RTP TV An 8 Part Live CME Webcast

RTP TV: An 8 -Part Live CME Webcast Series Part VII – Non-Hodgkin Lymphoma/ Chronic Lymphocytic Leukemia Tuesday, July 26, 2011 7: 30 PM – 8: 30 PM ET

Neil Love, MD Research To Practice Miami, Florida Stephanie A Gregory, MD The Elodia Kehm Chair of Hematology Professor of Medicine Director, Section of Hematology Rush University Medical Center Chicago, Illinois John P Leonard, MD Richard T Silver Distinguished Professor of Hematology and Medical Oncology Professor of Medicine, Weill Cornell Medical College New York, New York

Disclosures for Moderator Neil Love, MD Dr Love is president and CEO of Research To Practice, which receives funds in the form of educational grants to develop CME activities from the following commercial interests: Allos Therapeutics, Amgen Inc, Astellas Pharma Global Development Inc, Astra. Zeneca Pharmaceuticals LP, Aureon Laboratories Inc, Bayer Health. Care Pharmaceuticals/Onyx Pharmaceuticals Inc, Biogen Idec, Boehringer Ingelheim Pharmaceuticals Inc, Bristol-Myers Squibb Company, Celgene Corporation, Cephalon Inc, Daiichi Sankyo Inc, Dendreon Corporation, Eisai Inc, EMD Serono Inc, Genentech Bio. Oncology, Genomic Health Inc, Im. Clone Systems, a wholly owned subsidiary of Eli Lilly and Company, Lilly USA LLC, Millennium: The Takeda Oncology Company, Mundipharma International Limited, Myriad Genetics Inc, Novartis Pharmaceuticals Corporation, OSI Oncology, Sanofi and Seattle Genetics

Disclosures for Stephanie A Gregory, MD Advisory Committee Amgen Inc, Cephalon Inc, Genentech Bio. Oncology, Novartis Pharmaceuticals Corporation, Spectrum Pharmaceuticals Inc Speakers Bureau Cephalon Inc

Disclosures for John P Leonard, MD Consulting Agreements Biogen Idec, Bristol-Myers Squibb Company, Celgene Corporation, Cephalon Inc, EMD Serono Inc, Genentech Bio. Oncology, Glaxo. Smith. Kline, Millennium: The Takeda Oncology Company, Novartis Pharmaceuticals Corporation, Pfizer Inc, Sanofi

Agenda — Non-Hodgkin Lymphoma/ Chronic Lymphocytic Leukemia • Module 1: Follicular Lymphoma (FL) • Module 2: Chronic Lymphocytic Leukemia (CLL) • Module 3: Mantle-Cell Lymphoma (MCL) • Module 4: Diffuse Large B-Cell Lymphoma (DLBCL) • Questions and answers


Dr Gregory (Case A): Follicular Lymphoma • 6/2004: 54 yo man with diffuse supraclavicular, bilateral axillary and inguinal and intra-abdominal adenopathy, largest 2. 8 cm • Lab results: All normal, including LDH • Inguinal node bx: Follicular lymphoma Grade II/III • BM bx: Positive for lymphoma (20%) • FLIPI score: 2
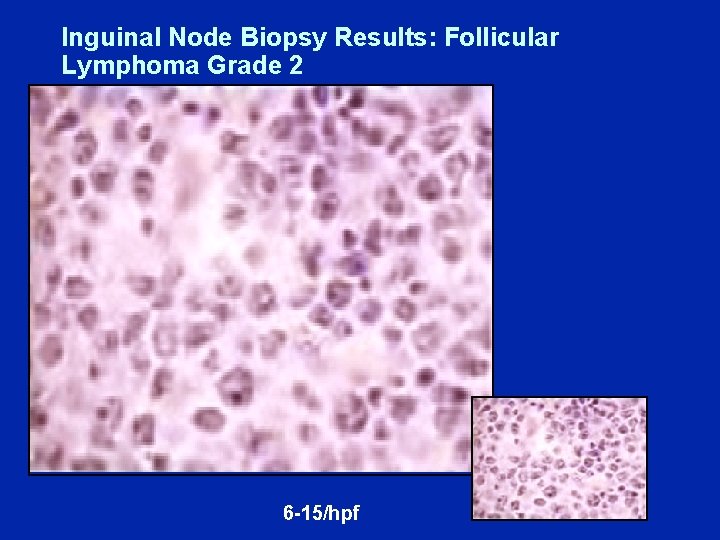
Inguinal Node Biopsy Results: Follicular Lymphoma Grade 2 6 -15/hpf

1. If this patient presented to you now in 2011, which first-line therapy would you generally recommend?
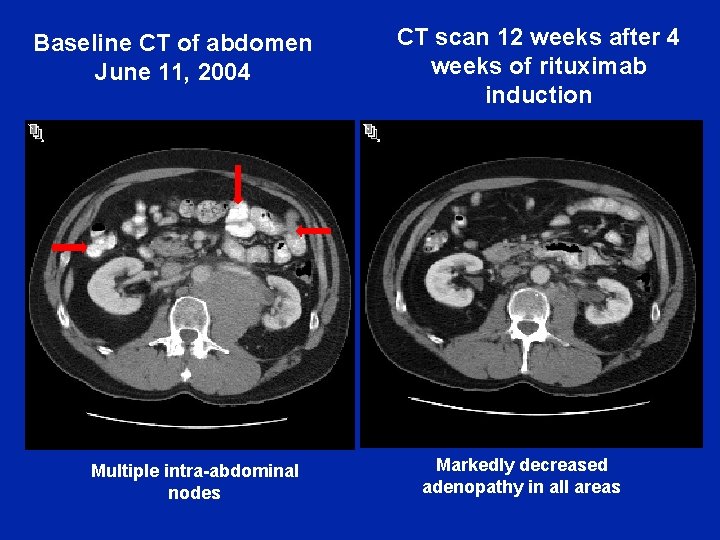
Baseline CT of abdomen June 11, 2004 Multiple intra-abdominal nodes CT scan 12 weeks after 4 weeks of rituximab induction Markedly decreased adenopathy in all areas

N = 600+ patients – closed ECOG 4402 RESORT Trial R E G I S T E R Induction rituximab 375 mg/m 2 weekly x 4 Restageweek 12 End study treatment ≥PR/CR <PR or PD R A N D O M I Z E Rituximab maintenance Single 375 mg/m 2 IV q 12 wk Continue to rituximab failure Rituximab re-treatment at progression Single 375 mg/m 2 IV q 4 wk Continue to rituximab failure Primary endpoint is time to rituximab failure

Dr Gregory (Case A): Follicular Lymphoma • 6/21/2011: Remains asymptomatic in complete remission • Labs: Normal except for mildly decreased Ig. G (794), Ig. A (144) and Ig. M (21) • Patient has had no infections • CT scans every 6 months – no evidence of disease • Remains on clinical trial w/o Rx interruption

