Rocks Earths Rocks 2 Sedimentary and Metamorphic Environments





























- Slides: 35

Rocks: Earth’s Rocks 2: Sedimentary and Metamorphic Environments

Sedimentary Rocks Sedimentary rocks are formed by the accumulation and hardening of sediment. Three kinds of sediment: Clastic sediment - consisting of particles derived from preexisting rocks (e. g. sand) Chemical sediment - consisting of mineral matter precipitated from a solution (e. g. salt) Biogenic sediment- consisting of materials produced by organisms (e. g. shells, bone, teeth, leaves, wood, etc. )

Clastic Sediment: Weathering When exposed at Earth’s surface, rocks are broken down by processes of weathering Mechanical weathering: physical breakup or disintegration of rocks without changes in their composition. This is accomplished mainly by physical agents such as water, wind and ice but can be aided by biological factors (e. g. widening of cracks in bedrock by tree roots). Chemical weathering: breakdown or decomposition of minerals due to chemical reaction of minerals with water or gases in the air.

Clastic Sediment: Mechanical Weathering Mechanical weathering basically serves to break rock into smaller particles. In areas where the degree of chemical weathering is very low (e. g. in cold, dry, regions of the Canadian Arctic), clastic sediment can consist almost entirely of small fragments of rock with no change in mineral makeup.

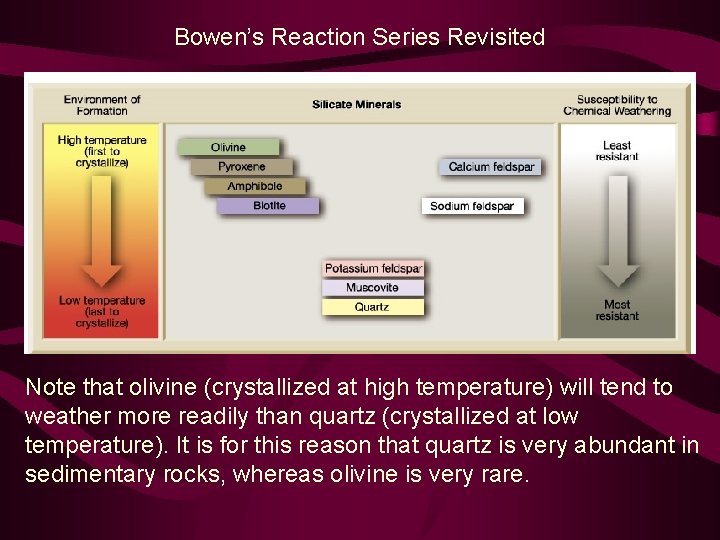
Clastic Sediment: Chemical Weathering Some minerals are more susceptible to weathering than others. For minerals in igneous rocks, resistance to weathering basically follows the reverse trend of same trend as the order of crystallization in Bowen’s Reaction Series. This is because high-temperature minerals are less stable at Earth’s surface than low-temperature minerals. Note that Earth’s surface is a lot cooler than the environments in which minerals form from magma; low-temperature minerals are most stable at Earth’s surface.

Bowen’s Reaction Series Revisited Note that olivine (crystallized at high temperature) will tend to weather more readily than quartz (crystallized at low temperature). It is for this reason that quartz is very abundant in sedimentary rocks, whereas olivine is very rare.

Products of Chemical Weathering Chemical weathering produces a number of minerals and free ions. The primary mineral products of weathering are quartz and clay – these products form the bulk of sedimentary particles. Iron oxides (hematite) can also be left behind as residue. The remaining material is dissolved in water, in the form of ions.

Climate and Sediment Composition Climate has a strong effect on the characteristics of clastic sediment. Sediment produced in cold, dry areas (where mechanical weathering dominates) tends to contain rock fragments of variable mineral content. Sediment produced in warm, wet areas (where chemical weathering dominates) tends to be composed largely of quartz, clay and iron oxides.

Clastic Sediment: Transportation Once dislodged (eroded), sedimentary particles can be transported away from their source area by: Gravity Water Wind Ice (Mass Wasting) (Rivers) (e. g. Dust Storms) (Glaciers)

Clastic Sediment: Deposition Sedimentary particles ultimately come to rest once the transporting medium can no longer carry them. As a general rule, smaller/lighter particles are deposited in less-agitated conditions than larger/heavier particles when transported by wind or water. Sedimentary particles can therefore experience some degree of sorting. Beach sand tends to be well sorted, due to constant wave action. Sedimentary particles transported and deposited rapidly by events such as mudflows tend be poorly sorted. beach mudflow Well-sorted sediment Poorly sorted sediment

Depositional Environments Clastic sediments can be deposited in a wide variety of settingscharacteristics of clastic sedimentary rocks can provide information on where their constituent sediments were originally deposited. We will look at some of these environments in greater detail as the term progresses.

