – Skin biopsy: Dermal lymphatic invasion • Staging CT scans: Positive axillary LNs, suspicious liver lesion – Liver FNA: Positive for breast cancer
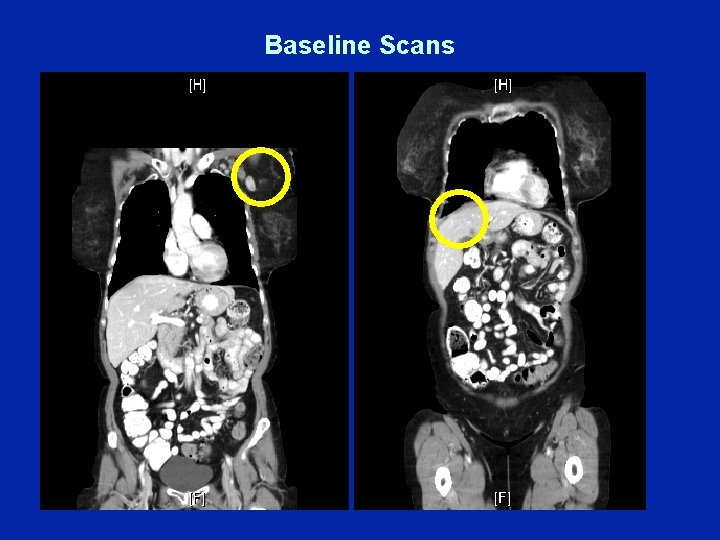
Baseline Scans

