RMTC Anabolic Steroid Studies Richard Sams Ph D

RMTC Anabolic Steroid Studies Richard Sams, Ph. D Florida Racing Laboratory University of Florida and Scott D. Stanley, Ph. D Kenneth L. Maddy Equine Chemistry Laboratory University of California at Davis

Objectives l Characterize plasma concentrations of the anabolic steroids in exercised Thoroughbred horses (geldings and mares) of racing age after administration of race track or label dose commercial and compounded formulation in a sufficient number of animals to obtain statistically meaningful results.

Why study plasma? l l Subject to less variability than urine concentrations Easier to perform assay on plasma than urine l l Anabolic steroids are detectable in plasma by modern LC-MS methods for clinically relevant periods Anabolic steroids are excreted in urine as metabolites or mixtures of metabolites that more require extensive sample preparation.

Responsibilities for RMTC Studies l Protocols for AAS studies l l Drug administrations and sample collections l l Developed by working advisory group including Drs. Walter Hyde, Scott Stanley, Lawrence Soma, and Richard Sams conducted under the direction of Dr. Patrick Colahan at the Florida Equine Performance Laboratory at the University of Florida Sample analyses l conducted under the direction of Dr. Richard Sams at the Florida Racing Laboratory at the University of Florida and Dr. Scott Stanley at the Kenneth L. Maddy Equine Analytical Chemistry Laboratory at the University of California at Davis

General Description of Studies l Experimental animals l l Source of AAS l l administered at the label dose for horses or at a commonly used race track dose Dosing regimen l l purchased from commercial sources or a compounding pharmacy Dose l l Thoroughbred horses (geldings and mares; aged 3 -10 years) that are subjected to a conditioning regimen on a treadmill administered once IM or subcutaneously Sample Collection schedule l Blood and urine samples were collected from each horse according to a pre-determined schedule. Samples were collected until the AAS was no longer detectable in two successive blood samples

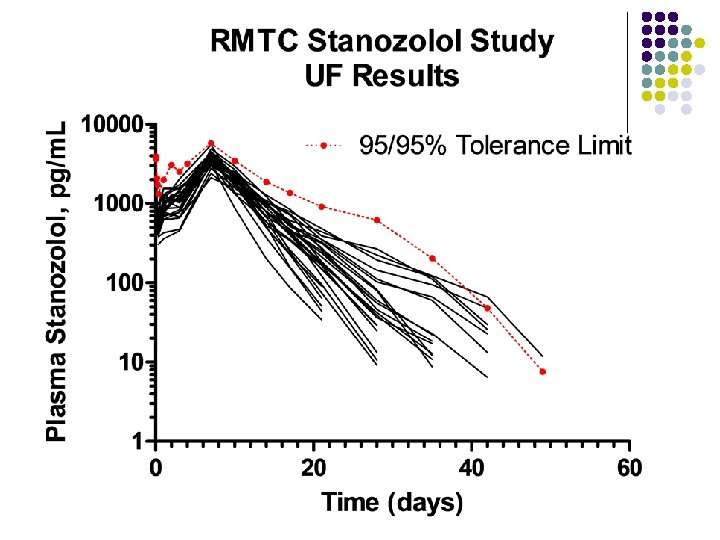
General Description of Studies l Plasma AAS concentrations l l determined by liquid chromatographic-mass spectrometric methods developed and validated in the two analytical laboratories. Plasma concentrations from the two laboratories are being compared and differences, if any, investigated. Plasma threshold concentrations corresponding to the “ 95/95 tolerance limit” are being calculated by standard methods. Urine concentrations l l to be determined later relationships between plasma and urine concentrations to be investigated.

