Rovibronic bands of the A X electronic transition

~ ~ Rovibronic bands of the A – X electronic transition of CH 3 O 2 and CD 3 O 2 detected with cavity ringdown absorption in the NIR Patrick Rupper, Erin N. Sharp and Terry A. Miller Laser Spectroscopy Facility, Department of Chemistry, The Ohio State University, Columbus, OH 43210

Motivations: Combustion, Atmospheric Chemistry, and Fundamental Interest Ø Ø Alkyl peroxy radicals (RO 2 • ) are key reaction intermediates in low temperature combustion of hydrocarbons (fossil fuels) as well as to many chemical reactions that occur in the atmosphere Atmospheric chemistry: final products of RO 2 • chemistry are ROOH (acid rain), PAN (peroxy acetyl nitrate), NO 2 (ozone cycle)1 Methyl peroxy (CH 3 O 2) is the simplest alkyl peroxy radical → starting point for subsequent investigation of larger alkyl peroxy radicals Effect of the internal methyl rotation on the spectrum 1) Lightfoot et al. , Atmos. Envir. 26 A, 1805 (1992)
Spectroscopic Detection and Characterization CH 3 O 2 J. A. Jafri and D. H. Phillips, J. Am. Chem. Soc. 112, 2586 (1990) Very recently: ionization detection techniques → parent cation only stable for CH 3 O 2 - H. B. Fu, Y. J. Hu, and E. R. Bernstein, JCP 125, 014310 (2006) - G. Meloni, P. Zou, S. J. Klippenstein, M. Ahmed, S. R. Leone, C. A. Taatjes, and L. Osborn, 128, 13559 (2006) Radicals, 1 O. J. D. Nielsen and T. JACS J. Wallington, in Peroxyl (John Wiley and Sons, New York, 1997), pp. 72 -73 P. D. et al. , Atmos. 1805 JCP (1992) 3 Miller group and Y. Lightfoot P. Lee group (JCP 112, Envir. 