Rocks and Mineral Continued Metamorphic Rocks Metamorphic rocks


















- Slides: 19

Rocks and Mineral Continued

Metamorphic Rocks Metamorphic rocks are rocks that have "morphed" into another kind of rock. These rocks were once igneous or sedimentary rocks. � The rocks are under tons and tons of pressure, which fosters heat build up, and this causes them to change underground. � As a result, most of the thousands of rare minerals known to science occur in metamorphic rocks. � The presence of mineral layers, called foliation, is important to observe when identifying a metamorphic rock. �

Examples of Metamorphic Rocks � Schist Parent- Shale or Phyllite � Gneiss Parent- Granitic and volcanic rocks � Slate Parent- Shale � Phyllite Parent. Mudstones and shale � Marble Parent. Limestone � Quartzite Parent- Quartz sandstone

Physical Properties of Minerals � Physical Properties- Properties used to visually identify a specific mineral � Color � Luster � Transparency � Crystal systems � Cleavage � Fracture � Hardness � Specific Gravity � Streak

Color � Color is the first thing someone notices when they view a mineral. Color is also one of the big reasons that attract people to minerals. Color in minerals is caused by the absorption, or lack of absorption, of various wavelengths of light. Many minerals come in different colors and some minerals' colors are identical to other minerals' colors. � Ex. Quartz can come in pink, purple, white, golden yellow or smoky grey.

Luster � Luster is a description of the way light interacts with the surface of a crystal. � Types of Luster- 2 main categories Metallic - the look of metals (shiny) Ex. Gold, Silver, Galena, Pyrite Non-metallic- not shiny � Ex. Sulfur, Feldspar, Talc

Types of Luster Adamantine-Clear and Brilliant like a diamond (most gems) � Dull- a non-reflective surface of any kind � Earthy- the look of dirt or dried mud � Pearly- the look of a pearl � Waxy- the look of wax � Fibrous- the look of fibers � Greasy- the look of grease � Resinous- the look of resins such as dried glue or chewing gum � Vitreous-the most common luster, it simply means the look of glass �

Transparency Transparent- light enters and exits the surface of the substance in relatively undisturbed fashion. Can see all visible light and can distinguish an object when looking through it. � Ex. Diamond � Translucent- light enters and exits the surface of the substance in relatively undisturbed fashion. Can see light through it, but can not clearly distinguish an object while looking through it. � Ex. Quartz � Opaque- If the light can not even penetrate the surface of the substance. � Ex. Feldspar �

Crystal Systems � 1. 2. 3. 4. 5. 6. 7. A mineral has flat sides called faces. These faces are flat and join at different angles. The joined faces make a certain shape. Cubic- Crystals grow in the shape of a cube Tetragonal- square bottom and top face with a rectangular body. Orthorhombic- a rectangular prism with a rectangular base Monoclinic-They form a rectangular prism with a parallelogram as its base. Triclinic. Hexagonal- face forms a hexagon Trigonal-similar to a cube that has been compressed to one side.

Cleavage › Crystal cleavage is a smooth break producing what appears to be a flat crystal face. › Cleavage is reproducible, meaning that a crystal can be broken along the same parallel plane over and over again › All cleavage must parallel › All cleavage planes of a mineral must match that mineral's crystal system › The same mineral will always, always have the same cleavage › Cleavage occurs in minerals that have specific planes of weakness � Ex. Fluorite= Octahedral Cleavage, Calcite= Rhombohedral, Halite=cubic

Fracture The way a Rock Breaks Types of Fracture � Conchoidal- smoothly curved fracture that looks like a piece of chipped or broken glass. Sometimes described as a clam-shell fracture. � Ex. Quartz, Obsidian � Uneven- basically self explanatory, the fracture does not have a pattern and can not be identically repeated � Ex. Anhydrite � Jagged- has sharp points or edges that catch on a finger that's rubbed across the surface. � Ex. Metals such as Copper �

