Rotational Spectrum And Ab Initio Calculations For Difluorosilyl

Rotational Spectrum And Ab Initio Calculations For Difluorosilyl Isocyanate Sean A. Peebles, Daniel A. Obenchain, Rebecca A. Peebles Dept. of Chemistry, Eastern Illinois University, Charleston, IL Michael H. Palmer School of Chemistry, University of Edinburgh, UK Gamil A. Guirgis, Zichao Wang, Jonel Lirjoni Dept. of Chemistry, College of Charleston, SC

Introduction Quasi-linear molecules characterized by: n Large amplitude motions n Low lying vibrationally excited states n Structures determined from different experimental methods (MW, IR, ED) can sometimes agree poorly n

Silyl isocyanates H 3 Si. NCO 1: Symmetric top in ground vibrational state; low barrier to linearity of heavy atom skeleton; nearly free internal rotation of the silyl group n F 3 Si. NCO: electron diffraction (ED) studies 2 give Si. NC angle of 160. 7(12)o n Cl 3 Si. NCO: IR/Raman results 3 suggested Si. NCO is linear; ED studies 4 give Si. NC angle 138. 0(4)o n Me 3 Si. NCO: Si. NC angle 150(3)o (ED)5 n Duckett, et al, J. Mol. Spectrosc. , 90, (1981), 374; 2 Airey, et al, J. Mol. Struct. , 8, (1971), 435; 3 Koster, Spectrochim. Acta 24 A, (1968), 395; 4 Hilderbrandt, Bauer, J. Mol. Struct. , 3, (1969), 325; 5 Kimura et al, J. Am. Chem. Soc. , 88, (1966), 416 1

Difluorosilyl isocyanate Microwave spectra n DFT and ab initio structures n Potential energy surfaces n Si. HF 2 internal rotation n Si. NC bending n

Ab initio calculations n Structures: Gaussian 09 and MOLPRO n n n Electric field gradients: GAMESS-UK Theoretical results at all levels favor a trans H-Si-N=C and trans Si-N=C=O n n B 3 LYP, MP 2, MP 4, CCSD(T) levels of theory with a 6 -311++G(3 df, 3 pd) basis But small energy differences between cis and trans H-Si-N=C orientations (at most 0. 45 k. J / mol) Microwave study gives rotational constants closer to those of a cis H-Si-N=C orientation (later)
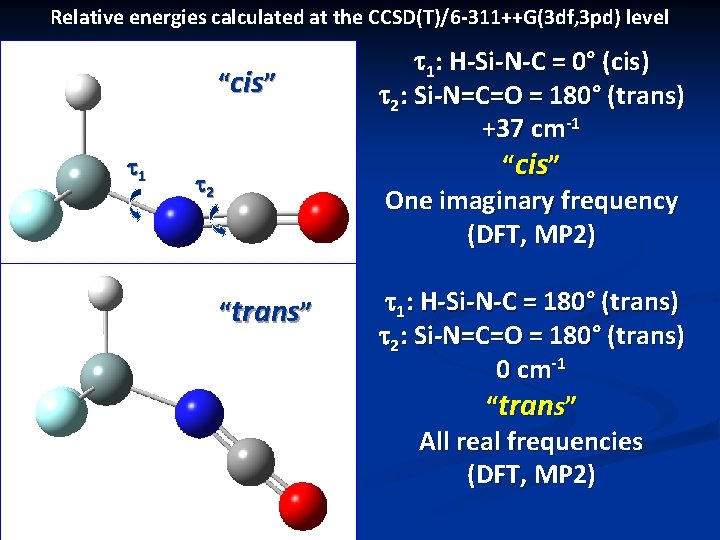
Relative energies calculated at the CCSD(T)/6 -311++G(3 df, 3 pd) level “cis” t 1 t 2 “trans” t 1: H-Si-N-C = 0° (cis) t 2: Si-N=C=O = 180° (trans) +37 cm-1 “cis” One imaginary frequency (DFT, MP 2) t 1: H-Si-N-C = 180° (trans) t 2: Si-N=C=O = 180° (trans) 0 cm-1 “trans” All real frequencies (DFT, MP 2)