An Intergroup Randomised Trial of Rituximab vs a Watch & Wait Approach in Patients with Advanced Stage, Asymptomatic, Non-Bulky Follicular Lymphoma Kirit M Ardeshna, Paul Smith, Wendi Qian, June Warden, Lindsey Stevens, Christopher FE Pocock, Fiona Miall, David Cunningham, John Davies, Andrew Jack, Jan Walewski, A Burhan Ferhanoglu, Ken Bradstock and David C Linch Ardeshna KM et al. Proc ASH 2010; Abstract 6.
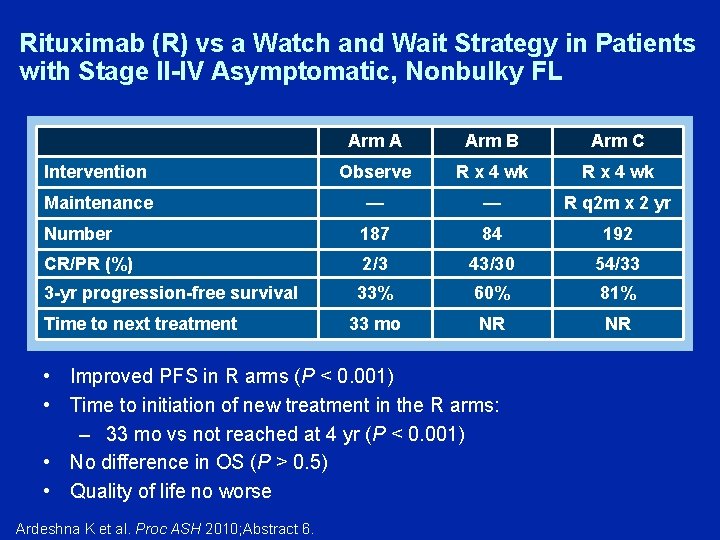
Rituximab (R) vs a Watch and Wait Strategy in Patients with Stage II-IV Asymptomatic, Nonbulky FL Arm A Arm B Arm C Intervention Observe R x 4 wk Maintenance — — R q 2 m x 2 yr Number 187 84 192 CR/PR (%) 2/3 43/30 54/33 33% 60% 81% 33 mo NR NR 3 -yr progression-free survival Time to next treatment • Improved PFS in R arms (P < 0. 001) • Time to initiation of new treatment in the R arms: – 33 mo vs not reached at 4 yr (P < 0. 001) • No difference in OS (P > 0. 5) • Quality of life no worse Ardeshna K et al. Proc ASH 2010; Abstract 6.
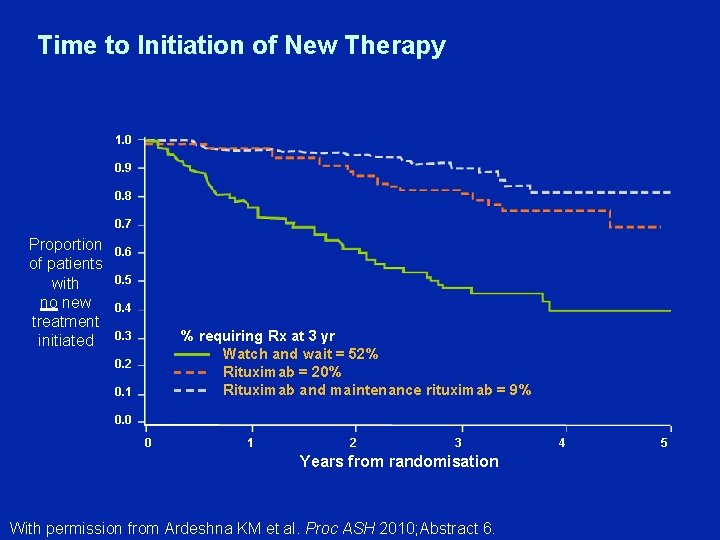
Time to Initiation of New Therapy 1. 0 0. 9 0. 8 0. 7 Proportion of patients with no new treatment initiated 0. 6 0. 5 0. 4 % requiring Rx at 3 yr Watch and wait = 52% Rituximab = 20% Rituximab and maintenance rituximab = 9% 0. 3 0. 2 0. 1 0. 0 0 1 2 3 Years from randomisation With permission from Ardeshna KM et al. Proc ASH 2010; Abstract 6. 4 5

Preliminary Results of Quality of Life (QOL) Analyses from the Intergroup Phase III Randomised Trial of Rituximab vs a Watch and Wait Approach in Patients with Advanced Stage, Asymptomatic, Non -Bulky Follicular Lymphoma (FL) Ardeshna KM et al. International Conference on Malignant Lymphoma 2011.


PRIMA: Rituximab Maintenance After R-Chemo • Two years of treatment: Every 8 weeks • Three-year progression-free survival R maintenance: 75% Control: 58% • Gr 3 -4 infection R maintenance: 24% Control: 17% • Treatment discontinued R maintenance: 4% Salles G et al. Lancet 2011; 377(9759): 42 -51. Control: 2%

PRIMA: Progression-Free Survival Progression-free survival Rituximab maintenance Observation HR p-value 74. 9% 57. 6% 0. 55 <0. 0001 Median follow-up of 36 months Salles G et al. Lancet 2011; 377(9759): 42 -51.

