RTEC 111 Bushong Ch 2 3 15 Technique

RTEC 111 Bushong Ch 2, 3 &15 Technique Math

Fundamentals of Radiologic Science RTEC 111 Bushong Ch. 2


Units of Measurement n n n This allows scientists to describe quantities. The fundamental units of measurement are mass, length and time. Two widely used systems of measurement

UNITS OF RADIATION MEASUREMENT • TO QUANTIFY THE AMOUNT OF RADIATION A PATIENT OR WORKER RECEIVES.

Conventional (British)Units vs. SI Units n Conventional (British) Units Used Since The 1920’s (foot, pound, second) ¡ n also called the US customary system 1948 - A System Of Units Based On Metric Measurements Was Developed By The International Committee For Weights And Measures. SI Units

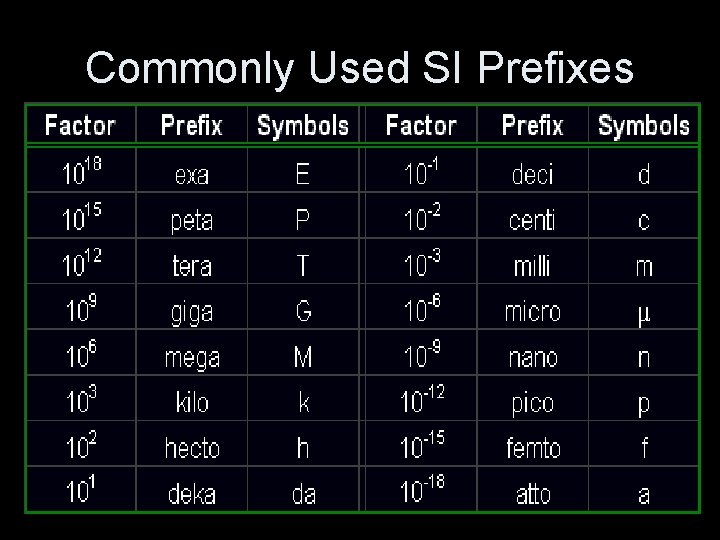
Commonly Used SI Prefixes

Important Radiology Units n Roentgen (R) is coulomb per kilogram (C/kg) n Radiation absorbed dose (rad) is gray (Gy) n Radiation equivalent man (rem) is seivert (Sv)
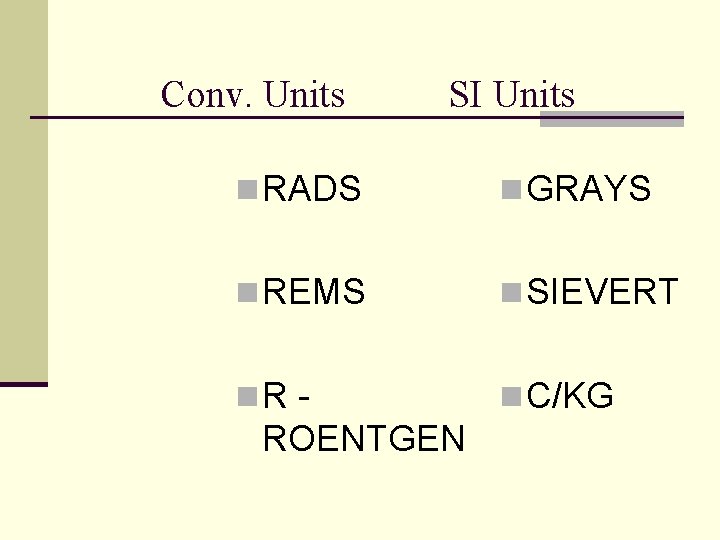
Conv. Units SI Units n RADS n GRAYS n REMS n SIEVERT n. R - n C/KG ROENTGEN

ROENTGEN (R) p p p SI = coulomb per kilogram (C/kg) or air kerma Gya The quantity of X-ray radiation Only exposure in air Output of the x-ray tube Does not indicate actual patient exposure or absorption