Experimental n Synthesis at College of Charleston, SC n n Microwave measurements at Eastern Illinois University n n 1 Fluorination of dichlorosilyl isocyanate 1 using antimony trifluoride at room temperature 475 MHz bandwidth chirped-pulse Fourier-transform microwave (CP-FTMW) spectrometer (7 -18 GHz) Balle-Flygare Fourier-transform microwave (FTMW) spectrometer operating in the (4 -18 GHz) F. Häfler, G. Jägerhuber, W. Veigl, Monatshefte für Chemie, 105, (1974), 539.
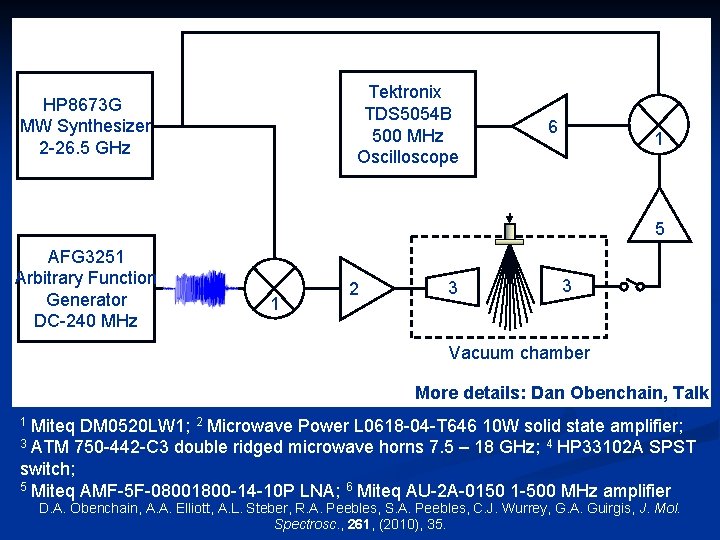
Tektronix TDS 5054 B 500 MHz Oscilloscope HP 8673 G MW Synthesizer 2 -26. 5 GHz 6 1 5 AFG 3251 Arbitrary Function Generator DC-240 MHz 1 2 3 3 Vacuum chamber More details: Dan Obenchain, Talk RH 07 1 Miteq DM 0520 LW 1; 2 Microwave Power L 0618 -04 -T 646 10 W solid state amplifier; 3 ATM 750 -442 -C 3 double ridged microwave horns 7. 5 – 18 GHz; 4 HP 33102 A SPST switch; 5 Miteq AMF-5 F-08001800 -14 -10 P LNA; 6 Miteq AU-2 A-0150 1 -500 MHz amplifier D. A. Obenchain, A. A. Elliott, A. L. Steber, R. A. Peebles, S. A. Peebles, C. J. Wurrey, G. A. Guirgis, J. Mol. Spectrosc. , 261, (2010), 35.

Experimental n 0. 8 mm General Valve Series 9 pulsed nozzle n n n Sample ~0. 5% in He/Ne at ~2 atm in both cases Initial scan 7 -18 GHz on CP-FTMW spectrometer n n n Run @ 6 Hz CP-FTMW and 10 Hz FTMW 475 MHz sections, pasted together Absolute frequencies determined by offsetting center frequency by 5 MHz Additional measurements on FTMW spectrometer n To confirm nuclear quadrupole hyperfine splittings and absolute frequencies