PRIMA: Quality of Life • EORTC QLQ-C 30 global health status mean scores in rituximab maintenance vs observation: – 75. 5 (95% CI 72. 8 -78. 2) vs 75. 2 (95% CI 72. 0 -78. 4), p-value = 0. 89 • Mean adjusted FACT-G total scores at the end of treatment in rituximab maintenance vs observation: – 86. 6 (95% CI 85. 0 -88. 3) vs 87. 2 (95% CI 85. 3 -89. 1), p-value = 0. 68 Salles G et al. Lancet 2011; 377(9759): 42 -51.
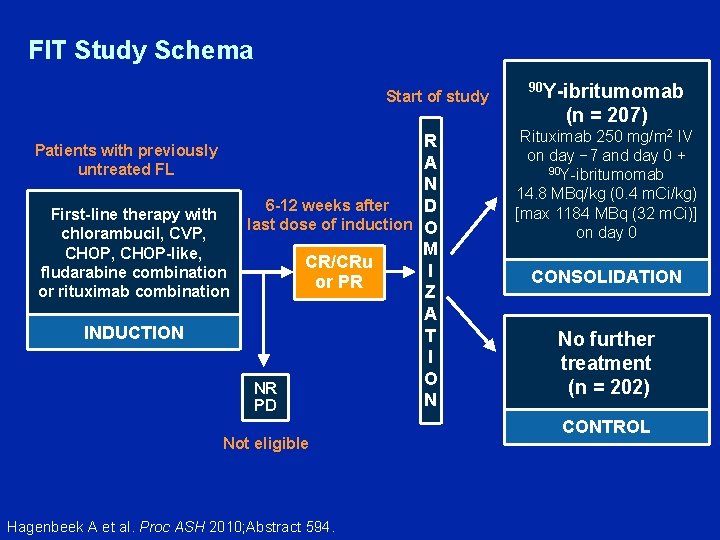
FIT Study Schema Start of study R Patients with previously A untreated FL N 6 -12 weeks after D First-line therapy with last dose of induction O chlorambucil, CVP, M CHOP, CHOP-like, CR/CRu I fludarabine combination or PR or rituximab combination Z A INDUCTION T I O NR N PD Not eligible Hagenbeek A et al. Proc ASH 2010; Abstract 594. 90 Y-ibritumomab (n = 207) Rituximab 250 mg/m 2 IV on day − 7 and day 0 + 90 Y-ibritumomab 14. 8 MBq/kg (0. 4 m. Ci/kg) [max 1184 MBq (32 m. Ci)] on day 0 CONSOLIDATION No further treatment (n = 202) CONTROL
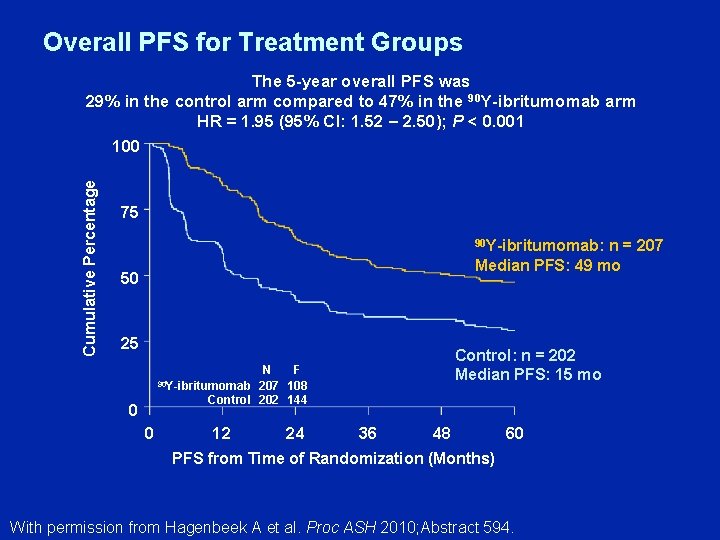
Overall PFS for Treatment Groups Cumulative Percentage The 5 -year overall PFS was 29% in the control arm compared to 47% in the 90 Y-ibritumomab arm HR = 1. 95 (95% CI: 1. 52 – 2. 50); P < 0. 001 100 75 90 Y-ibritumomab: n = 207 Median PFS: 49 mo 50 25 Control: n = 202 Median PFS: 15 mo N F 90 Y-ibritumomab 207 108 Control 202 144 0 0 12 24 36 48 60 PFS from Time of Randomization (Months) With permission from Hagenbeek A et al. Proc ASH 2010; Abstract 594.

• N = 676 • Rituximab (n = 340) vs bortezomib/rituximab (n = 336) • Progression-free survival: 11. 0 vs 12. 8 months, HR = 0. 82, p-value = 0. 039 • ≥Gr 3 AEs: 21% vs 46% • Serious AEs: 11% vs 18%
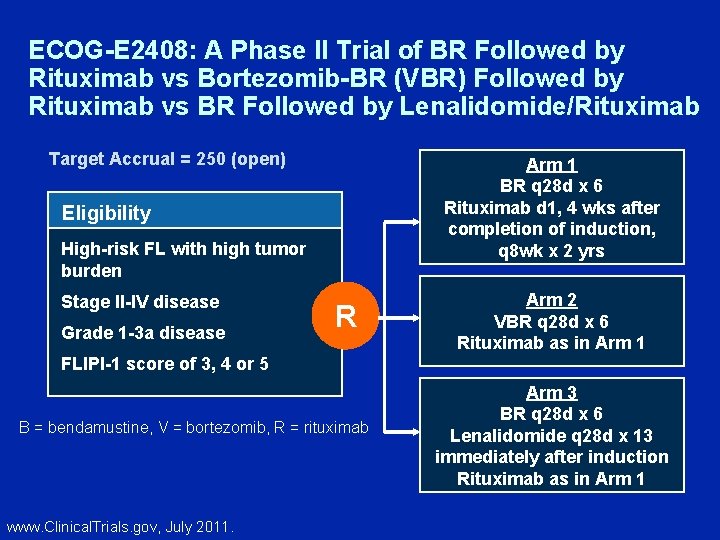
ECOG-E 2408: A Phase II Trial of BR Followed by Rituximab vs Bortezomib-BR (VBR) Followed by Rituximab vs BR Followed by Lenalidomide/Rituximab Target Accrual = 250 (open) Arm 1 BR q 28 d x 6 Rituximab d 1, 4 wks after completion of induction, q 8 wk x 2 yrs Eligibility High-risk FL with high tumor burden Stage II-IV disease Grade 1 -3 a disease R Arm 2 VBR q 28 d x 6 Rituximab as in Arm 1 FLIPI-1 score of 3, 4 or 5 B = bendamustine, V = bortezomib, R = rituximab www. Clinical. Trials. gov, July 2011. Arm 3 BR q 28 d x 6 Lenalidomide q 28 d x 13 immediately after induction Rituximab as in Arm 1

Dr Leonard (Case B): Follicular Lymphoma • 8/2006: Currently 61 yo otherwise healthy female artist w/2 -cm groin node; asymptomatic • Bx: Follicular Grade II/III • Imaging: Diffuse lymphadenopathy (LAN) 2 -3 cm range • Observed 3 years, slowly progressive disease • Imaging: Mass over 12 cm • Labs remain normal including LDH

What is currently your usual preferred regimen for a patient with FL who requires initial treatment? 58 -year-old 75 -year-old R-CHOP 32% 5% R-bendamustine 36% 34% R-CVP 26% 32% Rituximab monotherapy 1% 24% Patterns of Care in Medical Oncology: Management of NHL and CLL 2010.

Dr Leonard (Case B): Follicular Lymphoma • Bendamustine/rituximab therapy is initiated • Excellent response, 75% LAN reduction, no symptoms • Tolerated well but dose reduction in last 2 cycles – Mild cytopenias, fatigue, nausea, IV hydration needed • 9/2010: R maintenance initiated every 2 months • Patient doing well
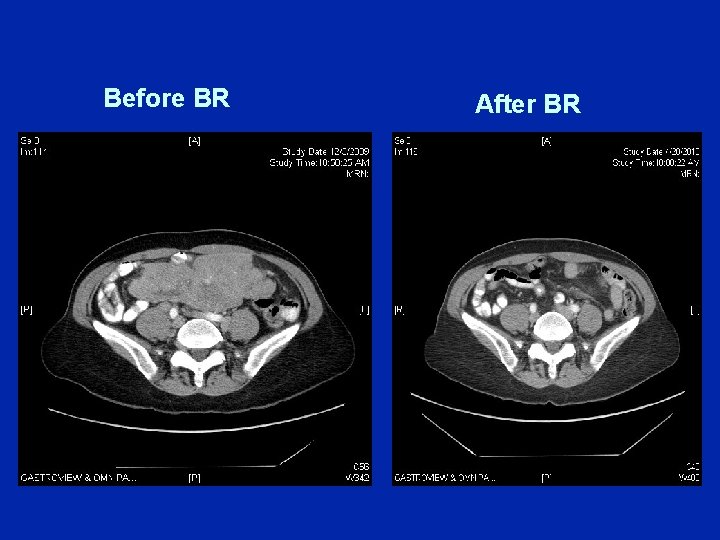
Before BR After BR