1. Generally what first-line, off-protocol therapy would you recommend?

Dr Burstein (Case A) • CALGB-40502 – Randomly assigned to ixabepilone/bevacizumab – Tolerates initial therapy well • 11/2010: Colitis symptoms, nausea, abdominal pain – CT: Non-specific colitis – Ixabepilone/bevacizumab discontinued • Paclitaxel • Summer 2011: Axillary lymphadenopathy and liver lesion decreased in size but still seen • CT: New subclinical changes in the original breast
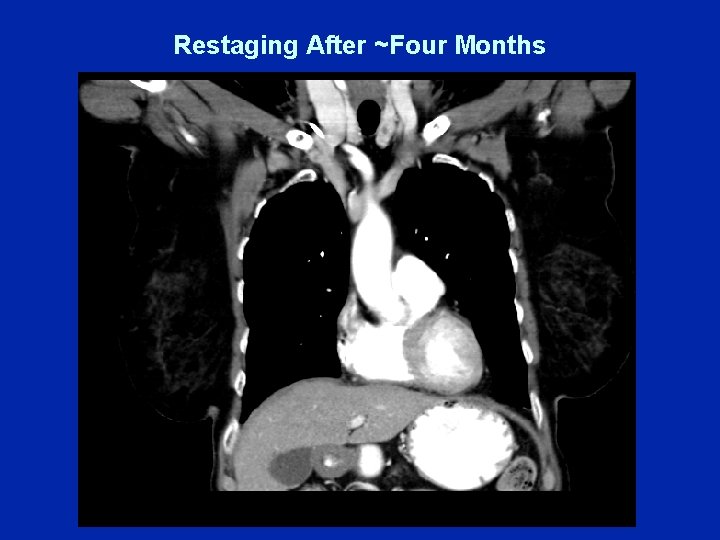
Restaging After ~Four Months
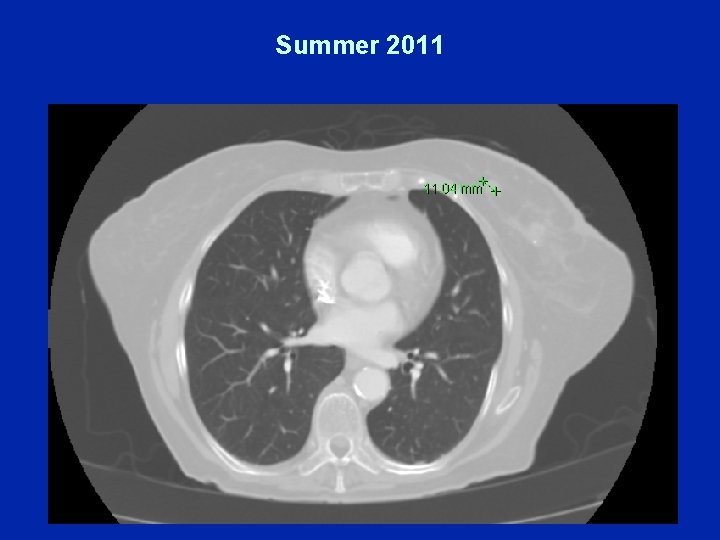
Summer 2011
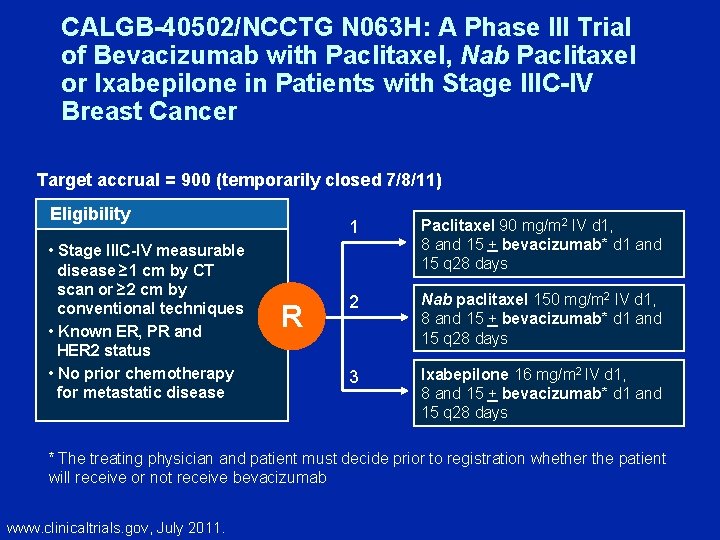
CALGB-40502/NCCTG N 063 H: A Phase III Trial of Bevacizumab with Paclitaxel, Nab Paclitaxel or Ixabepilone in Patients with Stage IIIC-IV Breast Cancer Target accrual = 900 (temporarily closed 7/8/11) Eligibility • Stage IIIC-IV measurable disease ≥ 1 cm by CT scan or ≥ 2 cm by conventional techniques • Known ER, PR and HER 2 status • No prior chemotherapy for metastatic disease R 1 Paclitaxel 90 mg/m 2 IV d 1, 8 and 15 + bevacizumab* d 1 and 15 q 28 days 2 Nab paclitaxel 150 mg/m 2 IV d 1, 8 and 15 + bevacizumab* d 1 and 15 q 28 days 3 Ixabepilone 16 mg/m 2 IV d 1, 8 and 15 + bevacizumab* d 1 and 15 q 28 days * The treating physician and patient must decide prior to registration whether the patient will receive or not receive bevacizumab www. clinicaltrials. gov, July 2011.
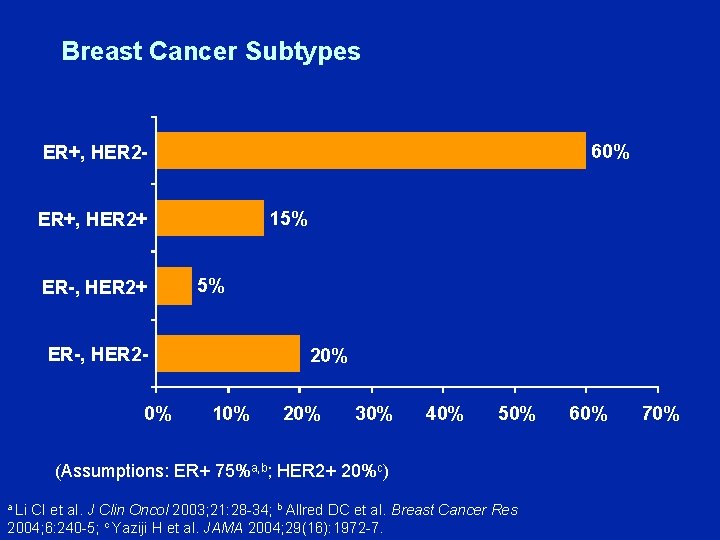
Breast Cancer Subtypes 60% ER+, HER 215% ER+, HER 2+ ER-, HER 2+ 5% ER-, HER 20% 10% 20% 30% 40% 50% (Assumptions: ER+ 75%a, b; HER 2+ 20%c) a Li CI et al. J Clin Oncol 2003; 21: 28 -34; b Allred DC et al. Breast Cancer Res 2004; 6: 240 -5; c Yaziji H et al. JAMA 2004; 29(16): 1972 -7. 60% 70%
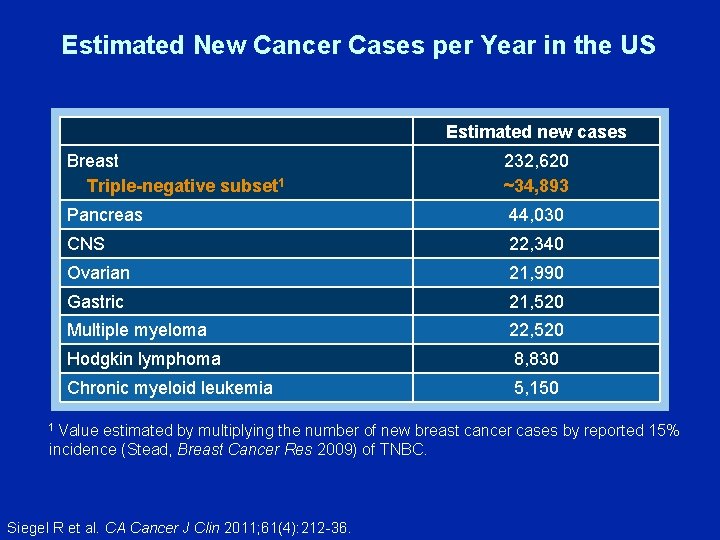
Estimated New Cancer Cases per Year in the US Estimated new cases Breast Triple-negative subset 1 232, 620 ~34, 893 Pancreas 44, 030 CNS 22, 340 Ovarian 21, 990 Gastric 21, 520 Multiple myeloma 22, 520 Hodgkin lymphoma 8, 830 Chronic myeloid leukemia 5, 150 1 Value estimated by multiplying the number of new breast cancer cases by reported 15% incidence (Stead, Breast Cancer Res 2009) of TNBC. Siegel R et al. CA Cancer J Clin 2011; 61(4): 212 -36.
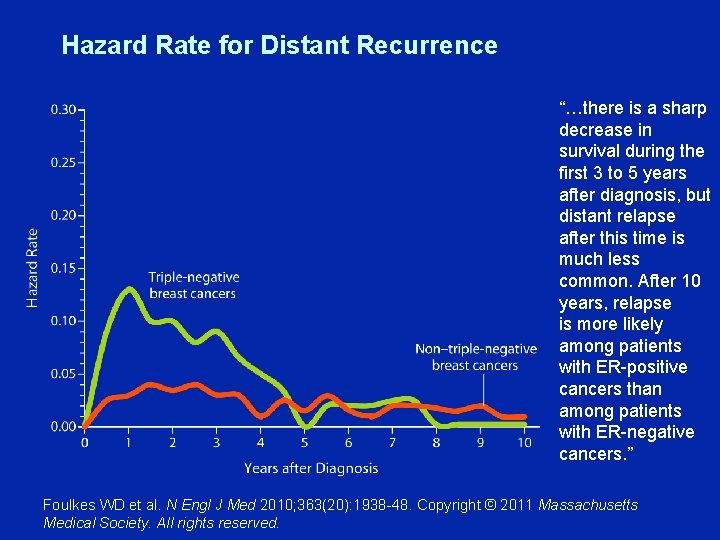
Hazard Rate for Distant Recurrence “…there is a sharp decrease in survival during the first 3 to 5 years after diagnosis, but distant relapse after this time is much less common. After 10 years, relapse is more likely among patients with ER-positive cancers than among patients with ER-negative cancers. ” Foulkes WD et al. N Engl J Med 2010; 363(20): 1938 -48. Copyright © 2011 Massachusetts Medical Society. All rights reserved.