Rotational Spectrum n Spectrum was quite congested n n n Lots of unassigned transitions (sample was impure) Predominantly c-type spectrum, with weaker a-type transitions observed Nuclear quadrupole hyperfine splittings were clearly visible on CP-FTMW spectrometer a c
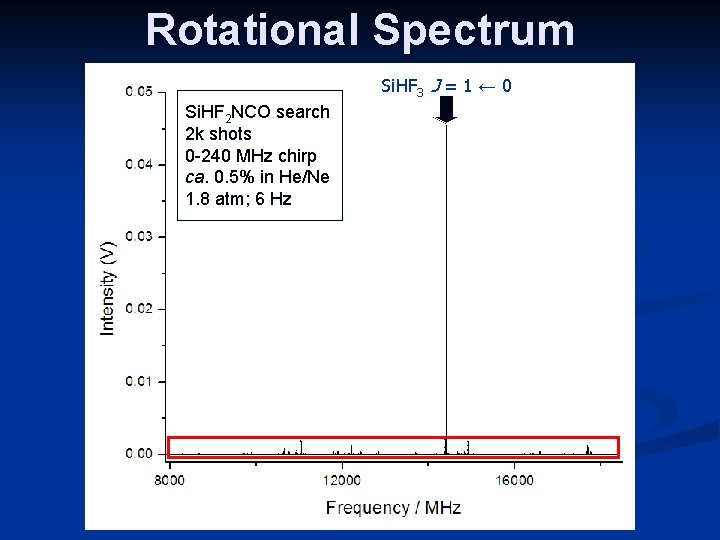
Rotational Spectrum Si. HF 3 J = 1 ← 0 Si. HF 2 NCO search 2 k shots 0 -240 MHz chirp ca. 0. 5% in He/Ne 1. 8 atm; 6 Hz
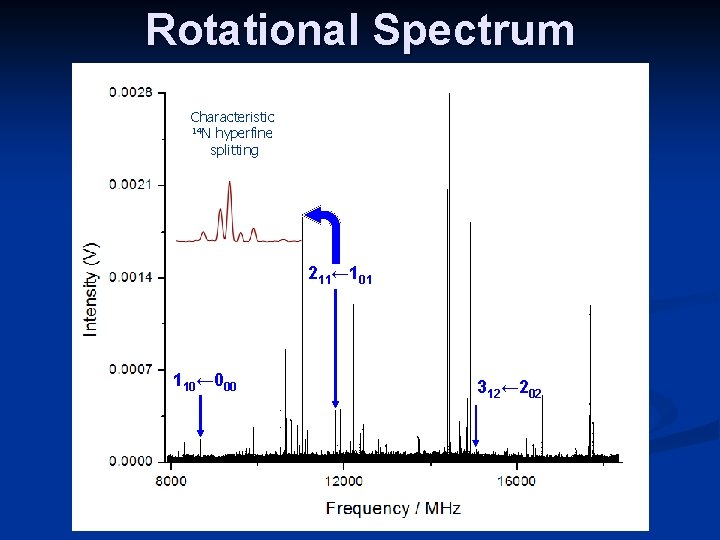
Rotational Spectrum Characteristic 14 N hyperfine splitting 211← 101 110← 000 312← 202
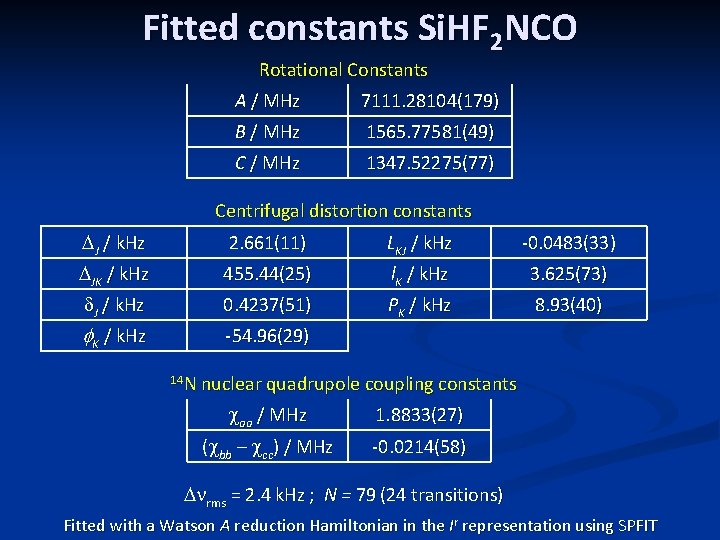
Fitted constants Si. HF 2 NCO Rotational Constants A / MHz 7111. 28104(179) B / MHz 1565. 77581(49) C / MHz 1347. 52275(77) Centrifugal distortion constants DJ / k. Hz 2. 661(11) LKJ / k. Hz -0. 0483(33) DJK / k. Hz 455. 44(25) l. K / k. Hz 3. 625(73) d. J / k. Hz 0. 4237(51) PK / k. Hz 8. 93(40) f. K / k. Hz -54. 