RAPID-FIRE QUESTIONS

Follicular Lymphoma • 59 yo man with bulky follicular lymphoma but minimally symptomatic. What is the optimal regimen? • After bendamustine fails, what is the next step? • Does rituximab change the natural history of follicular lymphoma? • Patient with poor performance status whose FL progressed on rituximab monotherapy

Dr Leonard (Case C): CLL • Currently 75 yo married businessman presents with mild lymphoadenopathy and lymphocytosis • PMH: Diabetes, hyperlipidemia • Followed off therapy • Develops progressive lymphocytosis (70 k) anemia, splenomegaly, fatigue • Trisomy 12 (intermediate risk)

2. Which first-line therapy would you generally recommend for this patient?
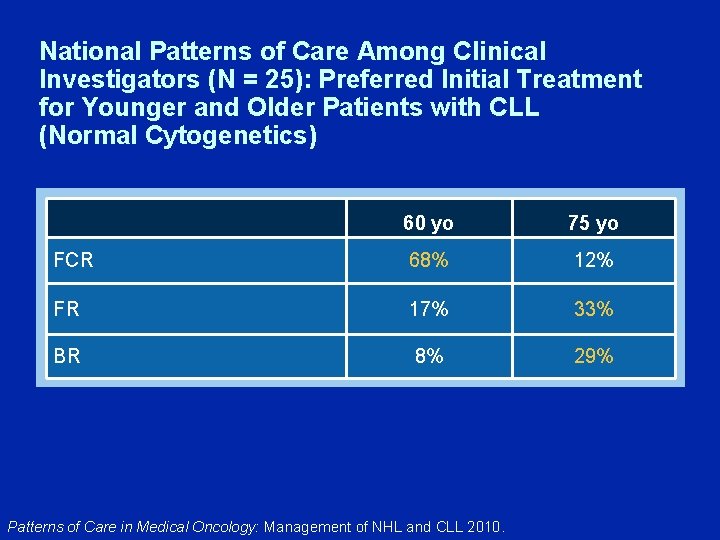
National Patterns of Care Among Clinical Investigators (N = 25): Preferred Initial Treatment for Younger and Older Patients with CLL (Normal Cytogenetics) 60 yo 75 yo FCR 68% 12% FR 17% 33% BR 8% 29% Patterns of Care in Medical Oncology: Management of NHL and CLL 2010.

Dr Leonard (Case C): CLL • F-R x 6 cycles (delayed cycle 6) • Tolerated well, mild cytopenias • Patient is active with minimal symptoms

Informative for Prognosis or Therapy Decision. Making? • • • Cytogenetics and/or FISH – t(11; 14) – t(11 q; v) – +12 – del(13 q) – del(17 p) Molecular Genetic Analysis – Immunoglobulin heavy chain variable gene (Ig. HV) mutation status Flow Cytometry or Immunohistochemistry – CD 38 – Zap 70

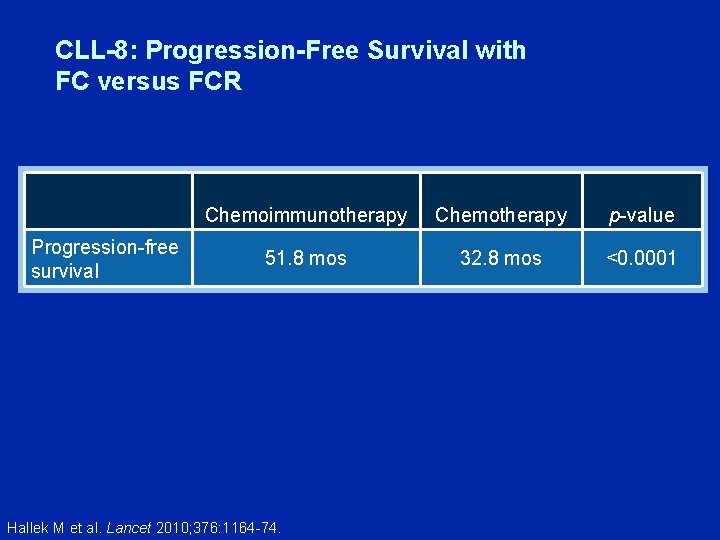
CLL-8: Progression-Free Survival with FC versus FCR Progression-free survival Chemoimmunotherapy Chemotherapy p-value 51. 8 mos 32. 8 mos <0. 0001 Hallek M et al. Lancet 2010; 376: 1164 -74.
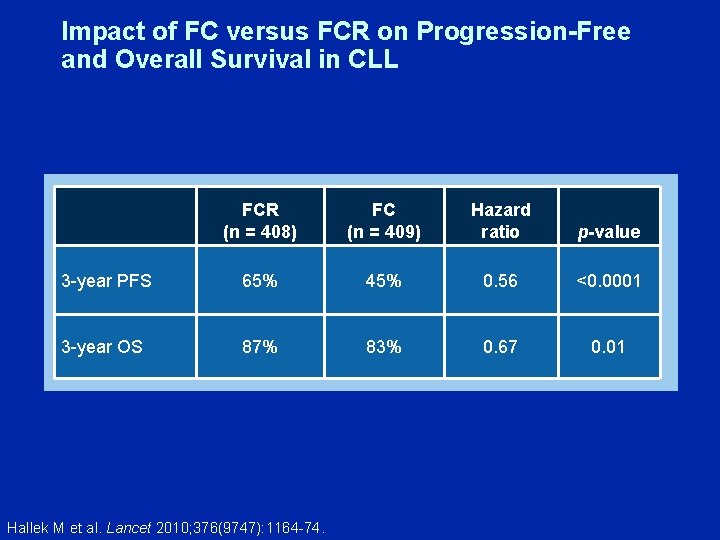
Impact of FC versus FCR on Progression-Free and Overall Survival in CLL FCR (n = 408) FC (n = 409) Hazard ratio p-value 3 -year PFS 65% 45% 0. 56 <0. 0001 3 -year OS 87% 83% 0. 67 0. 01 Hallek M et al. Lancet 2010; 376(9747): 1164 -74.