Published Guidelines for the Management of TNBC • NCCN: No specific algorithm – NCCN Guidelines 2010, v. 2 • ESMO: No specific algorithm – Cardoso F et al. Ann Oncol 2010; 21(Suppl): v 15 v 19 – Aebi S et al. Ann Oncol 2010; 21(Suppl): v 9 -v 14 – Balmana J et al. Ann Oncol 2010; 21(Suppl): v 20 v 22 • St Gallen: No specific algorithm – Goldhirsch A et al. Ann Oncol 2009; 20: 1319 -29

The Effect of p. CR of Bevacizumab and/or Antimetabolites Added to Standard Neoadjuvant Chemotherapy: NSABP Protocol B-40 Bear HD et al. Proc ASCO 2011; Abstract LBA 1005.
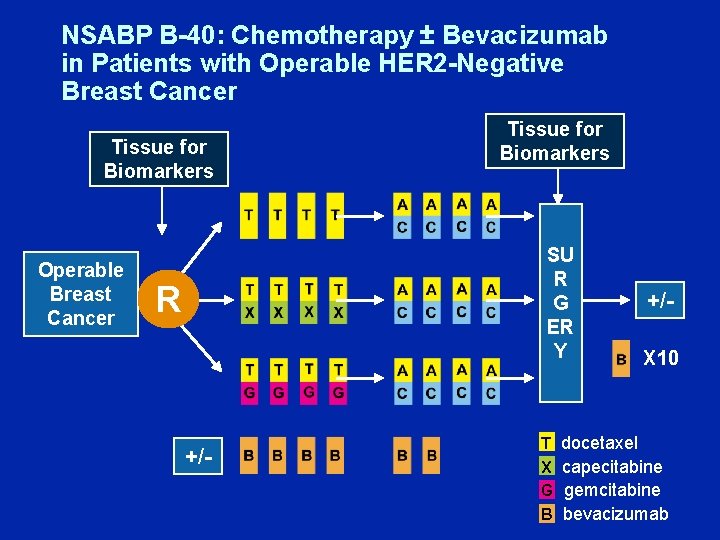
NSABP B-40: Chemotherapy ± Bevacizumab in Patients with Operable HER 2 -Negative Breast Cancer Tissue for Biomarkers Operable Breast Cancer Tissue for Biomarkers SU R G ER Y R +/- T X G B +/X 10 docetaxel capecitabine gemcitabine bevacizumab
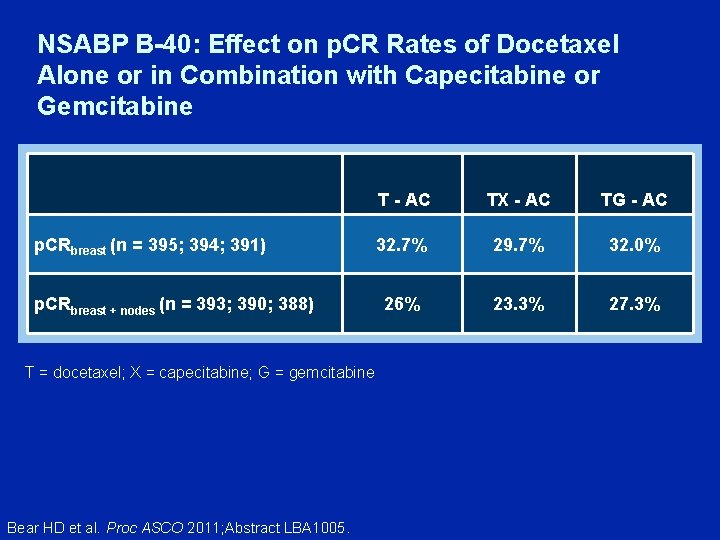
NSABP B-40: Effect on p. CR Rates of Docetaxel Alone or in Combination with Capecitabine or Gemcitabine p. CRbreast (n = 395; 394; 391) p. CRbreast + nodes (n = 393; 390; 388) T = docetaxel; X = capecitabine; G = gemcitabine Bear HD et al. Proc ASCO 2011; Abstract LBA 1005. T - AC TX - AC TG - AC 32. 7% 29. 7% 32. 0% 26% 23. 3% 27. 3%

NSABP B-40: Benefit of Adding Bevacizumab to Standard Chemotherapy • Benefit of bev predominant in HR+ and not TNBC patient subgroup • p. CRbreast (with bev vs without bev): – HR+ patients: 23. 3 vs 15. 2% (p = 0. 008) – TNBC patients: 51. 3 vs 47. 3% (p = 0. 44) Bear HD et al. Proc ASCO 2011; Abstract LBA 1005.