96(29) 14 N nuclear quadrupole coupling constants caa / MHz 1. 8833(27) (cbb – ccc) / MHz -0. 0214(58) Dnrms = 2. 4 k. Hz ; N = 79 (24 transitions) Fitted with a Watson A reduction Hamiltonian in the Ir representation using SPFIT
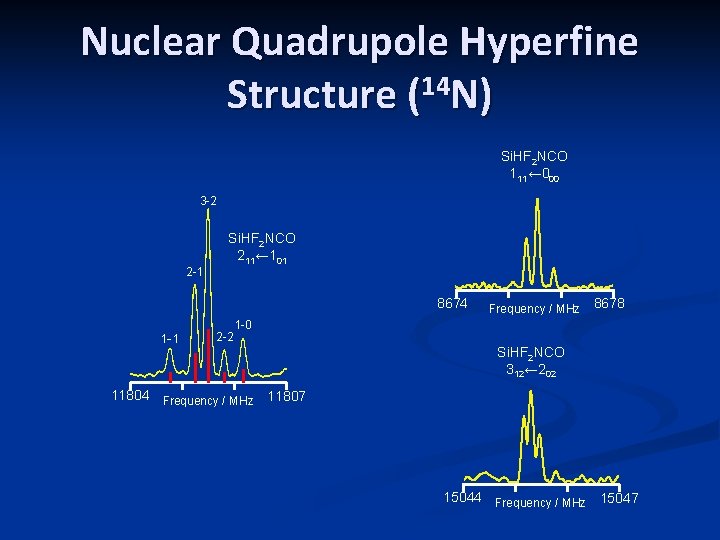
Nuclear Quadrupole Hyperfine Structure (14 N) Si. HF 2 NCO 111← 000 3 -2 2 -1 Si. HF 2 NCO 211← 101 8674 1 -1 2 -2 Frequency / MHz 8678 1 -0 Si. HF 2 NCO 312← 202 11804 Frequency / MHz 11807 15044 Frequency / MHz 15047
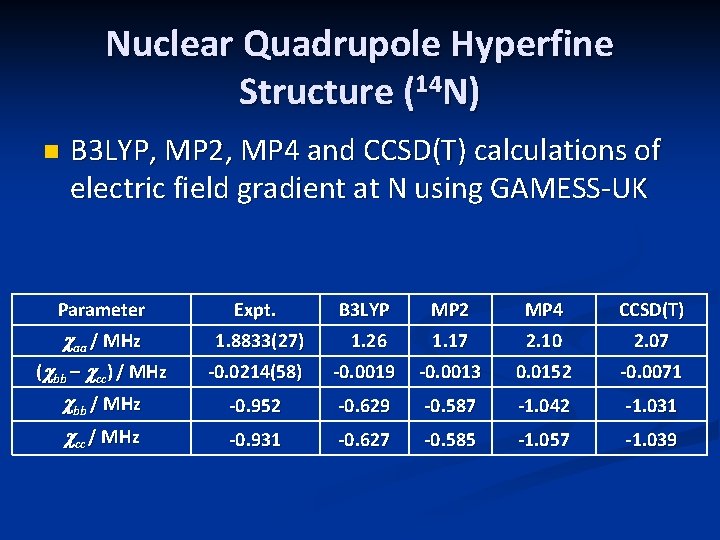
Nuclear Quadrupole Hyperfine Structure (14 N) n B 3 LYP, MP 2, MP 4 and CCSD(T) calculations of electric field gradient at N using GAMESS-UK Parameter Expt. B 3 LYP MP 2 MP 4 CCSD(T) caa / MHz (cbb – ccc) / MHz cbb / MHz 1. 8833(27) 1. 26 1. 17 2. 10 2. 07 -0. 0214(58) -0. 0019 -0. 0013 0. 0152 -0. 0071 -0. 952 -0. 629 -0. 587 -1. 042 -1. 031 ccc / MHz -0. 931 -0. 627 -0. 585 -1. 057 -1. 039