Phase III Trial of Combined Immunochemotherapy with FCR versus BR in Previously Untreated CLL Target Accrual = 550 (open) Eligibility Untreated Binet C CLL or Binet B or A with ≥ one of B-symptoms, progressive lymphocytosis, marrow failure; massive, progressive or painful splenomegaly; or massive lymph nodes or nodal clusters No 17 p deletion by FISH German CLL Study Group www. Clinical. Trials. gov, April 2011. FCR R BR

RAPID-FIRE QUESTIONS

Chronic Lymphocytic Leukemia • FCR versus BR for up-front therapy: Which agents, when and for whom? • What is the recommended dose of bendamustine for elderly patients? • Is there still a role for single-agent chlorambucil?

Chronic Lymphocytic Leukemia • What is the optimal use of alemtuzumab in CLL? • Role of bone marrow biopsy in CLL • Guidelines regarding re-treatment with rituximab: Role of ofatumumab

Dr Gregory (Case D): Mantle-Cell Lymphoma • 2/2008: 61 yo male w/progressive asymptomatic lymphadenopathy in left neck and right groin • 8/2008: Lymph node bx = MCL – Staging workup – CT CAP: Diffuse cervical, axillary, inguinal and mesenteric adenopathy, largest 4. 7 cm in the inguinal area – Labs: Normal including LDH and beta 2 microglobulin – BM bx: 10% involvement by lymphoma
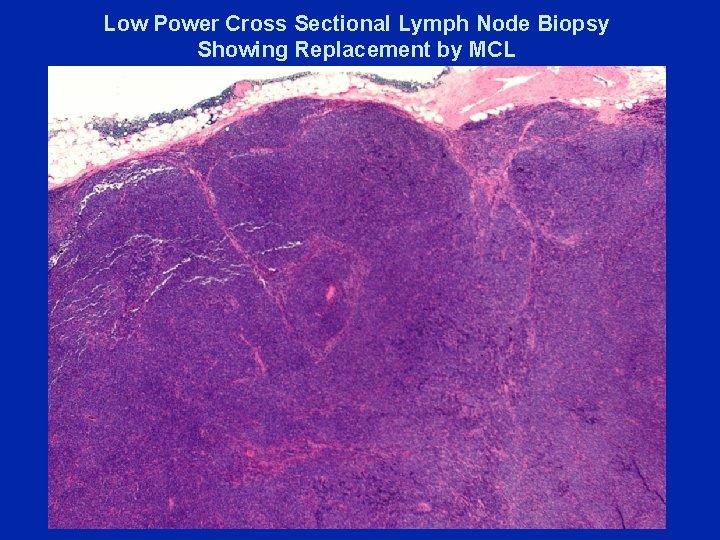
Low Power Cross Sectional Lymph Node Biopsy Showing Replacement by MCL
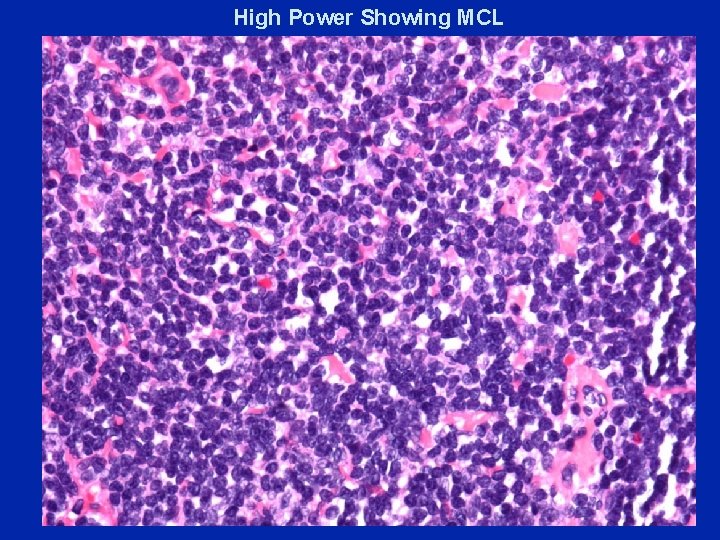
High Power Showing MCL
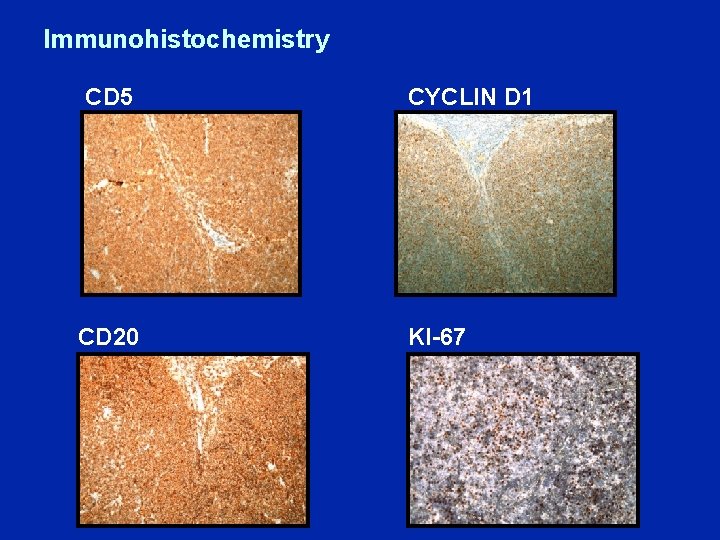
Immunohistochemistry CD 5 CD 20 CYCLIN D 1 KI-67
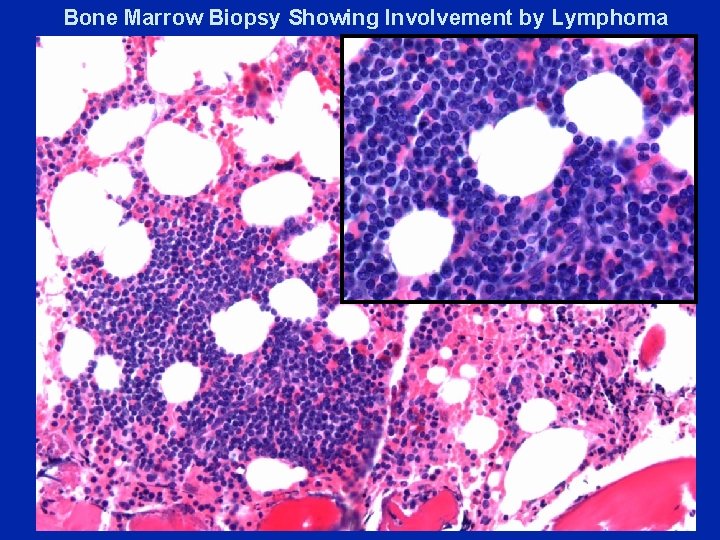
Bone Marrow Biopsy Showing Involvement by Lymphoma
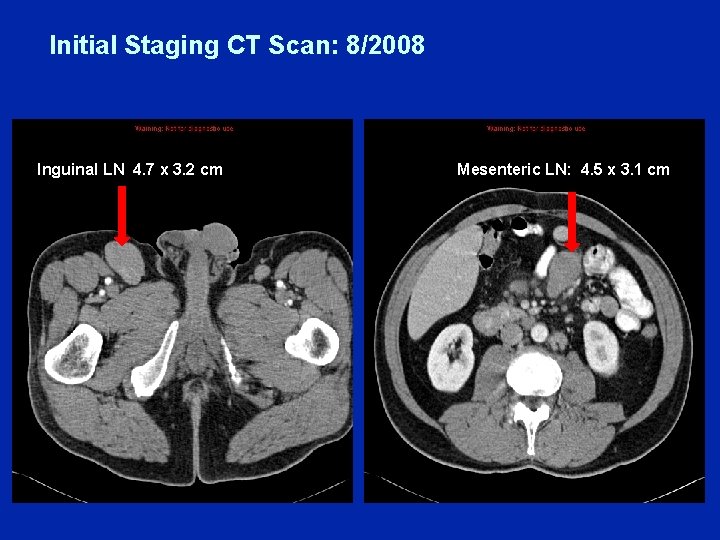
Initial Staging CT Scan: 8/2008 Inguinal LN 4. 7 x 3. 2 cm Mesenteric LN: 4. 5 x 3. 1 cm