RADIATION ABSORBED DOSE (RAD) SI = GRAY (Gy) n Measures the amount of energy absorbed in any medium (the patient) n 1 Gy = 100 rads n 1/100 or 0. 01 Gy = 1 rad n 1 centigray = 1 rad

REM / SIEVERT n 1 Sv = 100 rem n 1/100 Sv = 1 rem n 1 centisievert = 1 rem n Used for occupational exposure EMPLOYEE EXPOSURE

RADIATION EQUIVALENT MAN (rem) OR Effective dose SI UNITS = SIEVERT Not all types of radiation produce the same responses in living tissue n n n The unit of dose equivlaence, as expressed as the product of absorbed dose in RAD and the quality factor

Rem OR Sievert n SI UNITS = SIEVERT n 1 Sv = 100 rem n THE PRODUCT OF THE GRAY AND THE QUALITY FACTOR.

Rad VS. Rem n RAD’S X QUALITY FACTOR = REM n GRAY’S X QUALITY FACTOR = SIEVERT n QUALITY FACTOR FOR X-RAYS = 1 n So…… Rads = Rems

Rems & Rads n Sieverts & Grays n PAtient = r. Ads & gr. Ays n Employee (technologists) = r. Ems & si. Ev. Erts

U. S. to S. I. Conversion a trick to remember p U. S. = rads & rems p S. I. = grays & sieverts p Pennies p Dollars

Metric System n milli n m n 10 -3 n 5 mrem = 0. 005 rem n n 5000 mrem = 5 rems 500 mrem = 0. 5 rems 50 mrem = 0. 05 rems 5 mrem = 0. 005 rems

Radiology – units of measurement n n What units correlate with: Exposure Dose Effective dose

The Structure of Matter RTEC 111

Radiology Mechanics n Velocity ¡ The motion of an object can be described by the use of two terms velocity and acceleration. ¡ Velocity = speed ¡ What is the speed of x-rays in a vacuum?

X-Ray Properties n Travel in straight lines. n Travel at the speed of light, 3 X 108 meters per second in a vacuum or 299, 792, 458 m/s or 29, 979, 245, 800 cm/s n Can ionize matter.
Kinetic energy (KE) n n The energy associated with the motion of an object Kinetic energy depends on the mass of the object and the square of its velocity
Potential energy (PE) n The stored energy of position or configuration
Heat n Is the kinetic energy of the random motion of molecules

Atoms n n Elements: 112 substances have been identified ¡ 92 are naturally occurring and 20 more have been artificially produced The atom is the smallest particle of matter that has the properties of an element

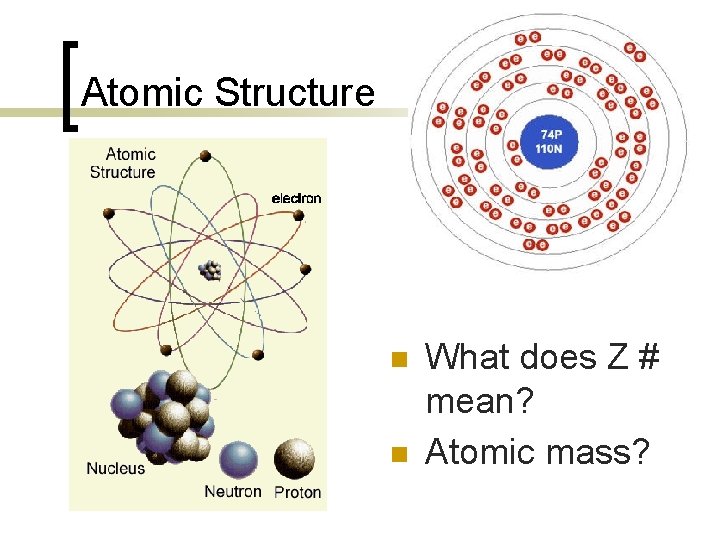
Atomic Structure n n What does Z # mean? Atomic mass?