Ab initio structural parameters: “cis/trans” Method B 3 LYP MP 2 (valence) MP 4 (full) CCSD(T) Total Energy/a. u. -658. 21937 -657. 10557 -657. 10571 -657. 15835 -657. 15834 -657. 15192 -657. 15209 H–Si (Å) 1. 458 1. 456 1. 451 1. 450 1. 456 1. 451 1. 456 1. 453 Si–N (Å) 1. 681 1. 682 1. 686 1. 688 1. 679 1. 687 1. 686 N=C (Å) 1. 197 1. 196 1. 204 1. 207 1. 212 1. 208 1. 206 N=C=O (°) 177. 7 178. 8 178. 7 177. 3 177. 2 177. 9 177. 4 177. 2 F–Si–N (°) 108. 9 109. 5 108. 7 109. 5 108. 5 109. 4 108. 6 109. 6 F–Si–N=C (°) 121. 3 123. 3 121. 4 121. 1 121. 2 121. 6 121. 1 C=O (Å) F–Si (Å) H–Si–N (°) Si–N=C (°) Experimental rotational constants: 1. 162 1. 169 1. 175 1. 173 1. 165 1. 164 A = 7111. 28104(179) MHz 1. 579 1. 581 1. 584 1. 587 1. 584 1. 586 1. 583 1. 585 B 111. 8 = 1565. 77581(49) MHz 112. 4 110. 8 112. 7 111. 3 112. 6 110. 7 171. 2 167. 7 158. 5 154. 9 163. 0 154. 6 157. 0 C 171. 2 = 1347. 52275(77) MHz A (MHz) 6940. 8 6626. 7 6973. 3 6327. 1 7067. 3 6451. 2 7106. 5 6330. 9 B (MHz) 1513. 5 1546. 1 1506. 6 1590. 7 1508. 0 1568. 4 1517. 8 1600. 7 C (MHz) 1311. 3 1347. 0 1303. 5 1394. 2 1302. 5 1372. 5 1311. 0 1405. 5
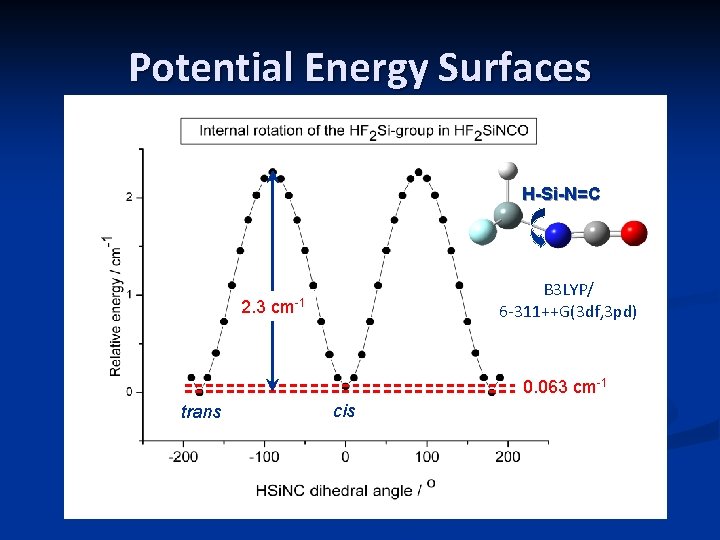
Potential Energy Surfaces H-Si-N=C 2. 3 B 3 LYP/ 6 -311++G(3 df, 3 pd) cm-1 0. 063 cm-1 trans cis