3. Which treatment would you generally recommend for this patient as first-line therapy? R-CHOP ASCT R-hyper-CVAD or other Ara-C-containing regimen BR Other

Dr Gregory (Case D): Mantle-Cell Lymphoma • 8/2008 - 1/2009: Patient treated on ECOG-E 1405 – 8/2008 - 1/2009: Randomized to Vc. R-CVAD – Randomized to SCT w/o rituximab maintenance • 4/2011: Patient remains in CR as of last office visit
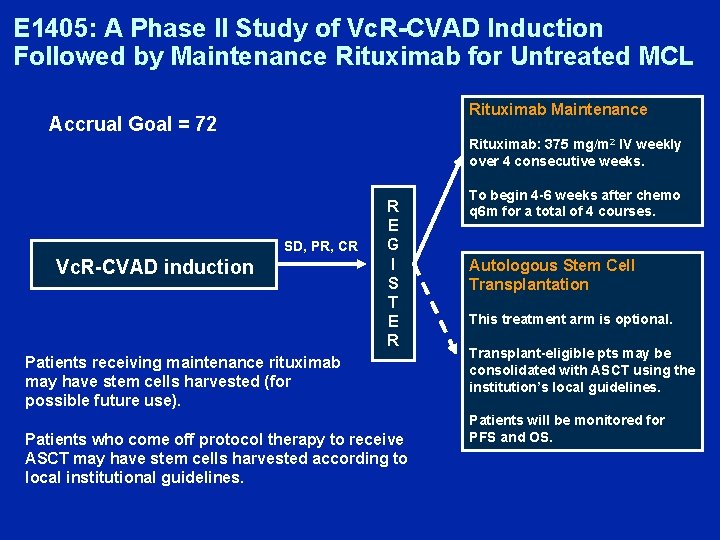
E 1405: A Phase II Study of Vc. R-CVAD Induction Followed by Maintenance Rituximab for Untreated MCL Rituximab Maintenance Accrual Goal = 72 Rituximab: 375 mg/m 2 IV weekly over 4 consecutive weeks. SD, PR, CR Vc. R-CVAD induction R E G I S T E R Patients receiving maintenance rituximab may have stem cells harvested (for possible future use). Patients who come off protocol therapy to receive ASCT may have stem cells harvested according to local institutional guidelines. To begin 4 -6 weeks after chemo q 6 m for a total of 4 courses. Autologous Stem Cell Transplantation This treatment arm is optional. Transplant-eligible pts may be consolidated with ASCT using the institution’s local guidelines. Patients will be monitored for PFS and OS.
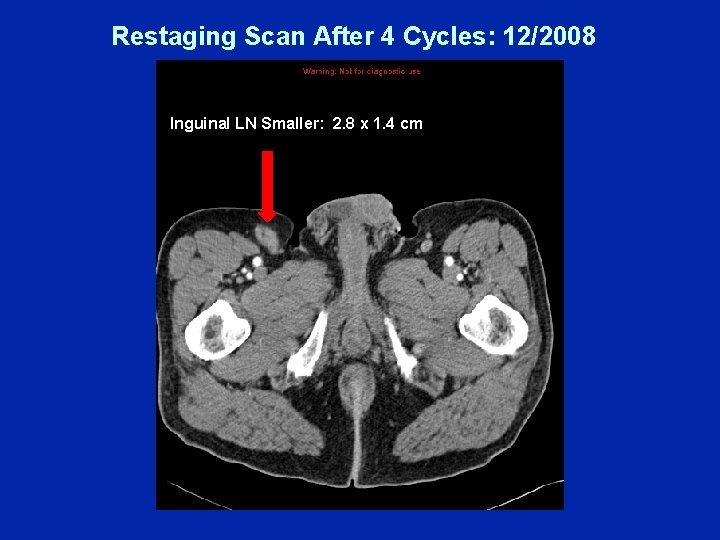
Restaging Scan After 4 Cycles: 12/2008 Inguinal LN Smaller: 2. 8 x 1. 4 cm
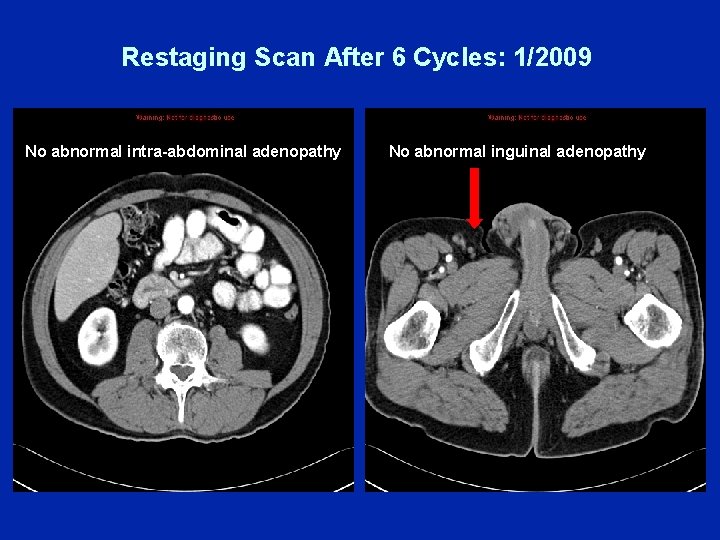
Restaging Scan After 6 Cycles: 1/2009 No No abnormal intra-abdominal adenopathy No abnormal inguinal adenopathy