Combining atoms n Atom + Atom = molecule n Molecule + Molecule = Compound n The smallest particle of an element is an atom; the smallest particle of a compound is a molecule

Elements n Chemical elements – determined by the # of protons n Isotopes – neutrons, atomic mass n Shells – electron orbits ¡ Ion or Ionization?
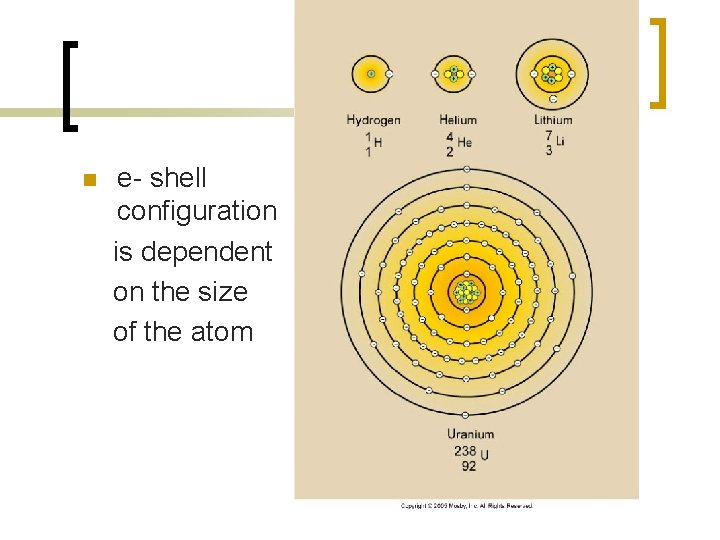
n e- shell configuration is dependent on the size of the atom
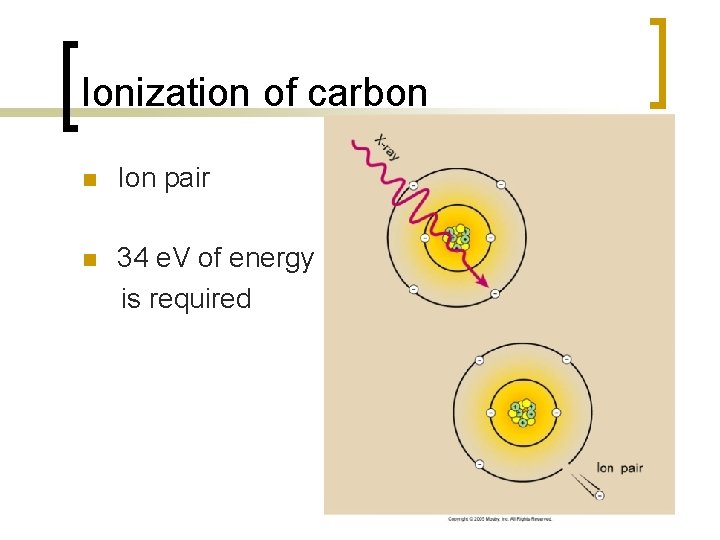
Ionization of carbon n Ion pair n 34 e. V of energy is required

Electron Arrangement n The maximum number of e- that can exist in each shell increase with distance from the nucleus n See table n No outer shell can contain more than eight e-

Periodic table of the elements n n n The table is organized by the number of e- in the outer or valence shell of an atom # of e- in the outermost shell = the period of that atom on the table The valence shell is important because it determines how that element will react and interact with other elements