Potential Energy Surfaces Si-N=C Barrier to linearity is ~5 cm-1 B 3 LYP/6 -311++G(3 df, 3 pd)

Comparison of bending potential n n Several potential fits (E/cm-1, x/rad where x is 180°-Si. NC) n E = 139. 8(479)x 2 n E = 8. 73(270)x 4 n E = 47. 0(170)x 2 – 11. 4(99)x 4 n E = 1993(536)x 2 – 422(134)x 4 + 27(10)x 6 – 0. 5 x 8 n E = 288. 4(19)x 2 – 43. 7 exp (-2. 7 x 2) n E = 112(44)x 2 -195(38) / (0. 2 – 0. 08 x 2) x 2: x 4 ratio (4. 7: 1) much larger for Si. HF 2 NCO compared to other related molecules n 1. 4: 1 F 3 Si. NCO n 0. 06: 1 Cl 3 Si. NCO n 0. 01: 1 H 3 Si. NCO

Far IR Spectrum n n Recorded at the Canadian Light. Source, Far-IR beamline in May 2010 n Bruker IFS 125 HR, 9 compartments n 0. 00096 cm-1 resolution n 450 – 1200 cm-1 region n 72 m path length n 6 m. Torr Si. HF 2 NCO Very busy spectrum; still being analyzed
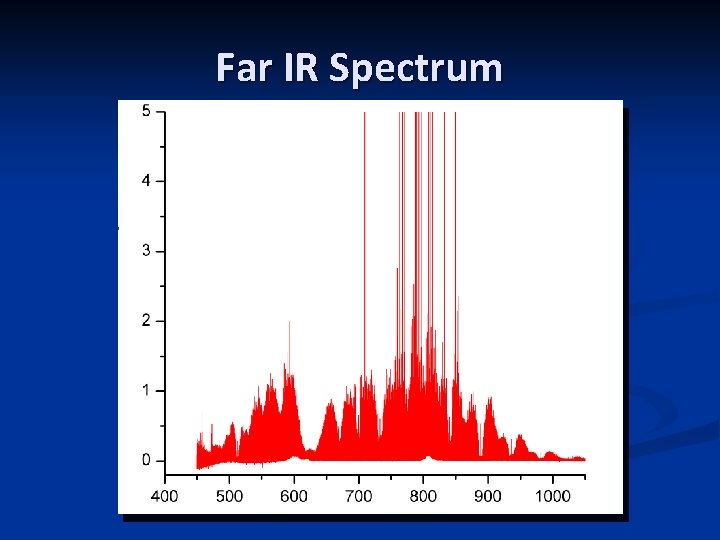
Far IR Spectrum
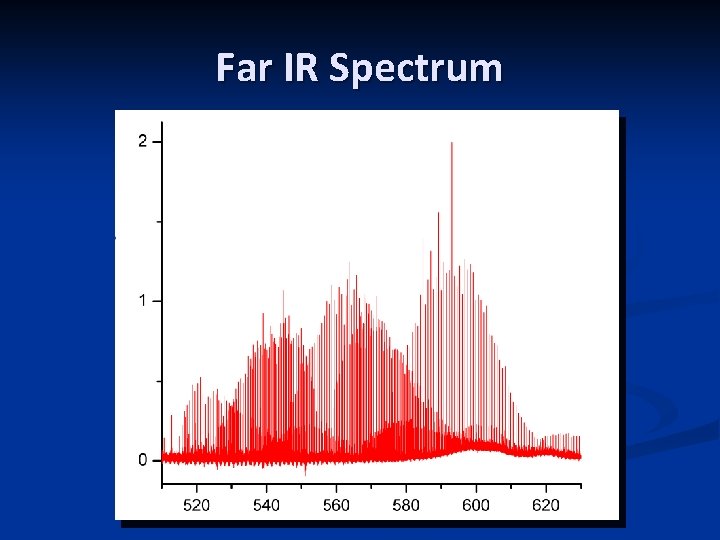
Far IR Spectrum

Conclusions n n n Two structures (cis and trans HSi. NC) lie very close in energy; barriers of 2. 3 cm-1 and ~5 cm-1 for Si. HF 2 rotation and Si. NC bending motions, respectively Distances for the cis and trans conformers typically agree to within 0. 006 Å at all levels of calculation Maximum differences between angles in cis and trans conformers are 0. 6 – 0. 8 degrees, except for the Si. NC angle