Sediment to Sedimentary Rock: Lithification Once buried, sediment undergoes changes that transform it into rock. This transformation is called lithification (lithos = stone) Compaction: As sediment layers are buried to deeper and deeper levels under successive sediment layers, sedimentary particles are squeezed together as the spaces between them decrease in size. The material thus becomes more rock-like. Cementation: Sediment grains can also become cemented together by minerals that precipitate from water remaining in the pore spaces between the grains. Grains therefore effectively become glued together.

Common Clastic Sedimentary Rocks Clastic sedimentary rocks are classified primarily by grain size. Sediment type Rock Type Gravel (pebbles, cobbles, boulders) Conglomerate Sand (sand-sized particles that are obviously gritty) Sandstone Silt (particles barely discernible under low magnification) Siltstone Clay (particles too small to be seen under low magnification) Shale

Chemical Sediment Water on Earth’s surface contains dissolved ions (electrically charged atoms or groups of atoms of various elements) principally derived from weathered minerals and volcanic gases. Dissolved Components in Seawater (Percent by Weight) Positive Ions Sodium (Na+): 30. 61 % Magnesium (Mg 2+ ): 3. 69 % Calcium (Ca 2+): 1. 16 % Potassium (K+): 1. 10 % Negative Ions Chloride (Cl-): 55. 04 % Sulphate (SO 42 -): 7. 68 % Others: 0. 72 % Include trace amounts of metals (gold, silver, zinc, lead, copper, etc. )

For example… HF HCl CO 2 Carried in water vapour HF HCl SO 2 Reacts with water in atmosphere H 2 SO 4 H 2 O Acid rain Pyroclastic debris (pulverized rock) Ions dissolved in seawater H+ Cl- SO 42 - F-

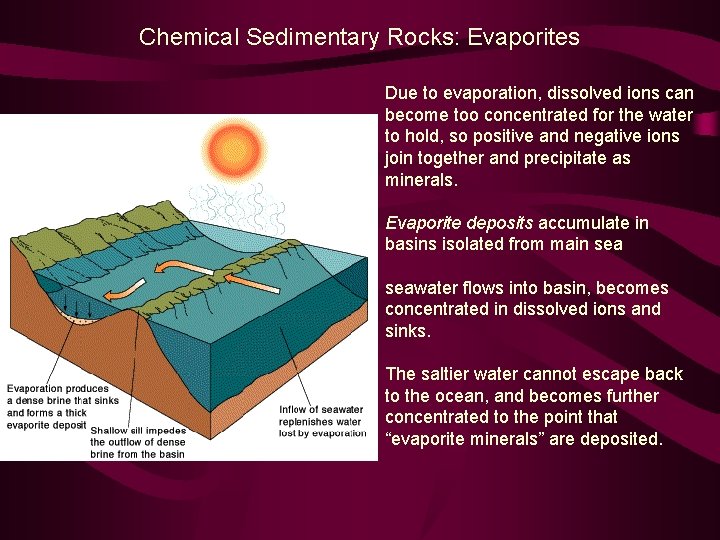
Chemical Sedimentary Rocks: Evaporites Due to evaporation, dissolved ions can become too concentrated for the water to hold, so positive and negative ions join together and precipitate as minerals. Evaporite deposits accumulate in basins isolated from main seawater flows into basin, becomes concentrated in dissolved ions and sinks. The saltier water cannot escape back to the ocean, and becomes further concentrated to the point that “evaporite minerals” are deposited.

Two common minerals in evaporite deposits are: Gypsum (calcium sulphate) and Halite (sodium chloride). In some cases, rocks can be composed exclusively of one of these minerals. A rock formed exclusively of halite is called rock salt (used as table salt) A rock formed exclusively of gypsum is called alabaster or “rock gypsum” (commonly used in sculpture)

Chemical Sedimentary Rocks: Chemical Limestones Limestone is a sedimentary rock dominated by the mineral calcite. In some caves and hotsprings, concentrations of calcium ions (Ca 2+) and the complex ion carbonate (CO 3)2 - can reach sufficient levels to allow the precipitation of calcite (Ca. CO 3). A travertine sample This forms a banded variety of limestone called travertine. Some forms of travertine also display pores that are produced from the liberation of gases by bacteria. Travertine with abundant pores

Travertine Structures Travertine stalactites on the ceiling of a cave (Lehman Caves, Nevada, U. S. A. ) Travertine terraces in Yellowstone National Park (Wyoming, U. S. A. )

Biogenic Sediment Biochemical sediment consists of materials that are produced by chemical processes associated with biological activity. So one can think of biochemical sediment as being chemical sediment formed through biological activities. Biogenic components include: Shells Bones Teeth Plant Remains

Biogenic Sedimentary Rocks Two common rocks that are formed from biogenic sediment are fossiliferous limestone, chert, and coal. Fossiliferous limestone is composed almost entirely of calcite shells (skeletons) of organisms. In many cases, the identity of the former owners of skeletal material can be identified. This piece of limestone contains broken “stem” segments of animal called a crinoid.