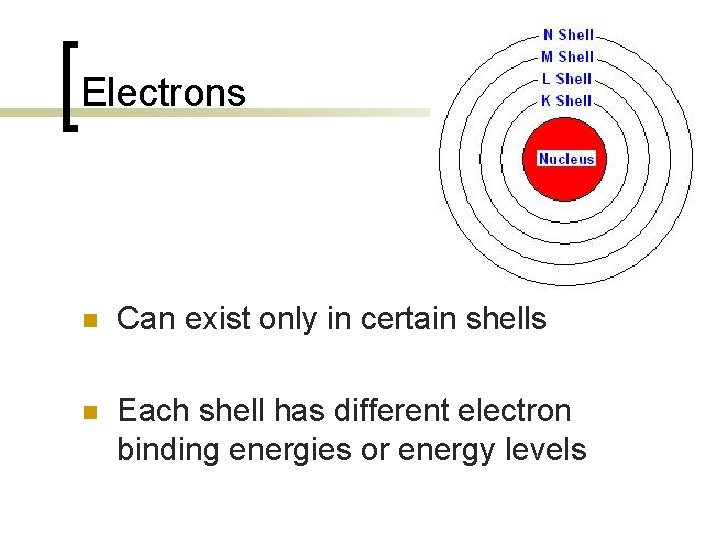
Electrons n Can exist only in certain shells n Each shell has different electron binding energies or energy levels

Electron binding energy n n The closer the e- is to the nucleus the more tightly it is bound and the higher the binding energy Also the larger the Z# of the atom the higher the binding energy for any given shell…. therefore more difficult of ionize larger atoms

Ionization potential n The energy required to ionize tissue atoms n How much energy is required to ionize tungsten’s K shell? n Pg 46

Types of Ionizing Radiation n All ionizing radiation can be classified into two categories ¡ n Particulate or electromagnetic radiation What type of radiation is used for diagnostic ultrasound or magnetic resonance imaging? Ionizing or Nonionizing?

Particulate Radiation n The emission of particles and energy from the nucleus in order to become stable n Radioactive elements are called radionuclides or radioisotopes
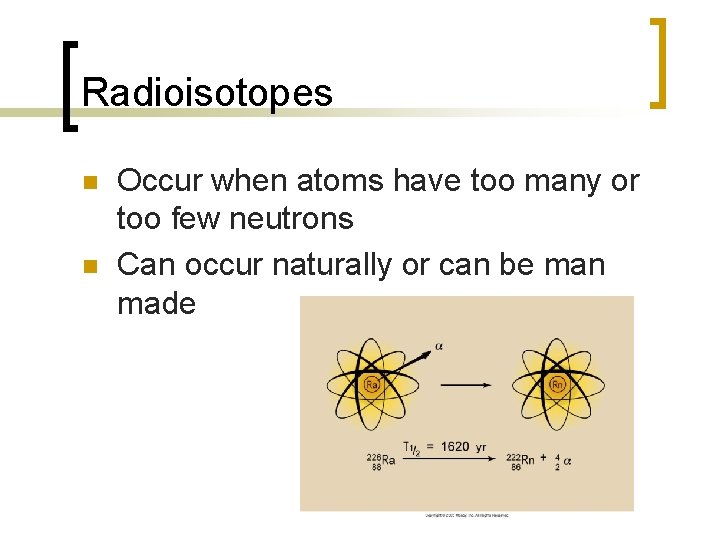
Radioisotopes n n Occur when atoms have too many or too few neutrons Can occur naturally or can be man made

Radioisotope decay n Decay from the nucleus to become stable ¡ n n Beta emission and Alpha emission Alpha & Beta particles can cause ionization because of high kinetic energy What form of energy does x-rays use to ionize

Radioactive Half-Life n The time required for a quantity of radioactivity to be reduced to one-half its original value n For radiology: Half-value layer ¡ To reduce the strength of the x-ray beam by 1/2

Photons vs Particles n n Particles cause ionization through kinetic energy Photons have no mass, no charge, travel at the speed of light and are considered energy disturbances in space. A form of EM energy. Photons travel at the speed of light or not at all.