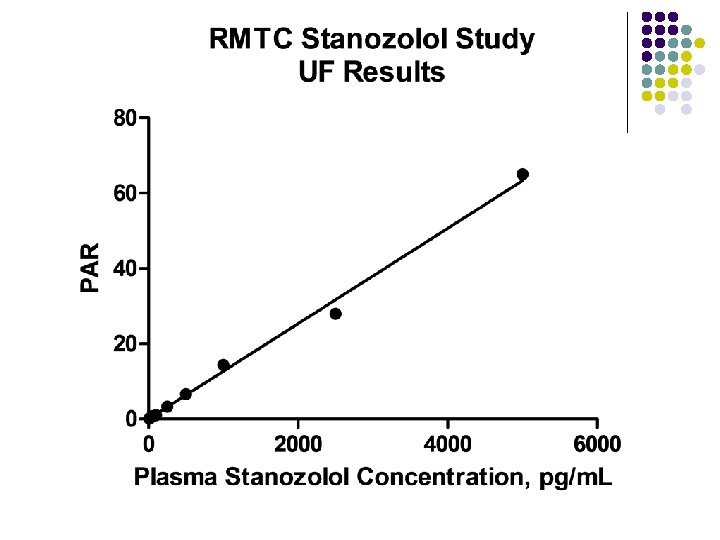
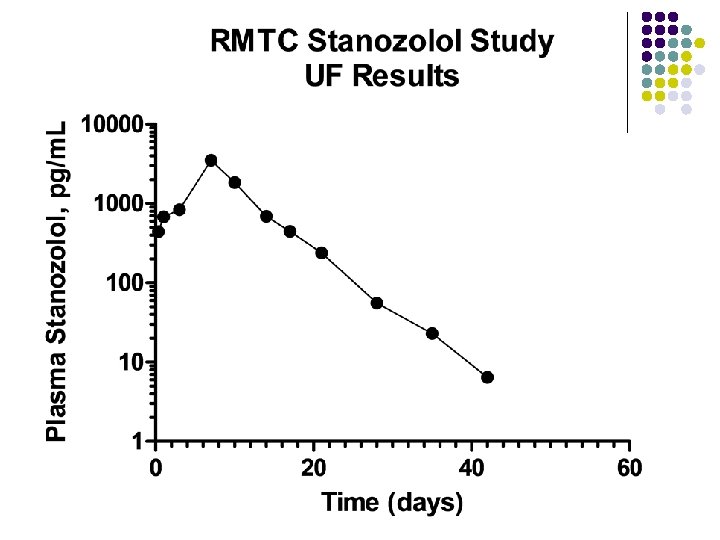
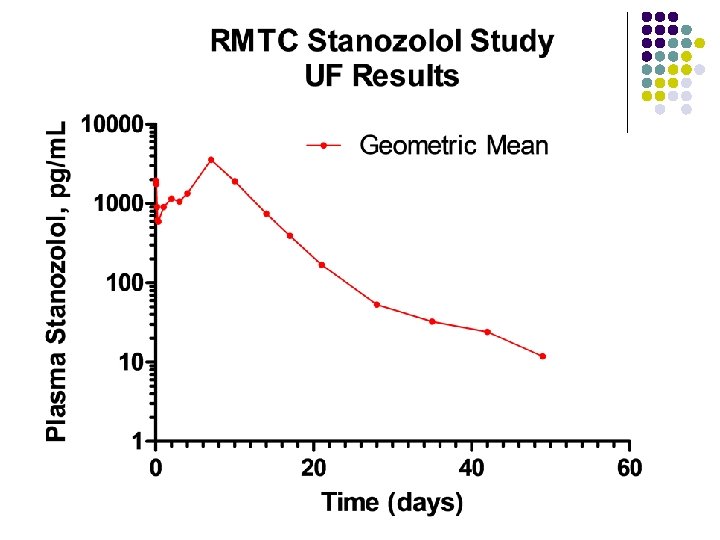
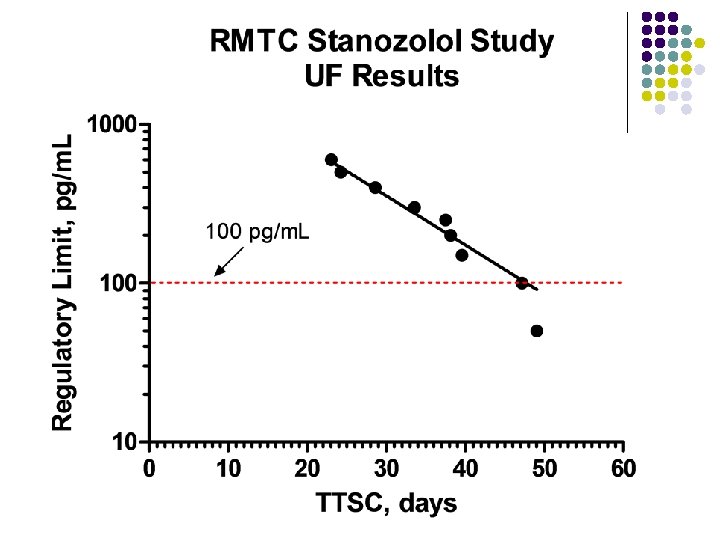
RMTC Stanozolol Study l Formulation l l Source l l l Frank’s Pharmacy, Ocala Florida Compounded product – Winstrol® is not available Dose l l Aqueous suspension 0. 55 mg/kg IM Subjects l 26 Thoroughbred horses