Neoadjuvant Bevacizumab and Anthracycline-Taxane Based Chemotherapy in 684 Triple Negative Primary Breast Cancers: Secondary Endpoint Analysis of the GEPARQUINTO Study (GBG 44) Gerber B et al. Proc ASCO 2011; Abstract 1006.
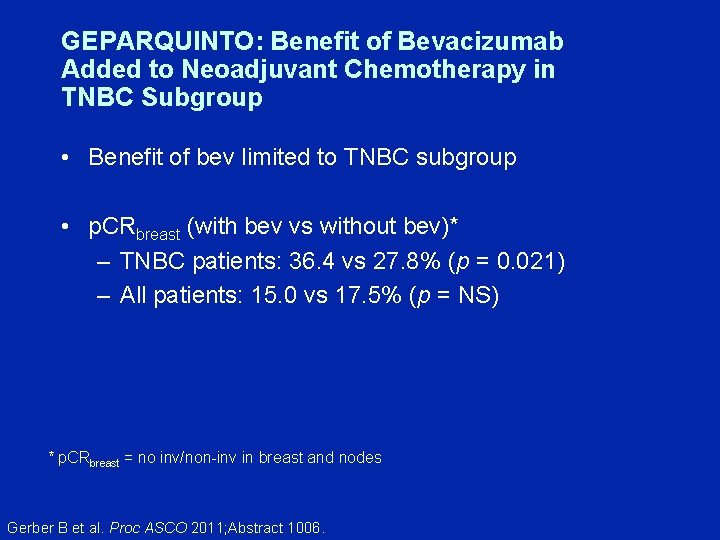
GEPARQUINTO: Benefit of Bevacizumab Added to Neoadjuvant Chemotherapy in TNBC Subgroup • Benefit of bev limited to TNBC subgroup • p. CRbreast (with bev vs without bev)* – TNBC patients: 36. 4 vs 27. 8% (p = 0. 021) – All patients: 15. 0 vs 17. 5% (p = NS) * p. CRbreast = no inv/non-inv in breast and nodes Gerber B et al. Proc ASCO 2011; Abstract 1006.
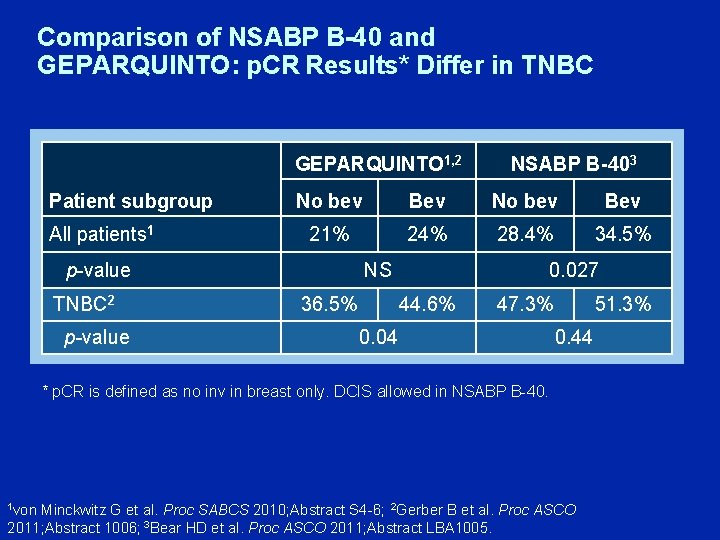
Comparison of NSABP B-40 and GEPARQUINTO: p. CR Results* Differ in TNBC GEPARQUINTO 1, 2 Patient subgroup All patients 1 No bev Bev 21% 24% 28. 4% 34. 5% p-value TNBC 2 p-value NSABP B-403 NS 36. 5% 0. 027 44. 6% 47. 3% 0. 04 51. 3% 0. 44 * p. CR is defined as no inv in breast only. DCIS allowed in NSABP B-40. 1 von Minckwitz G et al. Proc SABCS 2010; Abstract S 4 -6; 2 Gerber B et al. Proc ASCO 2011; Abstract 1006; 3 Bear HD et al. Proc ASCO 2011; Abstract LBA 1005.

CALGB-40603: The Triple-Negative Neoadjuvant Trial (TNNT) Target accrual = 362 (Open) 1 Eligibility • Stage II-IIIA resectable breast cancer ≥ 1 cm • HER 2 -negative (IHC 0 -1+ or FISH <2. 0); ER-/PR-poor • Registered on CALGB 150709 correlative tissue study www. clinicaltrials. gov, July 2011. R Paclitaxel 80 mg/m 2 x 12 dose dense (dd) AC x 4 2 Paclitaxel + dd AC as in arm 1; bevacizumab 10 mg/kg q 2 wk x 9 3 Paclitaxel + dd AC as in arm 1; carboplatin AUC 6 q 3 wk x 4 4 Paclitaxel + dd AC as in arm 1 + bevacizumab as in arm 2 + carboplatin as in arm 3
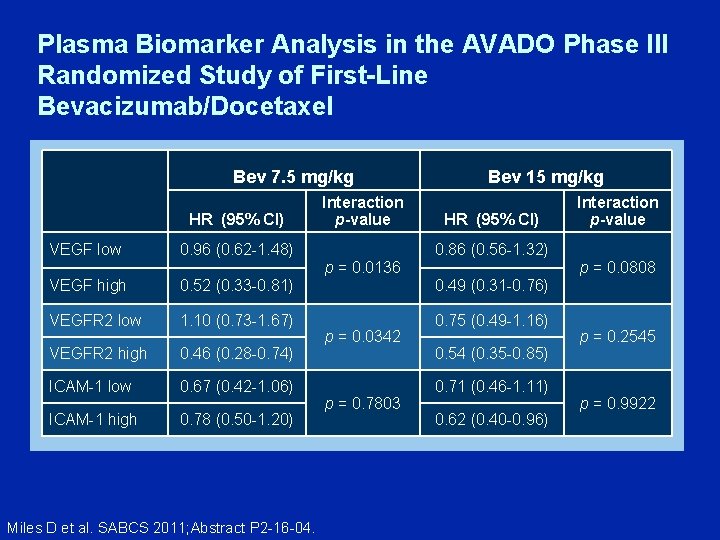
Plasma Biomarker Analysis in the AVADO Phase III Randomized Study of First-Line Bevacizumab/Docetaxel Bev 7. 5 mg/kg HR (95% CI) VEGF low Interaction p-value 0. 96 (0. 62 -1. 48) Bev 15 mg/kg HR (95% CI) 0. 86 (0. 56 -1. 32) p = 0. 0136 p = 0. 0808 VEGF high 0. 52 (0. 33 -0. 81) 0. 49 (0. 31 -0. 76) VEGFR 2 low 1. 10 (0. 73 -1. 67) 0. 75 (0. 49 -1. 16) VEGFR 2 high 0. 46 (0. 28 -0. 74) ICAM-1 low 0. 67 (0. 42 -1. 06) ICAM-1 high 0. 78 (0. 50 -1. 20) Miles D et al. SABCS 2011; Abstract P 2 -16 -04. Interaction p-value p = 0. 0342 p = 0. 7803 0. 54 (0. 35 -0. 85) 0. 71 (0. 46 -1. 11) 0. 62 (0. 40 -0. 96) p = 0. 2545 p = 0. 9922

Burstein HJ. J Clin Oncol 2011; 29(10): 1232 -5.