1069526 A, (2000), (2007)) 2 4 D. B. Atkinson, J. L. Spillman, JPCA 106, 8891 (2002) ~ ~ B-X Transition very strong, s ~ 10 -17 cm 2 routinely used in kinetics studies 1, 2 ~ B state - repulsive lack of selectivity ~ ~ A-X Transition weak, s ~ 10 -20 cm 2 less explored 3, 4 ~ A state - bound very selective { requires a tunable light source in the NIR and a sensitive technique
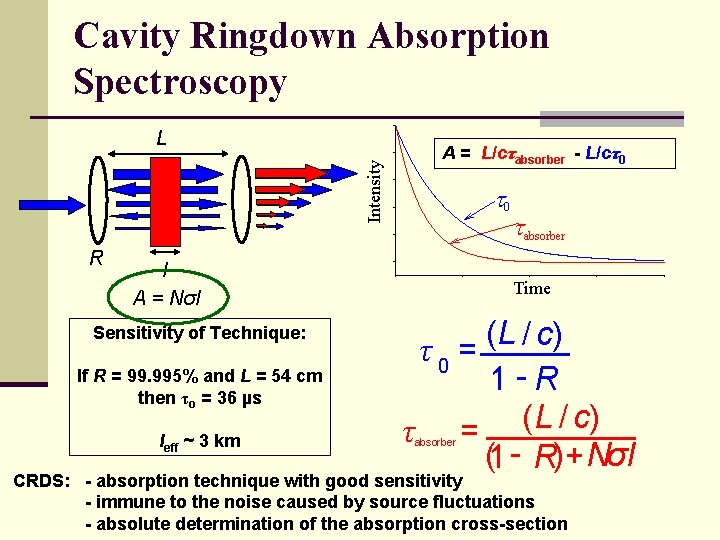
Cavity Ringdown Absorption Spectroscopy Intensity L R A = L/cτabsorber - L/cτ0 t 0 l A = Nσl Sensitivity of Technique: If R = 99. 995% and L = 54 cm then τo = 36 µs leff ~ 3 km tabsorber Time ( L / c) τ0 = 1 -R τ = ( L / c) (1 - R)+ Nσ l absorber CRDS: - absorption technique with good sensitivity - immune to the noise caused by source fluctuations - absolute determination of the absorption cross-section
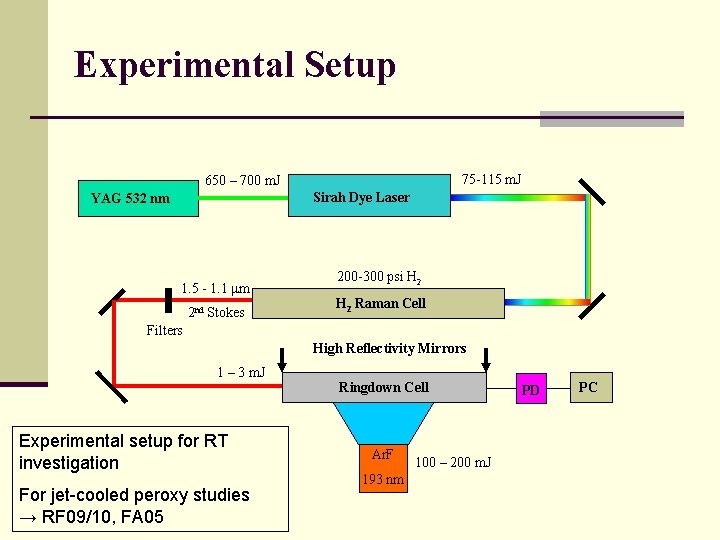
Experimental Setup 75 -115 m. J 650 – 700 m. J Sirah Dye Laser YAG 532 nm 1. 5 - 1. 1 mm 2 nd Stokes 200 -300 psi H 2 Raman Cell Filters High Reflectivity Mirrors 1 – 3 m. J Experimental setup for RT investigation For jet-cooled peroxy studies → RF 09/10, FA 05 Ringdown Cell Ar. F 193 nm 100 – 200 m. J PD PC

Peroxy Radical Production Reaction Initiation Acetone + hν 2 CH 3 + CO Production (within 1 - 5 µs completed) O 2 + CH 3 + M CH 3 O 2 + M Radical-Radical Losses (less than 10% during first 100 µs) R + M R 2 + M R + RO 2 2 RO RO 2 + RO 2 Products Typical experimental conditions: 60 Torr O 2, 90 Torr N 2 and 2 -3 Torr Acetone 100 µs time difference between photolysis and probing photolysis on photolysis off
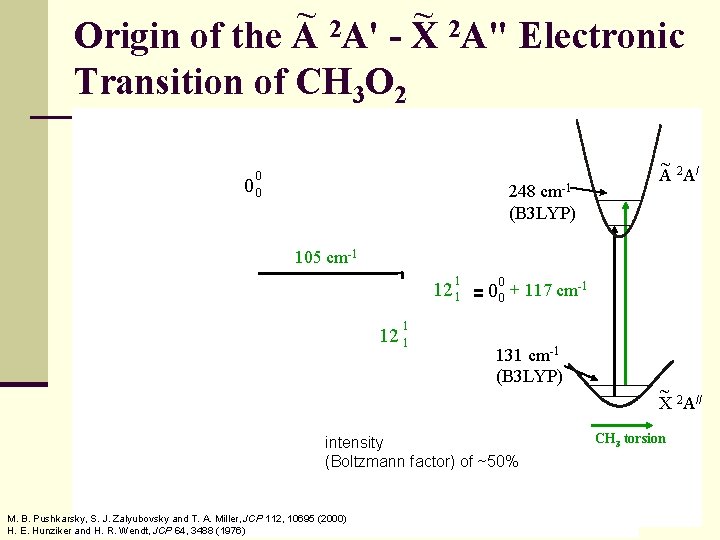