Biogenic Sedimentary Rocks A familiar rock that, in a loose sense, can also be considered a variety of fossiliferous limestone is chalk, which is made of microscopic skeletons of algae.

Chert is another common biochemical sedimentary rock. A few organisms, such as some microscopic planktonic organisms (as well as some sponges), have a skeleton made of silica. The silica can dissolve and form a “gel” on the seafloor. When this gel solidifies, it forms a finely crystalline rock called chert. As it is basically made of very fine-grained quartz crystals, it is very hard. Chert Silica skeletons of planktonic organisms

Coal is a special type of biogenic sedimentary rock that is largely composed of organic matter from plants. Coal seams represent large accumulations of organic matter that were deposited in swamps and were subsequently buried. swamp coal seam coal sample

Metamorphic Rocks Metamorphic rocks are formed by the transformation of preexisting rocks under the influence of high temperatures and pressures and chemically active fluids Three basic types of metamorphism: Contact metamorphism Regional metamorphism Metasomatism

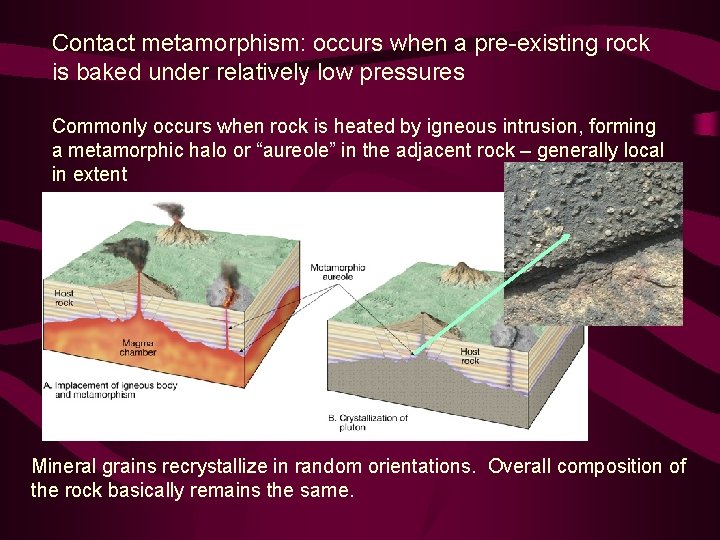
Contact metamorphism: occurs when a pre-existing rock is baked under relatively low pressures Commonly occurs when rock is heated by igneous intrusion, forming a metamorphic halo or “aureole” in the adjacent rock – generally local in extent Mineral grains recrystallize in random orientations. Overall composition of the rock basically remains the same.

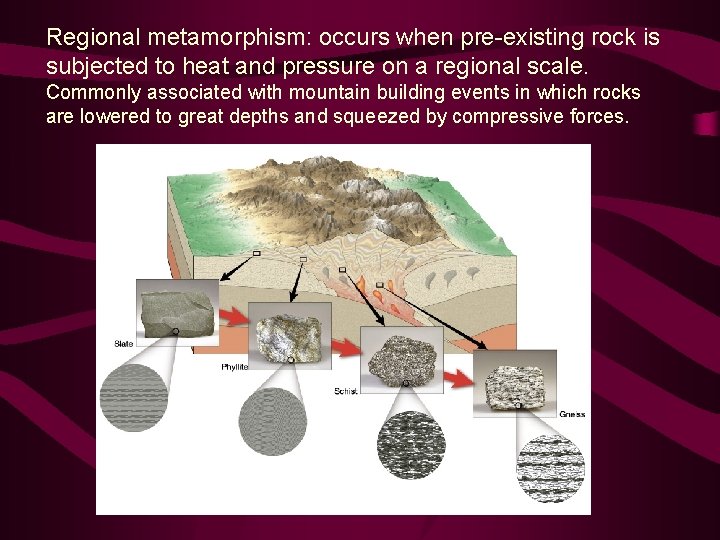
Regional metamorphism: occurs when pre-existing rock is subjected to heat and pressure on a regional scale. Commonly associated with mountain building events in which rocks are lowered to great depths and squeezed by compressive forces.