Rituximab Maintenance Significantly Prolongs Duration of Remission in Elderly Patients with Mantle Cell Lymphoma. First Results of a Randomized Trial of the European MCL Network Kluin-Nelemans J et al. European Hematology Association 2011; Presidential Symposium.
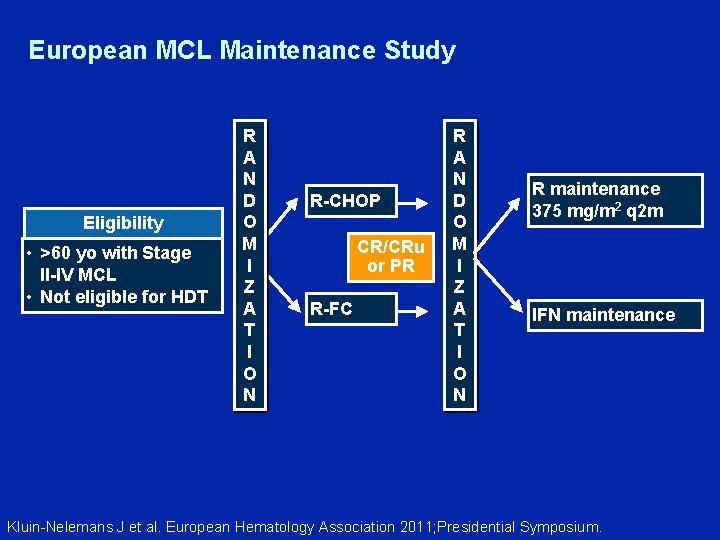
European MCL Maintenance Study Eligibility • >60 yo with Stage II-IV MCL • Not eligible for HDT R A N D O M I Z A T I O N R-CHOP CR/CRu or PR R-FC R A N D O M I Z A T I O N R maintenance 375 mg/m 2 q 2 m IFN maintenance Kluin-Nelemans J et al. European Hematology Association 2011; Presidential Symposium.
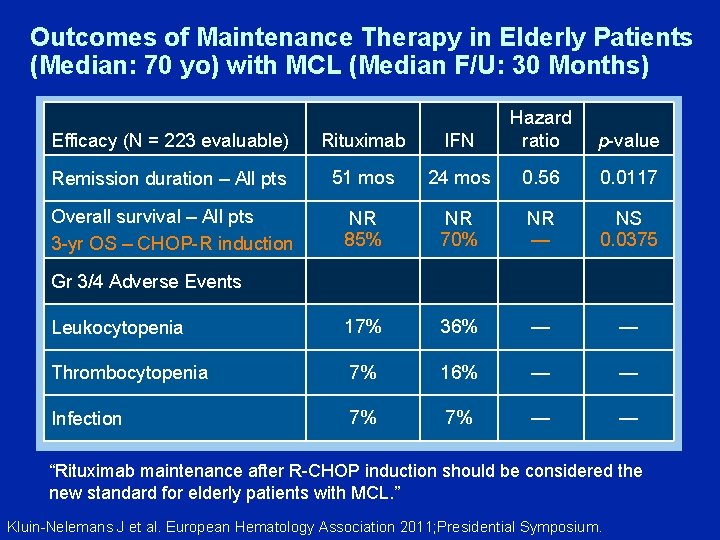
Outcomes of Maintenance Therapy in Elderly Patients (Median: 70 yo) with MCL (Median F/U: 30 Months) Efficacy (N = 223 evaluable) Rituximab IFN Hazard ratio Remission duration – All pts 51 mos 24 mos 0. 56 0. 0117 Overall survival – All pts 3 -yr OS – CHOP-R induction NR 85% NR 70% NR — NS 0. 0375 Leukocytopenia 17% 36% — — Thrombocytopenia 7% 16% — — Infection 7% 7% — — p-value Gr 3/4 Adverse Events “Rituximab maintenance after R-CHOP induction should be considered the new standard for elderly patients with MCL. ” Kluin-Nelemans J et al. European Hematology Association 2011; Presidential Symposium.

A Randomized Phase II Trial of Maintenance vs Consolidation Bortezomib Therapy Following Aggressive Chemo-Immunotherapy and ASCT for Previously Untreated MCL Protocol ID: CALGB-50403 Target Accrual = 150 (active, not recruiting) Treatment Chemo-Immunotherapy and ASCT www. Clinical. Trials. gov, April 2011. Bortezomib D 1, 8, 15, 22 every 56 days x 10 R Bortezomib D 1, 4, 8, 11 every 21 days x 4

Dr Leonard (Case E): Mantle-Cell Lymphoma • Currently 66 yo married female teacher – 1998: Presents with lymphocytosis, splenomegaly, originally CLL suspected • 2007: Dx of MCL on positive FISH for t(11; 14) • Watched for 9 years • 2/2010: Increased lymphocytosis, splenomegaly, anemia
Dr Leonard (Case E): Mantle-Cell Lymphoma • 2/2010: CHOP-R x 3 cycles, minimal response progression • Oral PEP-C x 2 months progression • BR x 6 cycles, excellent response for 4 months – Progresses 7/2010 • Part B R-hyper-CVAD (methotrexate/ARA-C) x 1 cycle, not tolerated well • 3/2011: Btk inhibitor (PCI-32765) on study – Excellent response for past 3 months
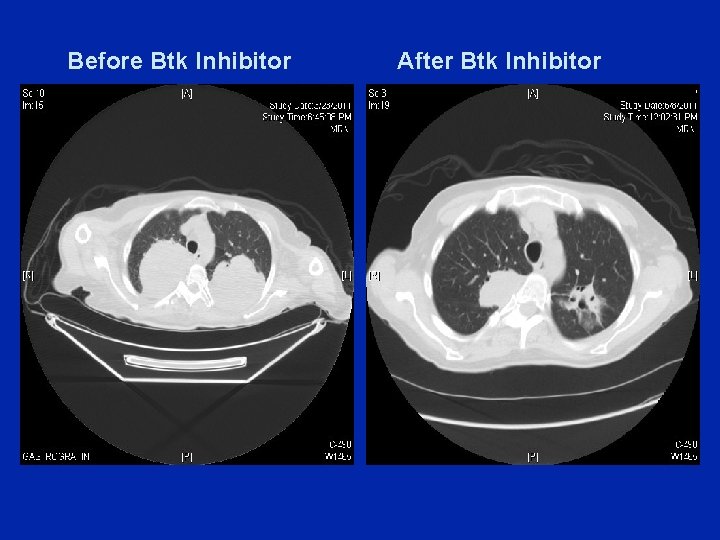
Before Btk Inhibitor After Btk Inhibitor