~ ~ 2 A' Origin of the A - X 2 A" Electronic Transition of CH 3 O 2 0 00 248 cm-1 (B 3 LYP) ~ A 2 A/ 105 cm-1 1 12 1 = 0 00 + 117 cm-1 1 12 1 131 cm-1 (B 3 LYP) intensity (Boltzmann factor) of ~50% M. B. Pushkarsky, S. J. Zalyubovsky and T. A. Miller, JCP 112, 10695 (2000) H. E. Hunziker and H. R. Wendt, JCP 64, 3488 (1976) ~ 2 // X A CH 3 torsion
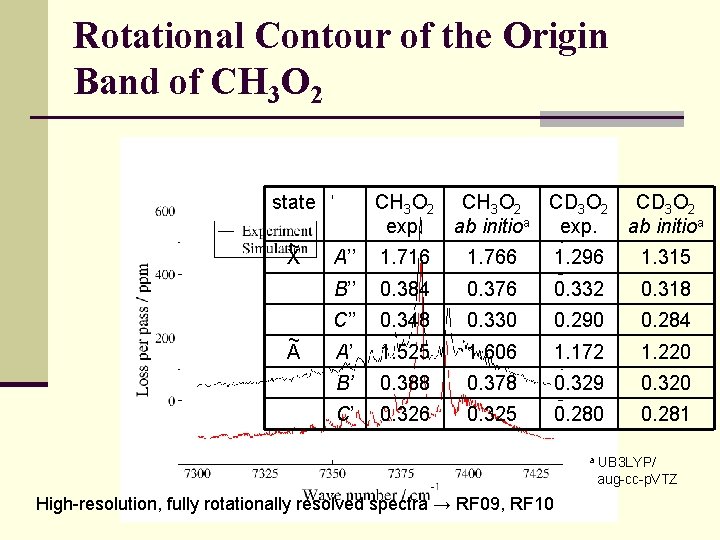
Rotational Contour of the Origin Band of CH 3 O 2 state ~ X ~ A CH 3 O 2 CD 3 O 2 exp. ab initioa A’’ 1. 716 1. 766 1. 296 1. 315 B’’ 0. 384 0. 376 0. 332 0. 318 C’’ 0. 348 0. 330 0. 290 0. 284 A’ 1. 525 1. 606 1. 172 1. 220 B’ 0. 388 0. 378 0. 329 0. 320 C’ 0. 326 0. 325 0. 280 0. 281 a High-resolution, fully rotationally resolved spectra → RF 09, RF 10 UB 3 LYP/ aug-cc-p. VTZ

Complete NIR Spectrum of CH 3 O 2 Mode Vibration origin cm-1 8 / cm-1 7 / cm-1 CH 3 O 2 COO bend exp. O-O abstretch initioa Origin 00 0 COO bend methyl O-Otorsion sym str. 81 1 7383 378 887 1211 122 a 80 1 UB 3 LYP/ aug-cc-p. VTZ C. -Y. Chung, C. -W. Cheng and Y. -P. Lee, 61 st Symposium on Molecular Spectroscopy, 2006 70 1 370 917 7011211 2 8011211 7011222 CD 3 O 2 exp. CD 3 O 2 ab initioa 7373 in rotational - similarity contour 348 agreement 343 - good with ab 824 initio harmonic 841 vibrational frequencies - similar kinetic behavior other bands ? → involve large amplitude motion between methyl and peroxy group (“methyl torsion”) Talk of Gabriel Just (MH 07)
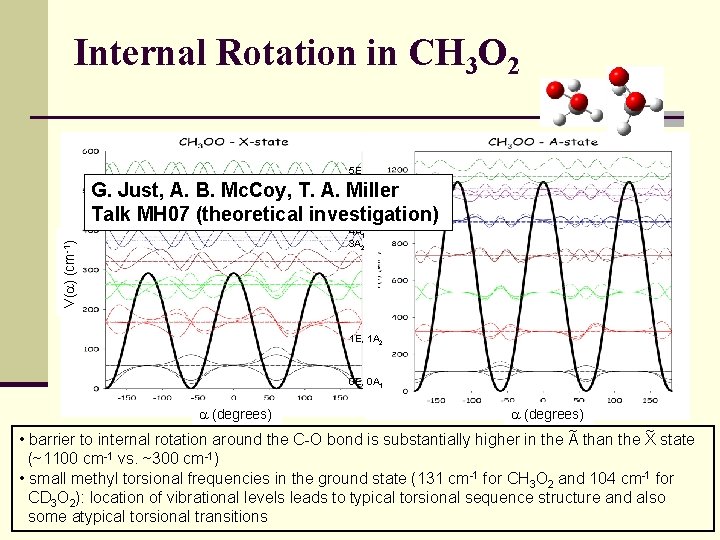
Internal Rotation in CH 3 O 2 5 E G. Just, A. B. Mc. Coy, T. A. Miller 4 E Talk MH 07 (theoretical investigation) V( ) (cm-1) 4 A 1 3 A 2 1 E, 1 A 2 0 E, 0 A 1 (degrees) ~ ~ • barrier to internal rotation around the C-O bond is substantially higher in the A than the X state (~1100 cm-1 vs. ~300 cm-1) • small methyl torsional frequencies in the ground state (131 cm-1 for CH 3 O 2 and 104 cm-1 for CD 3 O 2): location of vibrational levels leads to typical torsional sequence structure and also some atypical torsional transitions