Hardness- measure of the strength of the structure of the mineral relative to the strength of its chemical bonds. � Minerals with small atoms, packed tightly together with strong covalent bonds throughout tend to be the hardest minerals. � The softest minerals have metallic bonds or even weaker van der Waals bonds as important components of their structure � Hardness can be tested through scratching. A mineral can only be scratched by a harder substance. � Use Mohs Hardness Scale �

Moh’s Hardness Scale � Mohs Hardness Scale Talc 2. Gypsum 3. Calcite 4. Fluorite 5. Apatite 6. Orthoclase 7. Quartz 8. Topaz 9. Corundum (ruby and sapphire) 10. Diamond 1.

Specific Gravity- measure of the density of a mineral. � Non-metallic minerals tend to be of a low density � Metallic minerals tend to be of higher density � HEFTING- To use specific gravity, hold a mineral of unknown SG in one hand in the other hand a mineral of known SG preferably one near the average of 2. 75 and of the same size as the unknown mineral; then compare. � Ex. Galena vs. Quartz �

Streak � � � Streak is actually the color of the powder of a mineral. Streak is closely related to color, but is a different property because the color of the mineral may be different than the color of the streak The proper way to test for streak is to rub a mineral across a tile of white unglazed porcelain and to examine the color of the "streak" left behind. Two minerals that have similar outward color may have different colors when powdered. Ex. the minerals hematite and galena can be confused when both have a gray color. However, hematite's streak is reddish-brown, while galena's streak is lead gray.

Bowen’s Reaction Series When two tectonic plates collide one may be pushed down into the mantle. It heats and melts � Magma that is made will have different chemical composition depending on where it comes from. � Magmas from Continental Crust - rich in silicon and aluminum � Magmas from oceanic crust - rich in iron and magnesium � Composition determines which rocks will be made when magma cools �

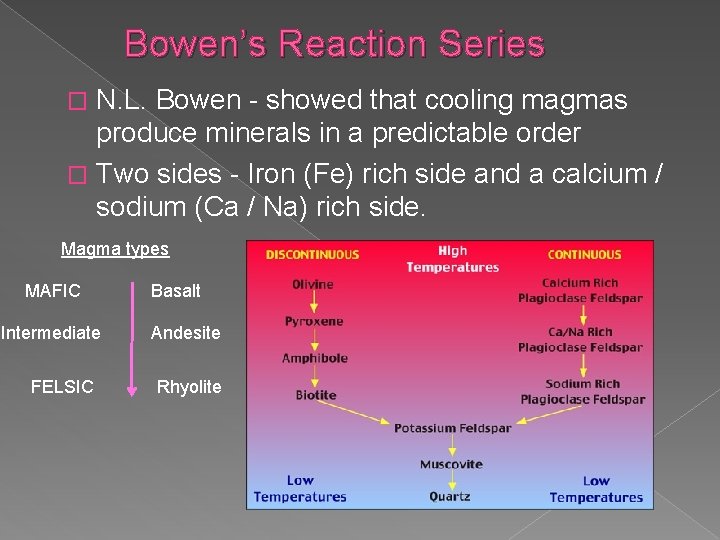
Bowen’s Reaction Series N. L. Bowen - showed that cooling magmas produce minerals in a predictable order � Two sides - Iron (Fe) rich side and a calcium / sodium (Ca / Na) rich side. � Magma types MAFIC Intermediate FELSIC Basalt Andesite Rhyolite

Bowen’s Reaction Series Olivine - First Fe rich mineral to form - in tetrahedra shape - easily weathered on the surface � Pyroxenes - Tetrahedra link in long chains - also weather easily � Amphiboles - double strands made from linking pyroxenes � Biotite Micas - amphibole chains make sheets �

Bowen’s Reaction Series � Calcium/Sodium Feldspar - from the Ca/Na side like olivine but Al is inside the tetrahedra instead of Silicon � Potassium Feldspars - as temp cools Ca and Na replace Al inside the tetrahedron � Quartz - very low temperatures - little left in magma except Si and O -three dimensional networks