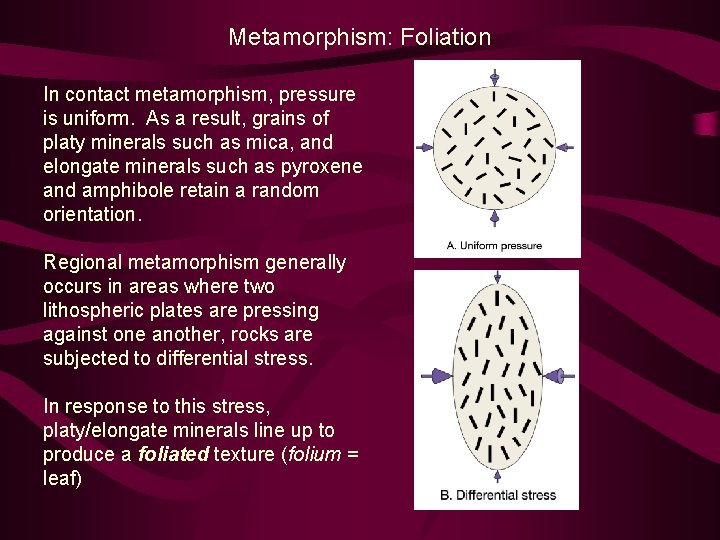
Metamorphism: Foliation In contact metamorphism, pressure is uniform. As a result, grains of platy minerals such as mica, and elongate minerals such as pyroxene and amphibole retain a random orientation. Regional metamorphism generally occurs in areas where two lithospheric plates are pressing against one another, rocks are subjected to differential stress. In response to this stress, platy/elongate minerals line up to produce a foliated texture (folium = leaf)

Common Metamorphic Rocks Non-foliated rocks Some metamorphic rocks, whether formed by contact or regional metamorphism always have a non-foliated (also called granoblastic) texture. This is because they lack platy/elongate minerals required to define foliation. Such rocks include quartzite (formed via metamorphism of quartz sandstone), and marble (formed via metamorphism of limestone). In both cases, mineral grains grow in size and form an interlocking texture. Fossils once present in the original sedimentary rock are obliterated due to this recrystallization.

Common non-foliated (granoblastic) metamorphic rocks Heat (+ pressure) Limestone Marble Heat (+ pressure) Quartz sandstone Quartzite

Common Foliated Metamorphic Rocks Foliated rocks Rocks containing platy/elongate minerals that subjected to regional metamorphism (and therefore) affected by differential pressure are typically foliated. Increasing intensity of metamorphism (called “grade”) results in increased size of mineral grains, and the development of distinct types of foliation. With increasing metamorphism, shale changes into the following rock types: Shale Slate Phyllite Schist Gneiss

Common foliated rocks Increasing temperature and pressure) Slate: very slight sheen due to growth of platy mica grains Phyllite: distinct sheen due to continued growth of platy mica grains Schist: very sparkly appearance due to large mica grains Gneiss: banded appearance due to separation of darkand light-coloured minerals

Metasomatism: occurs when fluids (generally water or carbon dioxide) react with a pre-existing rock and alter the chemical composition of minerals within the rock. In some cases, the fluid itself is involved. In others, substances dissolved in the fluid are involved. For example, the mineral olivine (which occurs in blocky crystals) reacts with water to form the mineral serpentine (with platy to fibrous crystals). 2 Mg 2 Si. O 4 olivine + 2 H 2 O water Mg 3 Si 2 O 5(OH)4 serpentine + Mg. O (in solution)

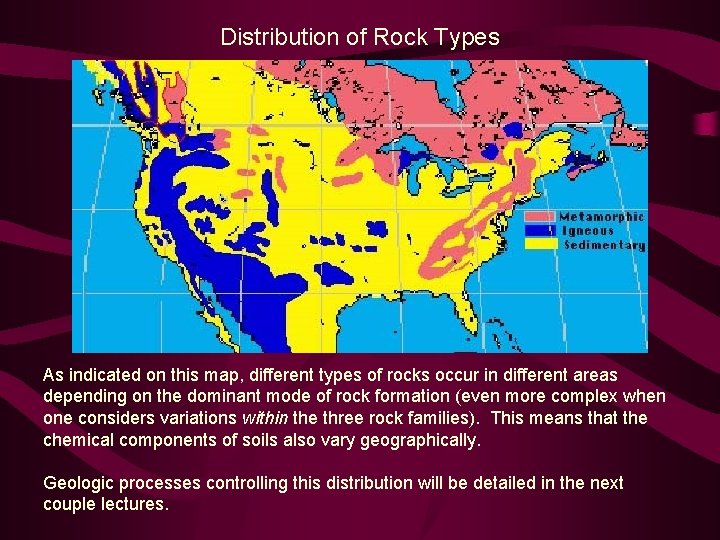
Distribution of Rock Types As indicated on this map, different types of rocks occur in different areas depending on the dominant mode of rock formation (even more complex when one considers variations within the three rock families). This means that the chemical components of soils also vary geographically. Geologic processes controlling this distribution will be detailed in the next couple lectures.

END OF LECTURE