Impact of Bevacizumab (Bev) on Efficacy of Second-Line Chemotherapy (CT) for Triple-Negative Breast Cancer: Analysis of RIBBON-2 Brufsky A et al. Proc ASCO 2011; Abstract 1010.
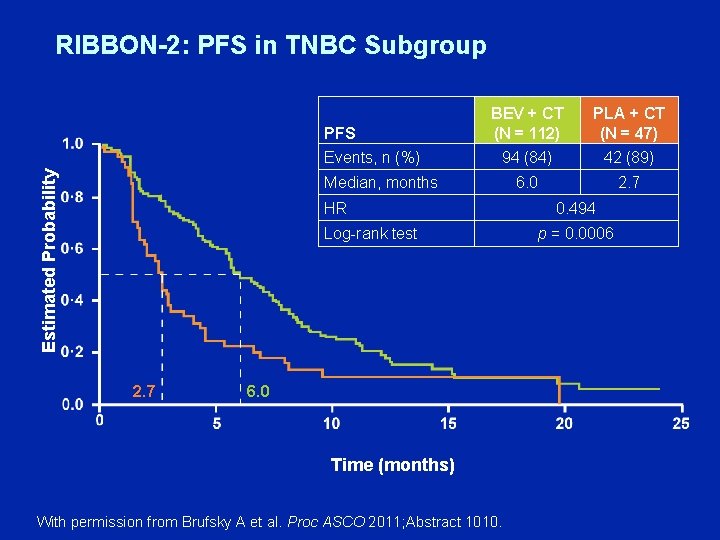
RIBBON-2: PFS in TNBC Subgroup Estimated Probability PFS Events, n (%) BEV + CT (N = 112) 94 (84) PLA + CT (N = 47) 42 (89) 6. 0 2. 7 Median, months HR Log-rank test 2. 7 6. 0 Time (months) With permission from Brufsky A et al. Proc ASCO 2011; Abstract 1010. 0. 494 p = 0. 0006
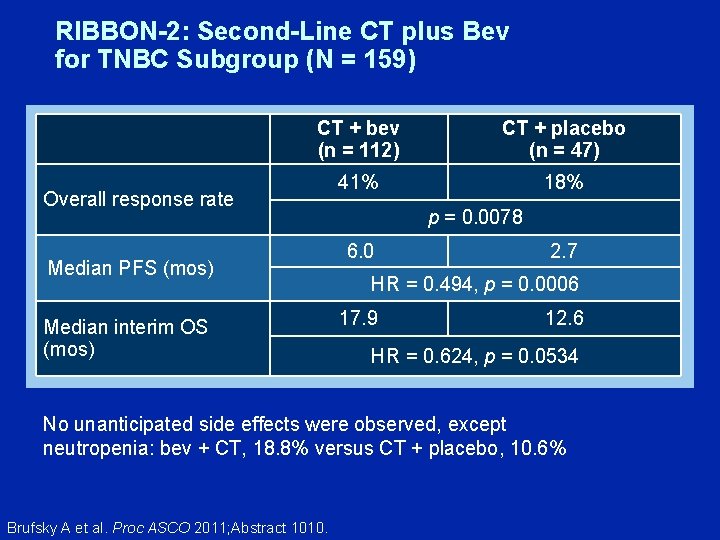
RIBBON-2: Second-Line CT plus Bev for TNBC Subgroup (N = 159) CT + bev (n = 112) CT + placebo (n = 47) 41% 18% Overall response rate Median PFS (mos) Median interim OS (mos) p = 0. 0078 6. 0 2. 7 HR = 0. 494, p = 0. 0006 17. 9 12. 6 HR = 0. 624, p = 0. 0534 No unanticipated side effects were observed, except neutropenia: bev + CT, 18. 8% versus CT + placebo, 10. 6% Brufsky A et al. Proc ASCO 2011; Abstract 1010.

Anti-angiogenic Agents Under Investigation in Breast Cancer • • • Motesanib Aflibercept IMC-18 F 1 Hu. MV 833 Ramucirumab AMG 386 Pazopanib Cabozantinib Foretinib www. clinicaltrials. gov, July 2011. • • Tivozanib Sunitinib Sorafenib Vandetanib Vatalanib Axitinib Semaximab Cediranib

Agents Currently Under Investigation for TNBC • • • Tigatuzumab Eribulin mesylate P 276 -00 Entinostat m. TOR inhibitors – Temsirolimus – Everolimus • Apatinib • NK 012 www. clinicaltrials. gov, July 2011. • Cediranib • Met. MAb • PARP inhibitors – Iniparib (BSI-201) – Olaparib (AZD-2281) – Veliparib (ABT-888) – PF-01367338 • MK 2206 • Irinotecan (patients with CNS disease)


Dr Geyer (Case B) • 11/2008: 42 yo premenopausal woman with moderately ER+, weakly PR+, HER 2 -neg T 3 N 0 M 0 poorly differentiated breast carcinoma • NSABP B-40: Completes docetaxel/gemcitabine + bevacizumab AC + bevacizumab per protocol • 7/2009: Lumpectomy + SLNB – Multifocal yp. T 2 N 1 a residual disease • 3. 6 x 2. 5 -cm: ER-neg, weakly PR+, HER 2 -neg • 1. 7 x 1. 2 -cm: Strongly ER+, moderately PR+, HER 2 -neg – 1 out of 4 SLNs positive • Post-operative seroma – RT delayed, ALND not completed – Resumed bevacizumab and RT after seroma improved • Tamoxifen and clodronate (on SWOG-S 0307) • 9/2010: Elevated estradiol, initiated leuprolide