RMTC Stanozolol Study l Method l LC-MS l Validated at UF and UCD laboratories § § § l Linear LOQ = 25 pg/m. L LOD ~10 pg/m. L Sample collections l l l Stopped at 63 days Plasma concentrations below LOD for 2 successive collections Plasma concentration above LOD at 49 days

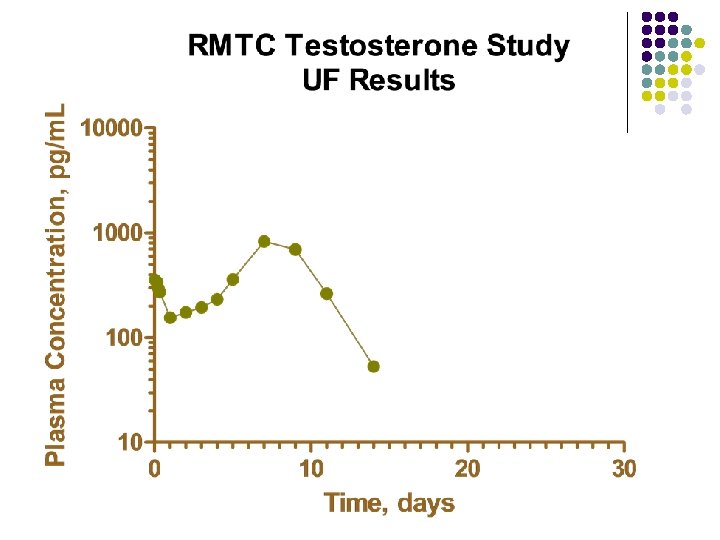
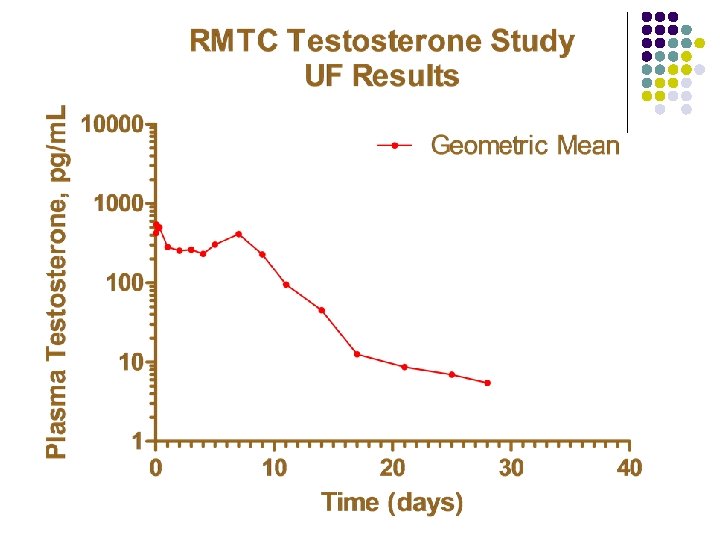
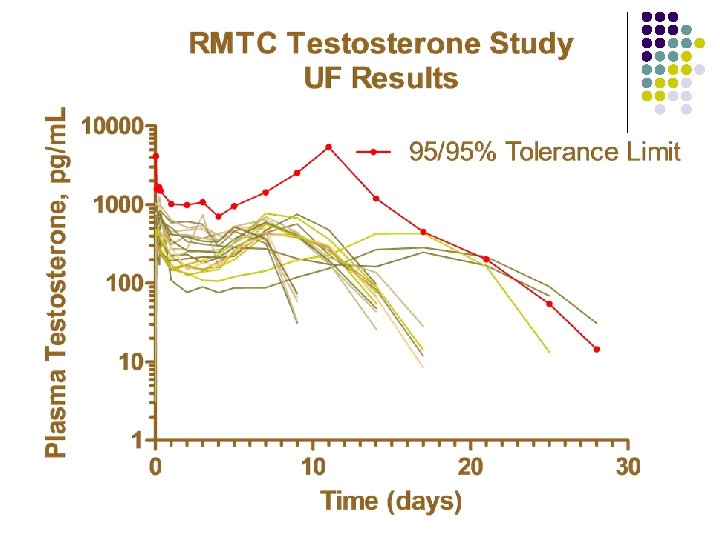
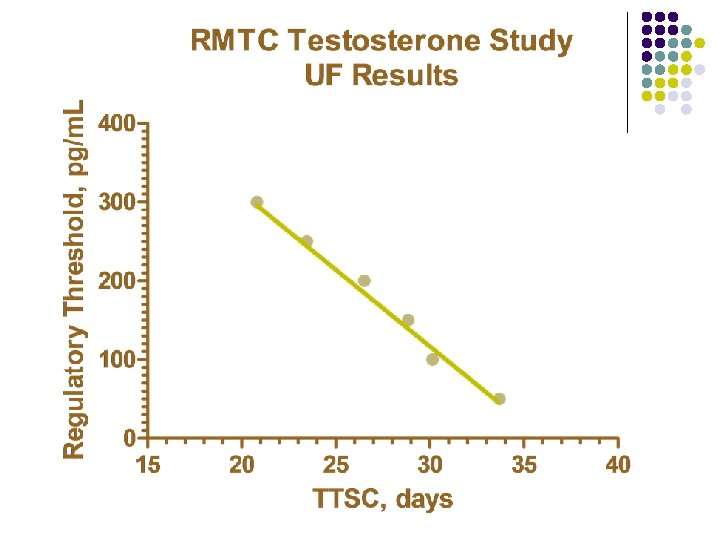
RMTC Testosterone Study l Formulation l l Source l l l Frank’s Pharmacy, Ocala Florida Compounded product Dose l l Aqueous suspension 0. 15 mg/kg subcutaneously Subjects l 20 Thoroughbred horses

