Rounding Summary Whole numbers Decimal Numbers Fill in
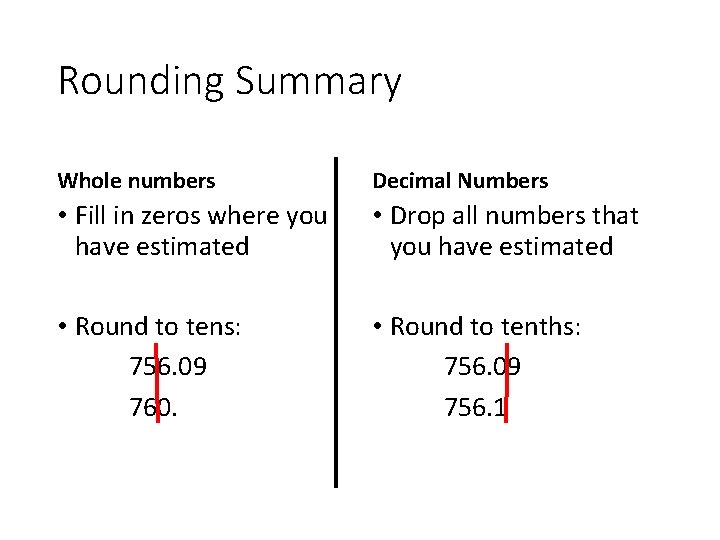
Rounding Summary Whole numbers Decimal Numbers • Fill in zeros where you have estimated • Drop all numbers that you have estimated • Round to tens: 756. 09 760. • Round to tenths: 756. 09 756. 1

Rounding Practice • Quiz- Trade • You each get a card. When you are not paired up your hand is up. • Move around and pair up. • You will answer each question • Trade Cards.

What’s in that beam? J. J. Thomson, 1897 • Cathode Ray Tube Experiments to measure the charge to mass ratio of the beam. • Particles in beam travel farther than hydrogen atoms. • The particles are 1000 times smaller than hydrogen atoms.

Thomson’s Model of the Atom 1897 Uniform base of positive charge with negative charges mixed within (electrons)

Ernest Rutherford, 1911 Gold Foil Experiment
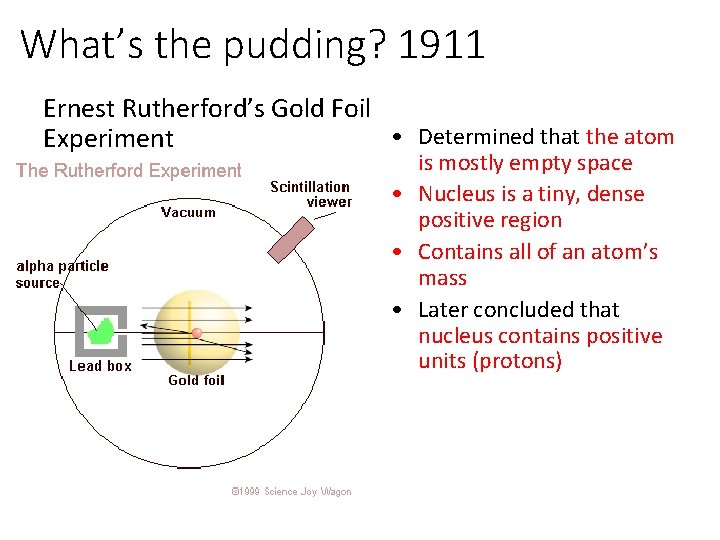
What’s the pudding? 1911 Ernest Rutherford’s Gold Foil • Determined that the atom Experiment is mostly empty space • Nucleus is a tiny, dense positive region • Contains all of an atom’s mass • Later concluded that nucleus contains positive units (protons)

James Chadwick, 1932 • After being released as a prisoner of WWI, Chadwick focused on radioactivity • Noticed discrepancies in the proton number and mass number • Used radioactive experiments to determine the neutral atom that has the mass of a proton (neutron)

Find Someone Who… • You will ask someone if they know the definition of the words in the left column • You will write their definition down • You will write their name • Once your done come back inside and glue on top half of pg 12

Atom Boards • Open up your boards • Let’s make a key • Red= Protons • Blue= Electrons • Clear= Neutrons

Build me an atom of Oxygen • How many proton’s did you put in? • How many electrons did you put in? • How many neutrons did you put in? • Atomic number= number of protons- determines element identity • Mass number= protons + neutrons

Isotopes-atoms of one element that have different mass number • Isotopes change the number of ____ • Build me an oxygen-18 atom • What does 18 represent? • Hyphen notation: element name – mass #
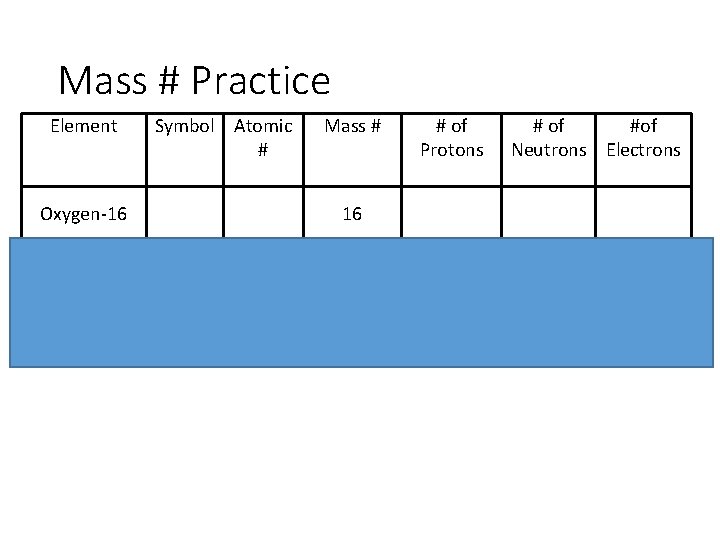
Mass # Practice Element Symbol Atomic # Oxygen-16 Mass # # of Protons # of #of Neutrons Electrons 16 Ru 58 26 56 23

Think it through • On the left page bottom half page

Ions- particles that carry a charge • Adding electrons creates an ANION- negatively charged atom • Removing electrons creates a CATION- positively charged atom
- Slides: 15