Conclusions n Single spectrum so far assigned in supersonic expansion n n A rotational constant seems more consistent with the “cis” conformer (with (H-Si-N-C) = 0° and (Si-N=C=O) = 180°) although theoretical B and C constants do tend to straddle experiment Plans to rescan with new sample if available to eliminate possibilities of additional states; need to return to analysis of Far-IR spectrum

Acknowledgments National Science Foundation, Research at Undergraduate Institutions CHE-0809387 n Prof. Brooks Pate, University of Virginia n n n Justin Neill Prof. Steve Cooke, University of North Texas n Smitty Grubbs

Fitted constants and correlation coefficients NEW PARAMETER (EST. ERROR) -- CHANGE THIS ITERATION 1 2 3 4 5 6 7 8 9 10 11 12 10000 A 20000 B 30000 C 200 Delta_J 1100 Delta_JK 40100 delta_j 42000 phi_KK 2200 L_KJ 43000 l_k 5000 P_K 110010000 chi_aa (14 110040000 0. 25*(chi_ MICROWAVE AVG = MICROWAVE RMS = 7111. 28104(179) 1565. 77581( 49) 1347. 52275( 77) -2. 6608(105)E-03 -0. 455439(254) -0. 4237( 51)E-03 -0. 054957(287) -0. 0483( 33)E-03 3. 625( 73)E-03 8. 93( 40)E-03 2. 8250( 41) -5. 34(145)E-03 0. 000101 MHz, IR AVG = 0. 002418 MHz, IR RMS = 0. 00000 -0. 0000 E-03 -0. 000000 0. 0000 E-03 -0. 0000 -0. 00 E-03 0. 00000 END OF ITERATION 4 OLD, NEW RMS ERROR= 0. 60451 1 2 -0. 413833 1 3 0. 653407 1 4 -0. 332442 1 5 -0. 891508 1 6 0. 256610 1 10 -0. 832296 1 11 -0. 006902 1 12 -0. 062738 2 1 -0. 413833 2 3 -0. 632047 2 7 -0. 740420 2 8 -0. 481448 2 9 0. 626159 2 10 0. 591041 2 11 -0. 013466 3 4 -0. 429297 3 5 -0. 766432 3 6 0. 559553 3 7 0. 804805 3 8 0. 669372 3 12 -0. 039300 4 1 -0. 332442 4 2 -0. 218163 4 3 -0. 429297 4 5 0. 313013 4 9 0. 397593 4 10 0. 384189 4 11 0. 000243 4 12 0. 047426 5 1 -0. 891508 5 6 -0. 319135 5 7 -0. 803903 5 8 -0. 936427 5 9 0. 850295 5 10 0. 879070 6 2 -0. 501717 6 3 0. 559553 6 4 -0. 175496 6 5 -0. 319135 6 7 0. 227794 6 11 0. 026423 6 12 -0. 006459 7 1 0. 727246 7 2 -0. 740420 7 3 0. 804805 7 8 0. 771868 7 9 -0. 940187 7 10 -0. 895444 7 11 -0. 016404 7 12 -0. 017502 8 4 -0. 330268 8 5 -0. 936427 8 6 0. 305493 8 7 0. 771868 8 9 -0. 877403 9 1 -0. 801903 9 2 0. 626159 9 3 -0. 773300 9 4 0. 397593 9 5 0. 850295 9 10 0. 984441 9 11 0. 021208 9 12 0. 019494 10 1 -0. 832296 10 2 0. 591041 10 6 -0. 282722 10 7 -0. 895444 10 8 -0. 940435 10 9 0. 984441 10 11 0. 031339 11 3 0. 006781 11 4 0. 000243 11 5 0. 002581 11 6 0. 026423 11 7 -0. 016404 11 12 -0. 022694 12 1 -0. 062738 12 2 -0. 025192 12 3 -0. 039300 12 4 0. 047426 12 8 -0. 018463 12 9 0. 019494 12 10 0. 017272 12 11 -0. 022694 1 2 2 3 4 5 5 6 7 8 8 9 10 10 11 12 7 4 12 9 6 2 11 8 4 1 10 6 3 12 8 5 0. 727246 -0. 218163 -0. 025192 -0. 773300 -0. 175496 0. 478854 0. 002581 0. 305493 -0. 171174 0. 869311 -0. 940435 -0. 262963 -0. 733369 0. 017272 -0. 036036 0. 031282 1 8 0. 869311 1 9 -0. 801903 2 5 0. 478854 2 6 -0. 501717 3 1 0. 653407 3 2 -0. 632047 3 10 -0. 733369 3 11 0. 006781 4 7 -0. 171174 4 8 -0. 330268 5 3 -0. 766432 5 4 0. 313013 5 12 0. 031282 6 1 0. 256610 6 9 -0. 262963 6 10 -0. 282722 7 5 -0. 803903 7 6 0. 227794 8 2 -0. 481448 8 3 0. 669372 8 11 -0. 036036 8 12 -0. 018463 9 7 -0. 940187 9 8 -0. 877403 10 4 0. 384189 10 5 0. 879070 11 1 -0. 006902 11 2 -0. 013466 11 9 0. 021208 11 10 0. 031339 12 6 -0. 006459 12 7 -0. 017502