RMTC Testosterone Study l Method l LC-MS l Validated at UF and UCD laboratories § § § l Linear LOQ = 25 pg/m. L LOD ~10 pg/m. L Sample collections l l l Stopped at 34 days Plasma concentrations below LOD for 2 successive collections Plasma concentration less than LOQ at 28 days

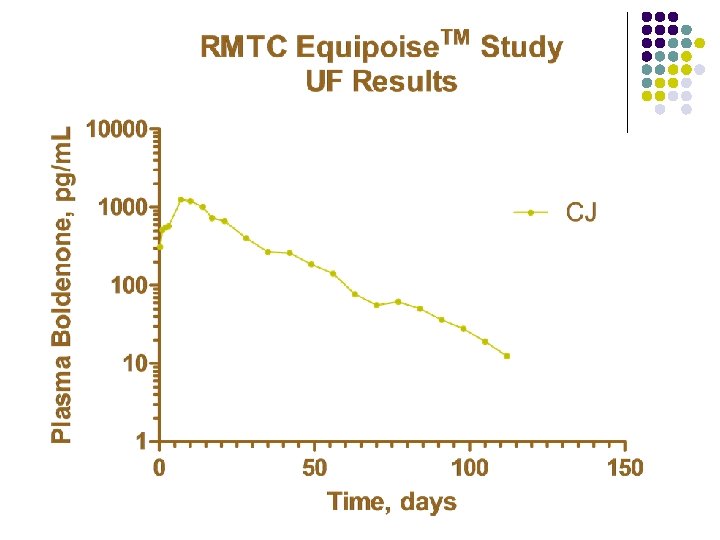
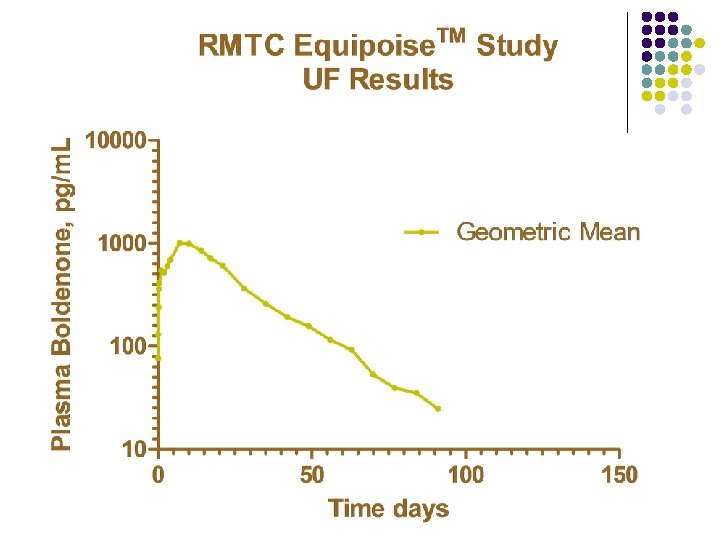
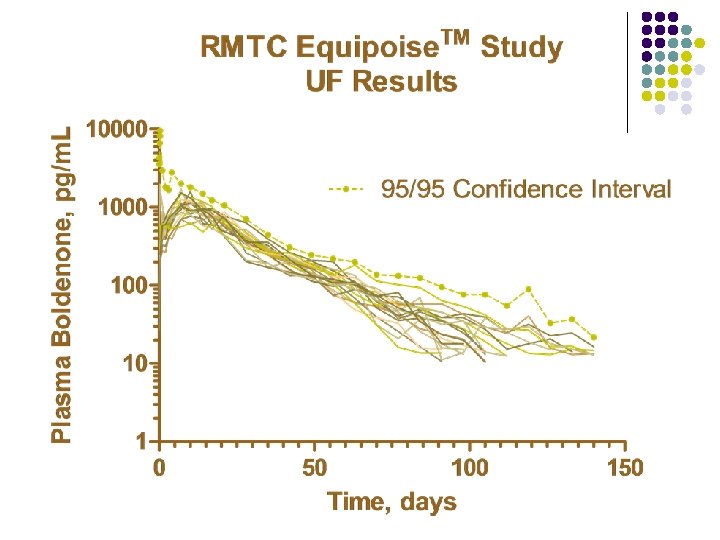
RMTC Boldenone Study l Formulation l l Source l l Fort Dodge, Manhattan, Kansas Dose l l boldenone undecylenate in oil (Equipoise®) 1. 1 mg/kg IM Subjects l 20 Thoroughbred horses

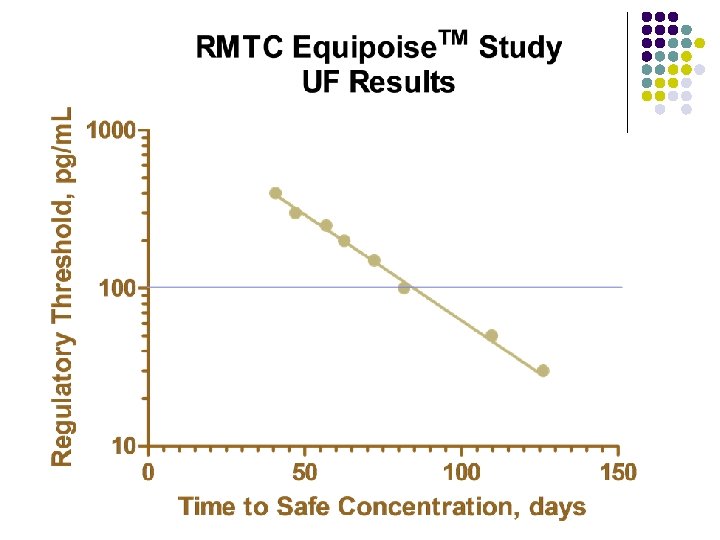
RMTC Boldenone Study l Method l LC-MS l Validated at UF and UCD laboratories § § § l Linear LOQ = 25 pg/m. L LOD ~10 pg/m. L Sample Collections l l l Stopped at 164 days Plasma concentrations below LOD for 2 successive collections Plasma concentration less than LOQ at 140 days