Dr Geyer (Case B) • 3/2011: Hepatomegaly, increased LFTs – CT CAP: Extensive hepatic mets, no other sites of disease – Liver biopsy: Triple-negative IDC • Asymptomatic, tumor markers normal, CTCs = 142 • Initiated ixabepilone

Local versus Central Laboratory Discrepancies in the Determination of Triple-Negative Breast Cancer (TNBC) Status in a Large Phase III (CIBOMA/ 2004 -01/GEICAM/2003 -11) Trial Assessing Adjuvant Capecitabine (C) Maintenance Therapy After Standard Chemotherapy (CT) in Early Breast Cancer (EBC) Patients Ruiz-Borrego M et al. Proc ASCO 2011; Abstract 1022. (Poster Discussion)

Local versus Central Laboratory Discrepancies in TNBC Status (CIBOMA/2004 -01/GEICAM/2003 -11) • N = 1, 441 patient samples sent for central laboratory confirmation • In 130 cases (9%) tumors were found to not be TNBC by central determination • 71% of discrepant results involved ER or PR status • 22% of discrepant results involved HER 2 status Ruiz-Borrego M et al. Proc ASCO 2011; Abstract 1022.


Dr Burstein (Case C) • 57 yo married woman with 2 children – Family hx: Sister (40 yo), maternal grandmother – Deleterious BRCA 2 mutation • 1997: DCIS + 4 -mm focus of high-grade IDC – Left mastectomy • 1998: Right mastectomy for DCIS • 2005: Left chest wall nodule – Biopsy: High-grade triple-negative IDC – Excision chest wall RT

2. Would you generally recommend “adjuvant” systemic treatment for this patient?
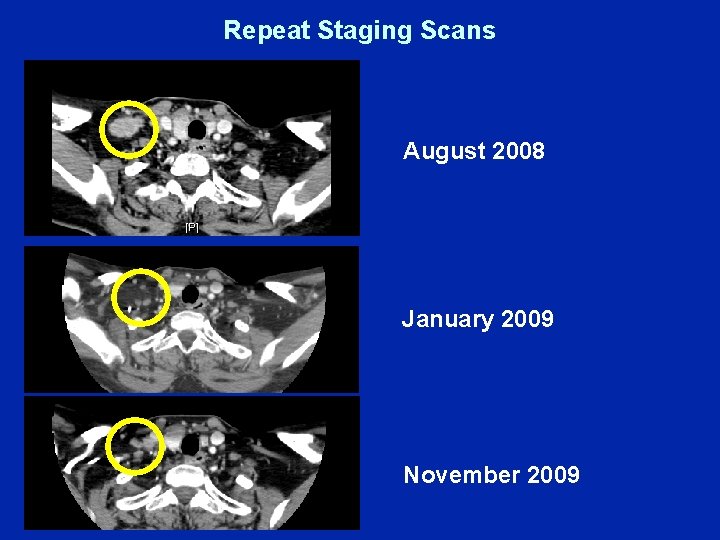
Repeat Staging Scans August 2008 January 2009 November 2009

Oral Poly(ADP-ribose) Polymerase Inhibitor Olaparib in Patients with BRCA 1 or BRCA 2 Mutations and Advanced Breast Cancer: A Proof-of-Concept Trial Tutt A et al. Lancet 2010; 376(9737): 235 -44.
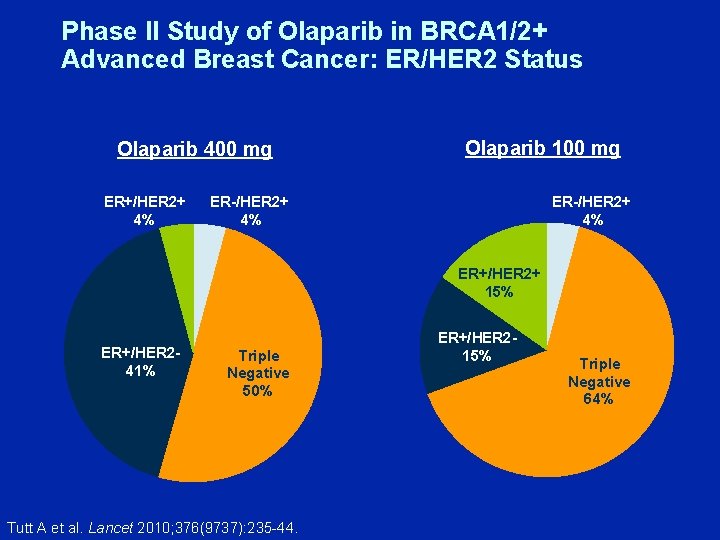
Phase II Study of Olaparib in BRCA 1/2+ Advanced Breast Cancer: ER/HER 2 Status Olaparib 400 mg ER+/HER 2+ 4% Olaparib 100 mg ER-/HER 2+ 4% ER+/HER 2+ 15% ER+/HER 241% Triple Negative 50% Tutt A et al. Lancet 2010; 376(9737): 235 -44. ER+/HER 215% Triple Negative 64%

Efficacy of Olaparib by BRCA Mutation Status Olaparib 400 mg bid Olaparib 100 mg bid BRCA 1 (n = 18) BRCA 2 (n = 9) BRCA 1 (n = 16) BRCA 2 (n = 11) Complete response, n (%) 1 (6%) 0 0 0 Partial response, n (%) 8 (44%) 2 (22%) 3 (19%) 3 (27%) Tutt A et al. Lancet 2010; 376(9737): 235 -44.