The Btk Inhibitor, PCI-32765, Induces Durable Responses with Minimal Toxicity in Patients with Relapsed/Refractory B-Cell Malignancies: Results from a Phase I Study Fowler N et al. Proc ASH 2010; Abstract 964.
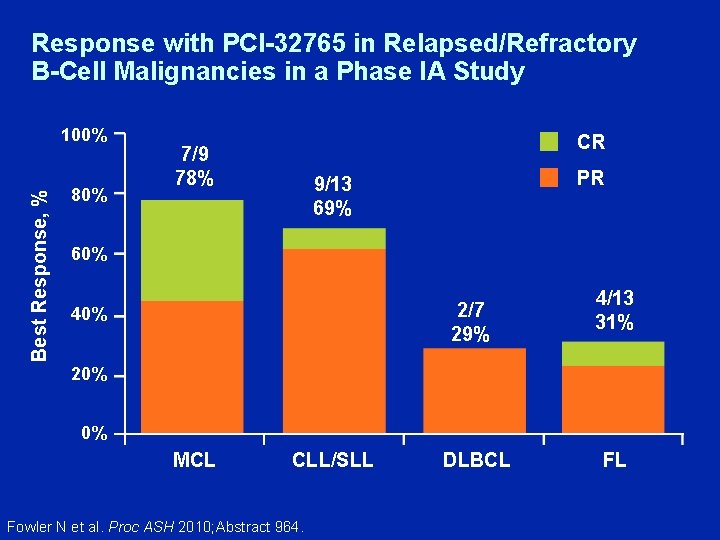
Response with PCI-32765 in Relapsed/Refractory B-Cell Malignancies in a Phase IA Study Best Response, % 100% 80% CR 7/9 78% PR 9/13 69% 60% 2/7 29% 40% 4/13 31% 20% 0% MCL CLL/SLL Fowler N et al. Proc ASH 2010; Abstract 964. DLBCL FL

Dr Gregory (Case F): DLBCL • 1999: 20 yo male college student with DLBCL – No B symptoms, no PMH – Positive for CD 20, CD 10 and CD 45 – IPI Score: 1 • 5/2000: Completes CHOP x 8 • Restaging CT after 4 cycles: 50% in mass size, negative Gallium scan • 6/2000: Restaging PET scan high SUV in mediastinum • Biopsy: DLBCL • 8/2000: ESHAP salvage therapy, BEAM, auto. SCT • Post-transplant consolidative RT to mediastinum • Repeat CT scans: Negative for 5 years • Patient lost to follow-up

Dr Gregory (Case F): DLBCL • 8/2009 (9 years later): Negative CTs • 1/2010: Bilateral inguinal adenopathy found on PE, largest 3. 7 cm left inguinal area • CT CAP: 2. 9 x 3. 7 cm, left inguinal lymph node, enlarged left para-aortic nodes and mass around rectal area • PET/CT: Consistent with abdominal lesion involving retroperitoneal lymph nodes, abdominal lymph nodes; celiac and pelvic lesion consistent with neoplasm, presumably lymphoma – Enlarged metabolically active lesion rectal region • MUGA: LVEF — 52%
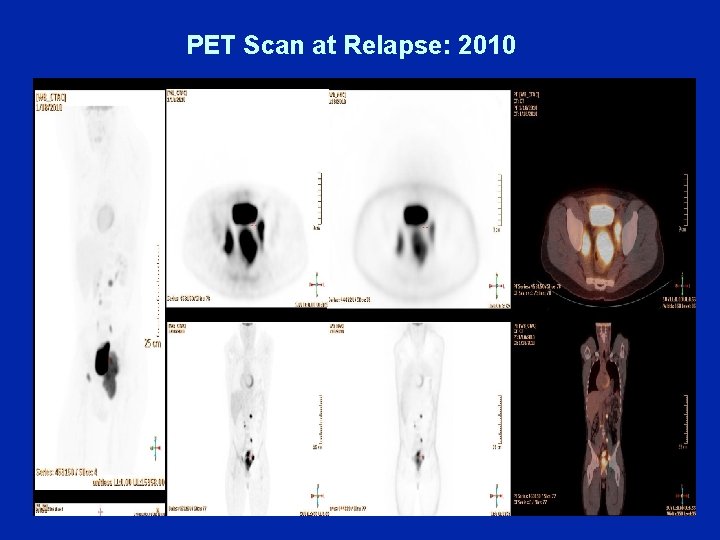
PET Scan at Relapse: 2010

Dr Gregory (Case F): DLBCL • Colonoscopy: Rectal erythema, edematous folds, friability and an aphthous ulcer; biopsy negative for DLBCL • BM bx: Negative • LN bx: DLBCL • Flow cytometry: Bright CD 45, CD 20, CD 19, CD 10 and monoclonal kappa • Labs: Normal, LDH 280 • Patient received 2 cycles of R-ICE and repeat auto transplant • Presently in complete remission one year post-transplant

Dr Gregory (Case F): DLBCL • IPI = 1 – A = Age of 30 – P = Performance Status - 1 – L = LDH: 280 – E = Extra Nodal -1 – S = Stage - IIE? (Was mass arising from rectum? Or was this a nodal mass pushing in on rectum? )

Maintenance with Rituximab After Autologous Stem Cell Transplantation in Relapsed Patients with CD 20 Diffuse Large B-cell Lymphoma (DLBCL): CORAL Final Analysis Gisselbrecht C et al. Proc ASCO 2011; Abstract 8004.

CORAL: EFS by Post-ASCT Maintenance Rituximab versus Observation Event-free survival Rituximab (n = 122) Observation (n = 120) p-value 45% 47% 0. 7435 PFS and OS also did not differ between post-ASCT maintenance rituximab and observation. Gisselbrecht C et al. Proc ASCO 2011; Abstract 8004.

4. Do you order specific screening tests for hepatitis for patients who are about to initiate treatment with rituximab and who have no history of or risk factors for hepatitis?

Hepatitis B Screening in Patients Receiving Anti-CD 20 Antibody (Rituximab) • No risk factors: – Hepatitis B surface antigen and core antibody • Risk factors or prior history of hepatitis B: – Add e-antigen • If positive, check viral load and consult with gastroenterologist NCCN Clinical Practice Guidelines in Oncology, Non-Hodgkin Lymphomas, v 3. 2011.

ECOG-E 3404: Response-Adapted Therapy for Aggressive NHL Based on Early PET Scanning Target Accrual: 99 + Eligibility Criteria Bulky Stage II or Stage III/IV DLBCL R-CHOP 4 Cycles PET SCAN - www. clinicaltrials. gov, July 2011. R-ICE 4 cycles R-CHOP 2 cycles

Interim Positron Emission Tomography Scans in Diffuse Large B-cell Lymphoma: An Independent Expert Nuclear Medicine Evaluation of the Eastern Cooperative Oncology Group E 3404 Study Horning SJ et al. Blood 2010; 115(4): 775 -7.

Interim PET Scans • PET scans read by three independent reviewers • Moderate agreement among readers (68% by ECOG criterion) • Proportion of positive PET scans relatively low (range 16% to 34%) • PET interpretation should be standardized Horning SJ et al. Blood 2010; 115(4): 775 -7.

RAPID-FIRE QUESTIONS

Diffuse Large B-Cell Lymphoma • What is the role of maintenance therapy in follicular lymphoma after transformation? • Use of bendamustine/rituximab in DLBCL • Would you ever consider a transplant at first remission? • Is there any clinical benefit to using dose-dense R-CHOP?

Schedule of Events Tuesday, August 2 Chronic Myeloid Leukemia Susan M O’Brien, MD Neil P Shah, MD, Ph. D
- Slides: 78