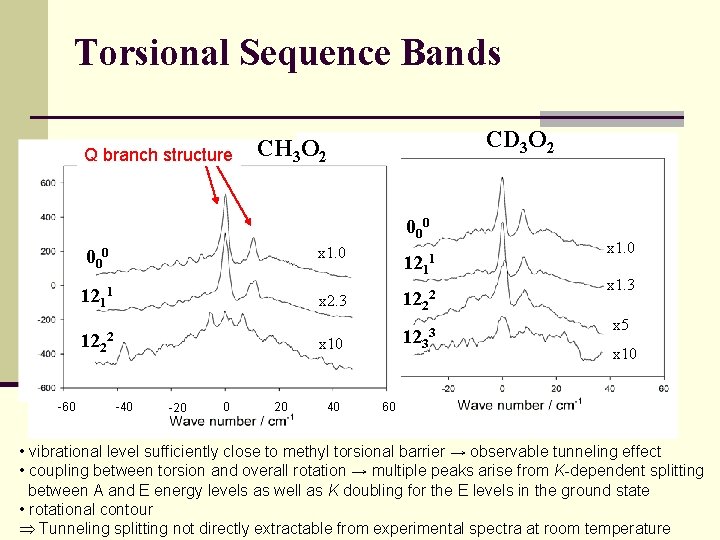
Torsional Sequence Bands Q branch structure CD 3 O 2 CH 3 O 2 000 1211 122 -60 2 -40 -20 0 20 x 1. 0 121 x 2. 3 122 x 10 1233 40 1 2 x 1. 0 x 1. 3 x 5 x 10 60 • vibrational level sufficiently close to methyl torsional barrier → observable tunneling effect • coupling between torsion and overall rotation → multiple peaks arise from K-dependent splitting between A and E energy levels as well as K doubling for the E levels in the ground state • rotational contour Tunneling splitting not directly extractable from experimental spectra at room temperature
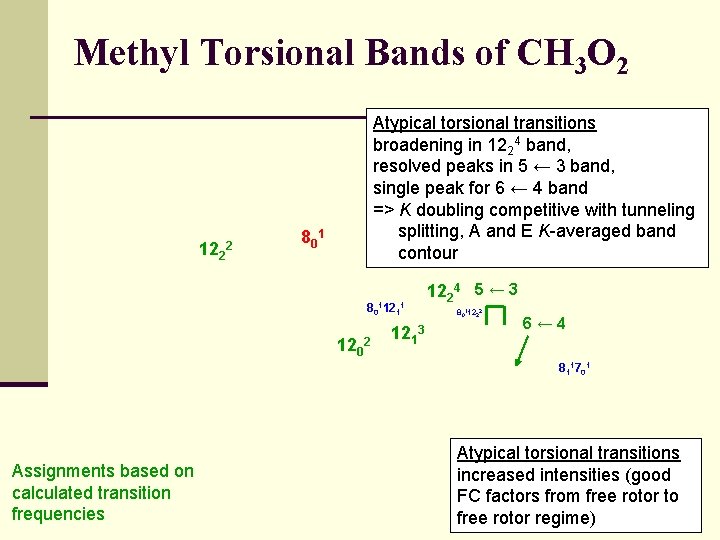
Methyl Torsional Bands of CH 3 O 2 122 2 Atypical torsional transitions broadening in 1224 band, resolved peaks in 5 ← 3 band, single peak for 6 ← 4 band => K doubling competitive with tunneling splitting, A and E K-averaged band contour 80 1 8011211 1202 1213 1224 5 ← 3 8011222 6← 4 811701 Assignments based on calculated transition frequencies Atypical torsional transitions increased intensities (good FC factors from free rotor to free rotor regime)

Conclusions n Studied normal and deuterated methyl peroxy radicals via CRDS under ambient conditions in the NIR n Observed several rovibronic bands → experimental vibrational frequencies n Methyl torsional mode plays significant role in the room-temperature spectrum → rich structure spectrum of typical sequence structure as well