Comparison of bending potential Si-N=C bending potential fitted to expression of the form: E = Ax 2 + Bx 3 + Cx 4 + Dx 5 + Ex 6 + Fx 7 + Gx 8 B 3 LYP 6 -311++G(3 df, 3 pd) Ax 2 term Cx 4 term Ratio x 2/x 4 terms H 3 Si. NCO 70. 6(42) 1200(10) 0. 059 F 3 Si. NCO * 32. 7(196) -24. 1(166) 1. 36 Cl 3 Si. NCO * 2. 16(65) -33. 80(1491) 0. 064 FCl 2 Si. NCO 5029(318) 6520(533) 0. 771 Cl. F 2 Si. NCO 5450(316) 7034(531) 0. 775 HF 2 Si. NCO 145404(8170) 190512(14020) 7. 63 * Fits for these two molecules are to the potential form: E = Ax 2 + Bx 4 + Cx 6 + Dx 8
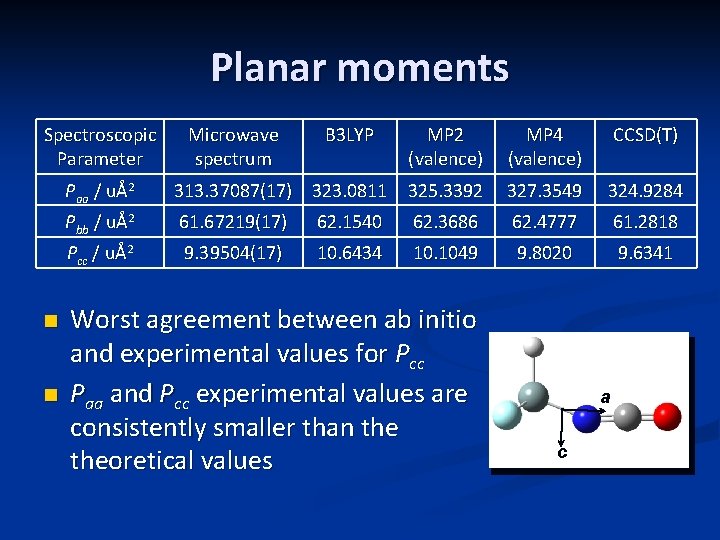
Planar moments Spectroscopic Parameter n n Microwave spectrum B 3 LYP MP 2 (valence) MP 4 (valence) CCSD(T) Paa / uÅ2 313. 37087(17) 323. 0811 325. 3392 327. 3549 324. 9284 Pbb / uÅ2 61. 67219(17) 62. 1540 62. 3686 62. 4777 61. 2818 Pcc / uÅ2 9. 39504(17) 10. 6434 10. 1049 9. 8020 9. 6341 Worst agreement between ab initio and experimental values for Pcc Paa and Pcc experimental values are consistently smaller than theoretical values a c
- Slides: 28