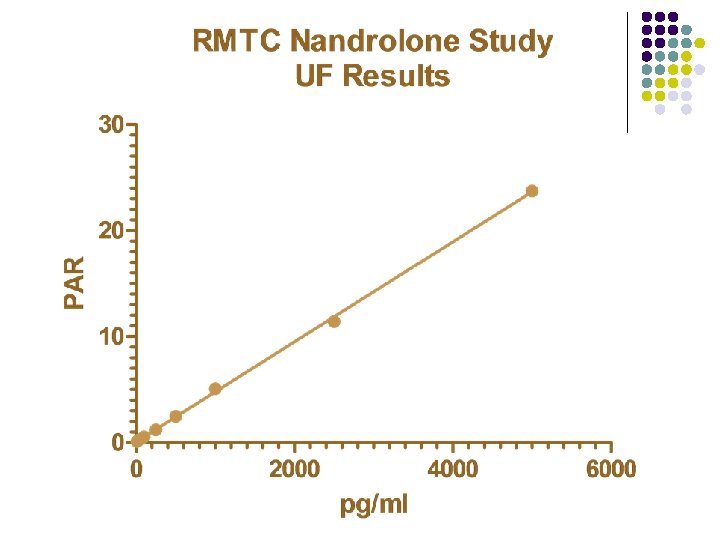
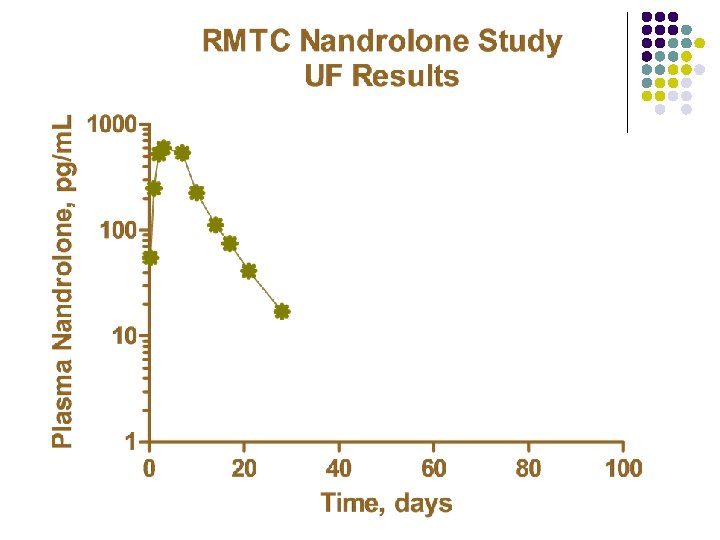
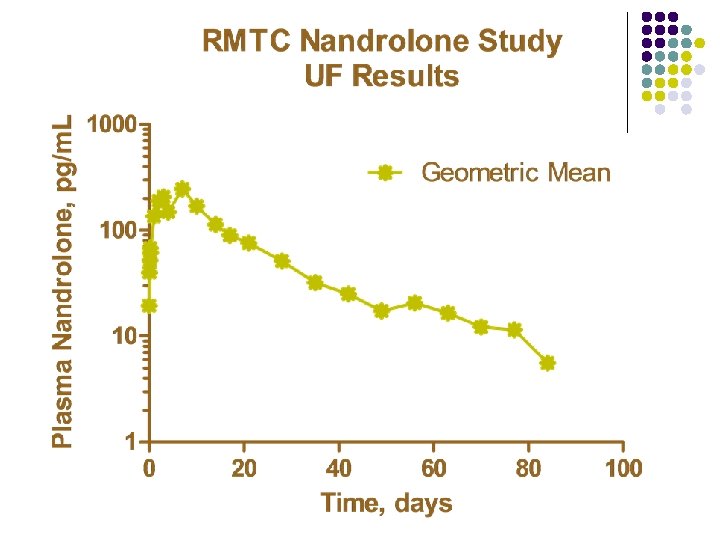
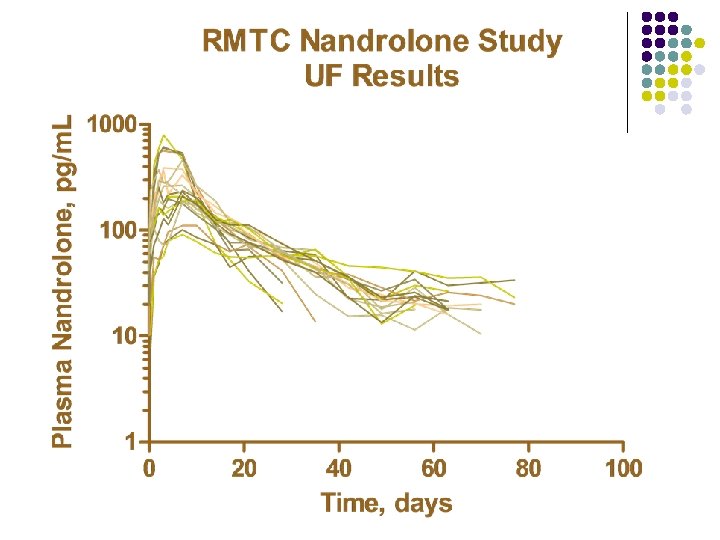
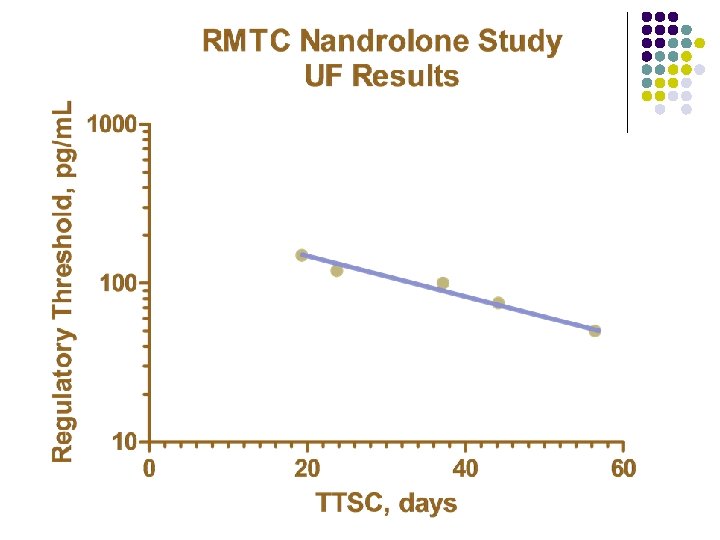
RMTC Nandrolone Study l Formulation l l Source l l Frank’s Pharmacy, Ocala, Florida Dose l l nandrolone decanoate in oil 0. 55 mg/kg IM Subjects l 20 Thoroughbred horses