as atypical transitions n Tunneling splittings comparable to the rotational contour in higher sequence bands n Ongoing and future work includes: - larger alkyl peroxy radicals → see talks TB 03 – TB 05 - unsaturated alkyl peroxy radicals - high-resolution spectroscopy of methyl peroxy → see talks RF 09/10

Acknowledgments n n n Prof. Terry A. Miller Erin Sharp Gabriel Just and Prof. Anne Mc. Coy Prof. Yuan-Pern Lee and his research group Miller group members n DOE for funding


Overview: Alkyl Peroxies: ~~ A-X Electronic Transitions T-conformer 1 a) Pushkarsky et al. , J. Chem. Phys. 112, 10695 -10698 (2000). 1 b) Zalyubovsky et al. J. Phys. Chem. A 109, 1308 -1315 (2005). 1 c) Glover et al. J. Phys Chem. A 109, 11191 -11197 (2005). 1 d) P. Rupper, E. N. Sharp and T. A. Miller, JPCA 111, 832 (2007), in preparation (2 x)
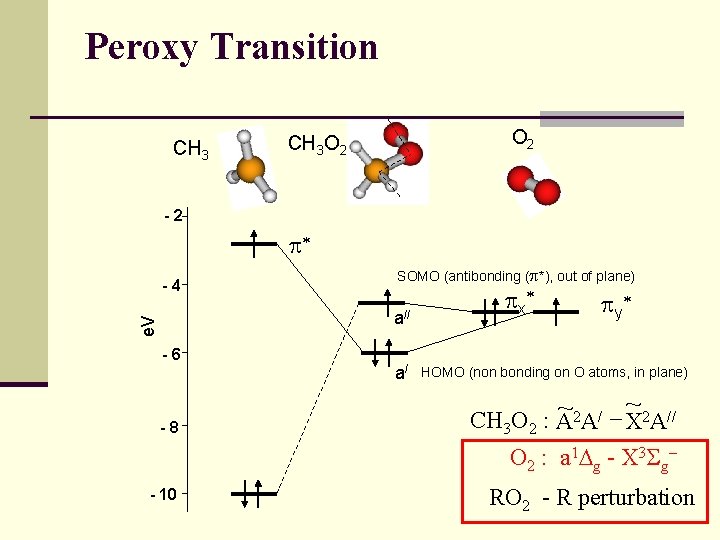
Peroxy Transition CH 3 O 2 CH 3 O 2 -2 p* -4 SOMO (antibonding (p*), out of plane) e. V a// -6 -8 a/ p x* p y* HOMO (non bonding on O atoms, in plane) ~ ~2 A/ X 2 A// CH 3 O 2 : A O 2 : a 1 Dg - X 3 Sg- - 10 RO 2 - R perturbation

CH 3 O 2 Simulation
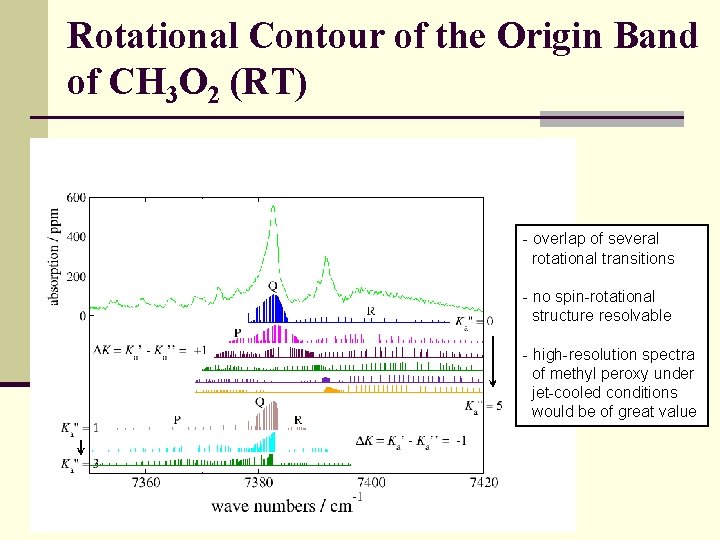
Rotational Contour of the Origin Band of CH 3 O 2 (RT) - overlap of several rotational transitions - no spin-rotational structure resolvable - high-resolution spectra of methyl peroxy under jet-cooled conditions would be of great value 7375
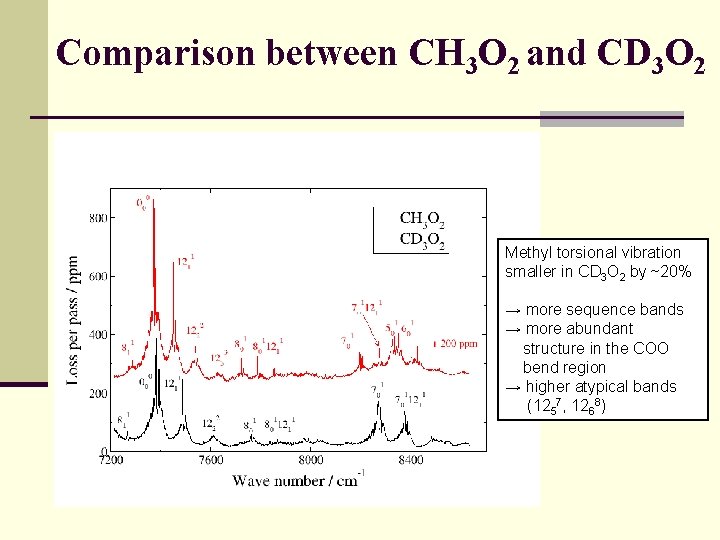
Comparison between CH 3 O 2 and CD 3 O 2 Methyl torsional vibration smaller in CD 3 O 2 by ~20% → more sequence bands → more abundant structure in the COO bend region → higher atypical bands (1257, 1268)
- Slides: 20