Can We Define Tumors That Will Respond to PARP Inhibitors? A Phase II Correlative Study of Olaparib in Advanced Serous Ovarian Cancer and Triple. Negative Breast Cancer Gelmon KA et al. Proc ASCO 2010; Abstract 3002.
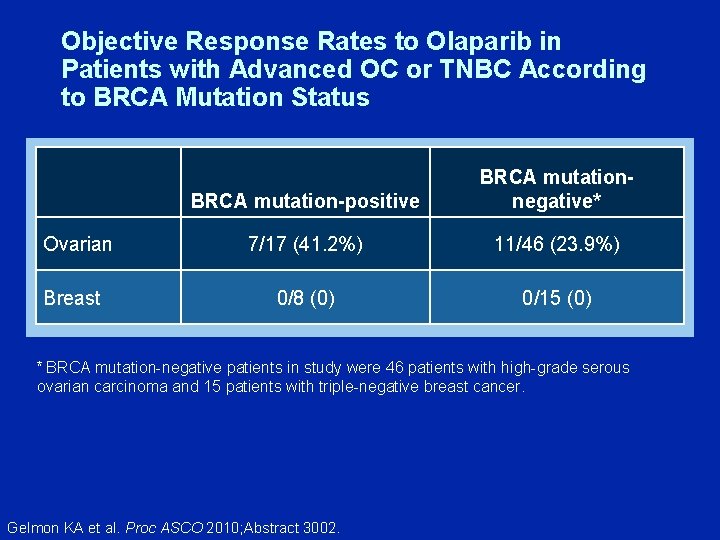
Objective Response Rates to Olaparib in Patients with Advanced OC or TNBC According to BRCA Mutation Status Ovarian Breast BRCA mutation-positive BRCA mutationnegative* 7/17 (41. 2%) 11/46 (23. 9%) 0/8 (0) 0/15 (0) * BRCA mutation-negative patients in study were 46 patients with high-grade serous ovarian carcinoma and 15 patients with triple-negative breast cancer. Gelmon KA et al. Proc ASCO 2010; Abstract 3002.

A Randomized Phase III Study of Iniparib (BSI-201) in Combination with Gemcitabine and Carboplatin in Metastatic Triple-Negative Breast Cancer (m. TNBC) O’Shaughnessy J et al. Proc ASCO 2011; Abstract 1007.
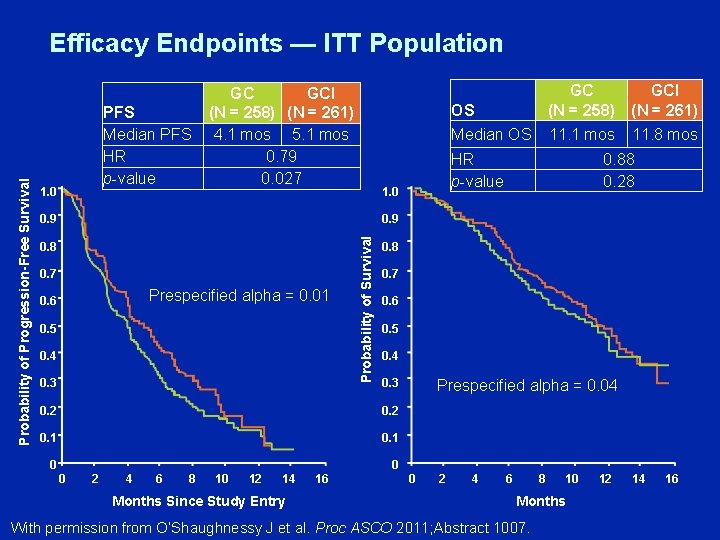
PFS Median PFS HR p-value 1. 0 GC GCI (N = 258) (N = 261) 4. 1 mos 5. 1 mos 0. 79 0. 027 0. 9 OS GC GCI (N = 258) (N = 261) Median OS 11. 1 mos HR p-value 1. 0 11. 8 mos 0. 88 0. 28 0. 9 0. 8 0. 7 Prespecified alpha = 0. 01 0. 6 0. 5 0. 4 0. 3 Probability of Survival Probability of Progression-Free Survival Efficacy Endpoints — ITT Population 0. 8 0. 7 0. 6 0. 5 0. 4 0. 3 0. 2 0. 1 0 Prespecified alpha = 0. 04 0 0 2 4 6 8 10 12 14 Months Since Study Entry 16 0 2 4 6 8 10 Months With permission from O’Shaughnessy J et al. Proc ASCO 2011; Abstract 1007. 12 14 16
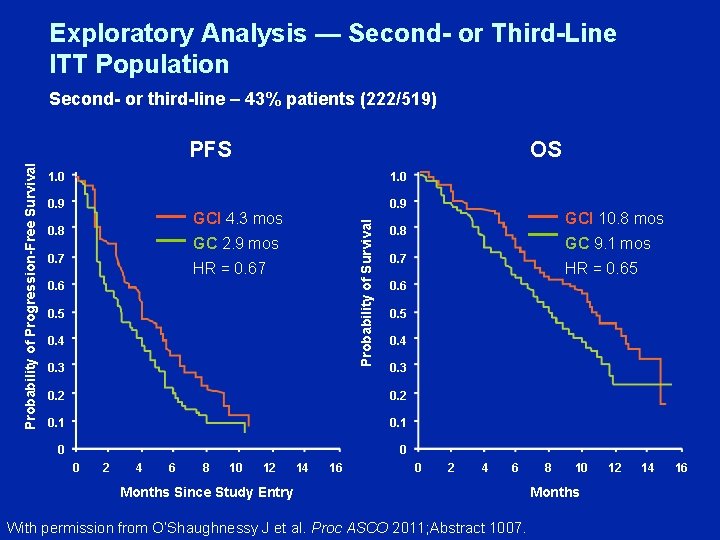
Exploratory Analysis — Second- or Third-Line ITT Population Second- or third-line – 43% patients (222/519) OS 1. 0 0. 9 GCI 4. 3 mos 0. 8 Probability of Survival Probability of Progression-Free Survival PFS GC 2. 9 mos 0. 7 HR = 0. 67 0. 6 0. 5 0. 4 0. 3 0. 1 0 0 4 6 8 10 12 14 16 HR = 0. 65 0. 6 0. 2 2 GC 9. 1 mos 0. 7 0. 2 0 GCI 10. 8 mos 0. 8 0 2 4 6 Months Since Study Entry With permission from O’Shaughnessy J et al. Proc ASCO 2011; Abstract 1007. 8 10 Months 12 14 16