RMTC Nandrolone Study l Method l LC-MS l Validated at UF and UCD laboratories § § § l Linear LOQ = 25 pg/m. L LOD ~10 pg/m. L Sample Collections l l l Stopped at 91 days Plasma concentrations below LOD for 2 successive collections Plasma concentrations less than LOQ at 77 days

Summary l Stanozolol l l Aqueous suspension from compounding pharmacy 0. 55 mg/kg intramuscular injection LOQ 25 pg/m. L No plasma concentrations above 25 pg/m. L after 49 days Substantial horse to horse variability

Summary l Testosterone l l l Aqueous suspension from compounding pharmacy 0. 15 mg/kg subcutaneous injection LOQ 25 pg/m. L No plasma concentrations above 25 pg/m. L after 28 days Substantial horse to horse variability

Summary l Boldenone l l l Equipoise™brand of boldenone undecylenate in oil 1. 1 mg/kg intramuscular injection LOQ 25 pg/m. L Plasma concentrations above 25 pg/m. L for about 140 days Horse to horse variability less than with other formulations

Summary l Nandrolone l l l Nandrolone decanoate in oil 0. 55 mg/kg intramuscular injection LOQ 25 pg/m. L No plasma concentrations above 25 pg/m. L after 77 days Substantial horse to horse variability

Acknowledgements l UF Equine Performance Laboratory l l l Bret Rice Jennifer Claflin Patrick Colahan, DVM Carly Brown Jennifer Pingel Stephanie Salas Kristy Smith Natalia Corredor Amber Davidson Allison Hreha Kevin Le. Blanc Ted Broome, DVM l UC Davis Laboratory l l Ben Moeller Scott Stanley, Ph. D Dan Mc. Kemie UF Racing Laboratory l l Nancy Szabo, Ph. D Carolyn Diaz Joy Guingab Katherine Wilding

Acknowledgements l The authors would like to acknowledge the financial support for these projects from the Racing Medication and Testing Consortium, Inc. and the Division of Pari-Mutuel Wagering, Florida Department of Business and Professional Regulations.
- Slides: 40