Role and Responsibilities of the Principal Investigator and

Role and Responsibilities of the Principal Investigator and Executive Committee Rebecca Torguson, MPH Director, CRI Clinical Research Organization Washington Hospital Center, Washington DC

Rebecca Torguson, MPH I have no real or apparent conflicts of interest to report.
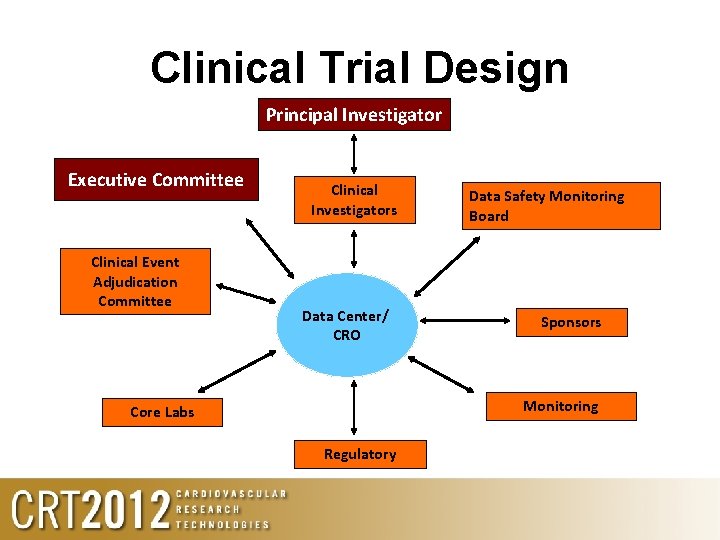
Clinical Trial Design Principal Investigator Executive Committee Clinical Event Adjudication Committee Clinical Investigators Data Center/ CRO Data Safety Monitoring Board Sponsors Monitoring Core Labs Regulatory

Principal Investigator ROLE: to facilitate the conduct of objective research that generates independent, high quality, and reproducible results. • • Oversee the design, conduct, and reporting of the trial as the lead clinician Maintain collaborative relationships and routine communication with all organizational and clinical players including: *Sponsor *Regulatory agencies and advisors *Study Committees *Site Investigators and staff *Clinical Research Organization *Core Laboratories *Data Center *Other study team members Direct compliance, financial (IIT), personnel, and other related aspects of the research project To assure research is conducted in accordance with Local and Federal regulations and sponsoring agency policies and procedures.

Roles and Responsibilities Pre-Initiation: Principal Investigator • Protocol development • Grant application and budget development • Regulatory submissions and approvals – Assist sponsors with achieving approval of IDE or IND applications or hold such themselves in the case of an Investigator initiated trial • Selection of trial collaborators – Data Center, Core Laboratories, Monitors, CRO/ARO, etc. • Logistical planning – Data collection, ensure to correct variables are collected to assess the trial endpoints • Site recruitment

Roles and Responsibilities Pre-Initiation: Principal Investigator Site Selection: • Qualified site investigator, through training and experience • • • Responsible for the safety of participants at the site Conduct the study according to the protocol and Investigator Brochure Comply with local and federal regulations Personally conduct or supervise the trial – ensure training of designees Obtain informed consent and promptly report adverse events Investigator and site interest Adequacy of staffing, facility and potential patient population Competing studies Timeline for IRB approvals, and other start-up activities

Roles and Responsibilities Enrollment: Principal Investigator • Facilitate Investigator Meetings • Communicate with sites and staff on study progression (eg: Newsletters, teleconferences, etc) • Review unexpected events, adverse events, and protocol deviations • Ensure data quality and integrity is maintained with periodic review • Receive feedback from DSMC and EC

Roles and Responsibilities End of Study: Principal Investigator • Close out of sites: – ensuring reconciliation of drug/device inventory, – regulatory obligations • Data query resolution and data lock • Ensure all financial obligations are met • Final report completion and submission to regulatory agency • Publication(s)

Executive Committee ROLE: The Executive Committee (EC), or steering committee, provides overall supervision and guidance of the conduct of the clinical trial. It is typically a team of clinicians, who may represent different disciplines related to the trial. • Operate in compliance with the EC charter, which outlines the purpose, scope of responsibilities and anticipated meeting schedule • Ensure all aspects of the trial are being conducted as efficiently as possible to achieve the objectives of the study, while maintaining data quality, integrity and patient safety without review of the data. • Offer advice and recommendations to the Principal Investigator and other members of the clinical trial team on all aspects of the trial, continuation, termination, or other decisions or changes being considered.

Roles and Responsibilities Pre-Initiation: Executive Committee • Final protocol review • Recommendations regarding clinical trial logistics • Sites, Study teams, etc.

Roles and Responsibilities Enrollment: Executive Committee • Conduct routine and emergency meetings as needed, and provide feedback and recommendations on trial conduct

Roles and Responsibilities End of Study: Executive Committee • Provide guidance for next steps with device or drug • Some may help establish or serve as the publication committee for the trial or series of trials to ensure fair and complete publication of the data
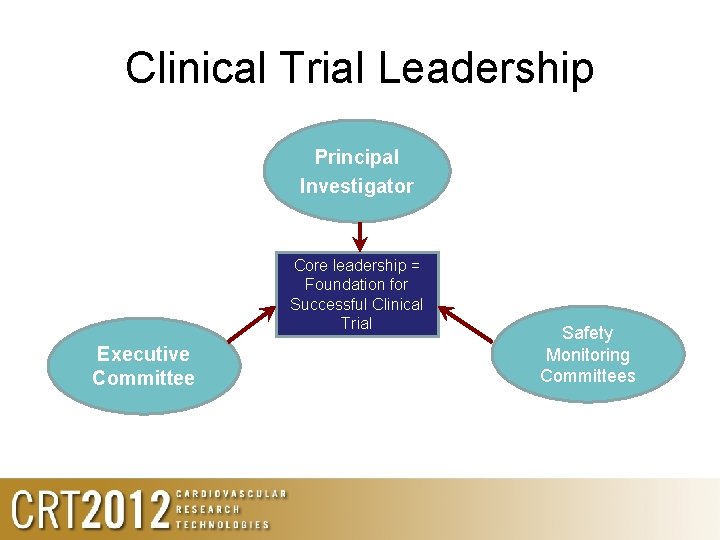
Clinical Trial Leadership Principal Investigator Core leadership = Foundation for Successful Clinical Trial Executive Committee Safety Monitoring Committees

Summary • The role and responsibilities of the Study PI are dynamic and warrant a lot of dedication from the PI to ensure all aspect of the trial are met • The role and responsibilities of the Study Executive Committee are also dynamic, but are more global than those of the Study PI
- Slides: 14