Dr Geyer (Case D) • 3/2006: 75 yo woman with clinical T 3 N 1 M 0 triplenegative, Grade III IDC – 5. 5 -cm mass – Ultrasound: 2 to 3 abnormal nodes

3. In general, what neoadjuvant therapy would you recommend?

Dr Geyer (Case D) • Neoadjuvant FEC x 3 – Mass reduced to 2 x 1. 5 cm • Weekly paclitaxel 80 mg/m 2 • Mastectomy – p. CR – Free of disease after 5 years

Submitted by William Adler, MD (Case E) • 86 yo woman with hypertension, severe kyphosis and moderate renal insufficiency • Presents with a 3. 5 -cm, Grade III triple-negative breast cancer with 8 positive nodes – No evidence of other disease – Good performance status • Patient desires adjuvant systemic therapy, even for modest benefit

4. In general, which adjuvant systemic therapy would you recommend for this patient? TAC x 6 cycles TC x 4 cycles TC x 6 cycles CMF x 6 cycles Dose-dense AC followed by dosedense paclitaxel Other


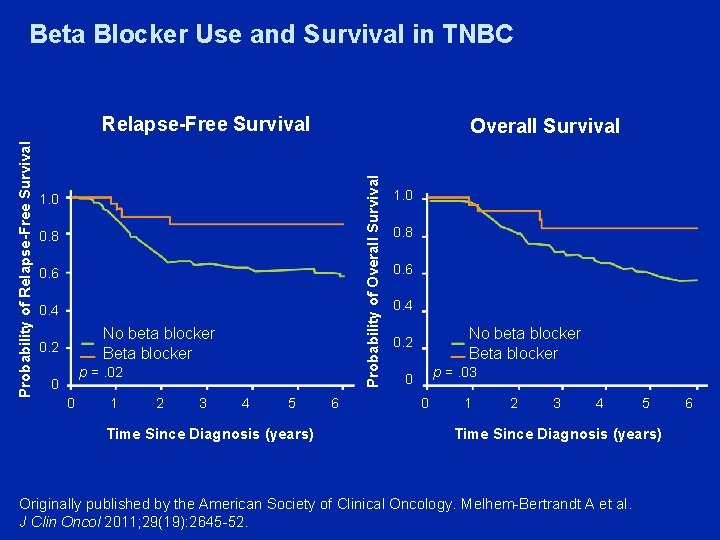
Beta Blocker Use and Survival in TNBC Overall Survival Probability of Relapse-Free Survival 1. 0 0. 8 0. 6 0. 4 No beta blocker Beta blocker 0. 2 p =. 02 0 0 1 2 3 4 5 Time Since Diagnosis (years) 6 1. 0 0. 8 0. 6 0. 4 No beta blocker Beta blocker 0. 2 p =. 03 0 0 1 2 3 4 5 Time Since Diagnosis (years) Originally published by the American Society of Clinical Oncology. Melhem-Bertrandt A et al. J Clin Oncol 2011; 29(19): 2645 -52. 6
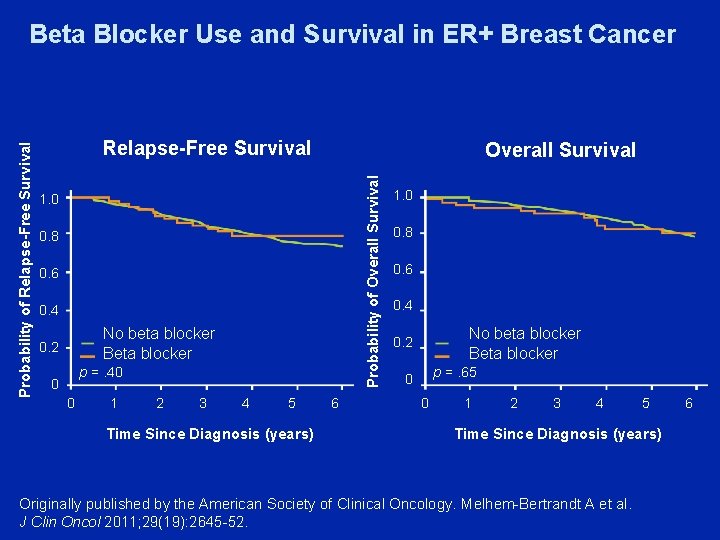
Relapse-Free Survival Overall Survival Probability of Relapse-Free Survival Beta Blocker Use and Survival in ER+ Breast Cancer 1. 0 0. 8 0. 6 0. 4 No beta blocker Beta blocker 0. 2 p =. 40 0 0 1 2 3 4 5 Time Since Diagnosis (years) 6 1. 0 0. 8 0. 6 0. 4 No beta blocker Beta blocker 0. 2 p =. 65 0 0 1 2 3 4 5 Time Since Diagnosis (years) Originally published by the American Society of Clinical Oncology. Melhem-Bertrandt A et al. J Clin Oncol 2011; 29(19): 2645 -52. 6


Schedule of Events Tuesday, July 26 Non-Hodgkin’s Lymphoma/Chronic Lymphocytic Leukemia Stephanie A Gregory, MD John P Leonard, MD Tuesday, August 2 Chronic Myeloid Leukemia Susan M O’Brien, MD Neil P Shah, MD, Ph. D
- Slides: 64