X-rays vs Gamma rays n n n Forms of EM energy Only difference between x-rays and gamma rays is their origin Only difference between alpha and beta particles is their origin

Origins n n n X-rays = outside the nucleus in the eshells Alpha & Beta particles = from the nucleus Gamma rays = from the nucleus ¡ As part of radioactive decay
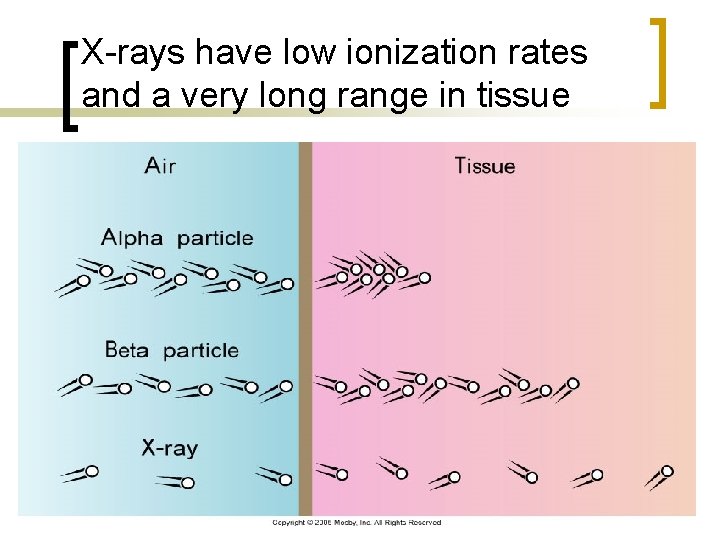
X-rays have low ionization rates and a very long range in tissue

Technique Calculations

What happens to primary? n When x-rays pass through a patient's body, three things can happen: (1) the x-ray photon is transmitted, passing through the body, interacting with the film, and producing a dark area on the film; (2) the x-ray photon is absorbed in an area of greater tissue density, producing lighter areas on the film; and (3) the x-ray photon is scattered and reaches the film causing an overall gray fog.

Density n The degree of overall blackening from the black metallic silver deposited in the emulsion. n Optical Density: range of human visibility


Densitometer
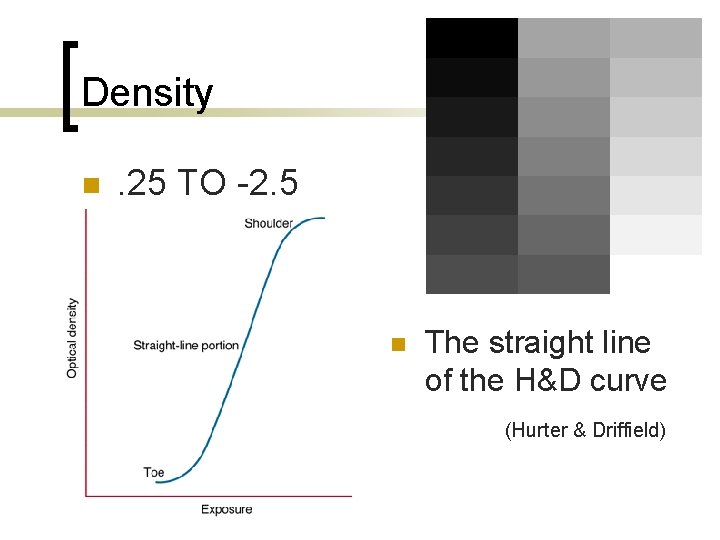
Density n . 25 TO -2. 5 n The straight line of the H&D curve (Hurter & Driffield)

Optical Density n n n Controlling factor: m. As determines the quantity of xrays What is the formula to determine quantity of x-rays?


m. As n n m. A X time (sec) = m. As The quantity of x-rays produced

To see changes in optical density n In order to see changes in optical density on a radiograph you must increase you m. As by at least 20 – 30% n To double the density on a radiograph you must double your m. As

k. Vp n Kilovolts peak n k. Vp Controls the Contrast n (for analog/film imaging)

Low vs High contrast n As a radiologic technologist, you will learn to distinguish between low- and high-contrast radiographs. A radiograph with large differences in density is a high-contrast, or shortscale, radiograph. If the density differences aren't as great, you have a low-contrast, or long-scale, radiograph.








k. Vp: Effects density & contrast n n n a. 15% rule: 15% k. Vp = doubling of exposure to the film 15% k. Vp = halving of exposure to the film b. 15% rule will always change the contrast of the image because k. V is the primary method of changing image contrast.

Its math time……
- Slides: 69