Rivaroxaban Should be the Anticoagulant of Choice in

Rivaroxaban Should be the Anticoagulant of Choice in High-Risk Non Valvular Atrial Fibrillation Con: Sanjay Kaul, MD Division of Cardiology Cedars-Sinai Heart Institute Professor, Cedars-Sinai Medical Center & Geffen School of Medicine at UCLA Los Angeles, California

Sanjay Kaul, MD I have no real or apparent conflicts of interest to report.

Le Roi est mort, vive le Roi ! "The King is dead. Long live the King. " King Charles VII of France

"The King is dead. Long live the King. " Manesh Patel, MD

Christopher B. Granger, MD

Interpreting The Results of ROCKET-AF • Statistical plan Issues for Discussion - Assessment of efficacy in non-inferiority & superiority trials - Appropriate population: ITT vs On treatment (or Per-protocol) - Proper “event window” (events post treatment discontinuation) - Double counting of hemorrhagic stroke (for efficacy and safety) • Adequacy of Warfarin management - Time in therapeutic range: surrogate vs marker of quality of care - Constancy or HESDE: comparison with historical and contemporary trials - Impact on outcomes • Benefit-risk assessment - Efficacy vs safety: overall and when warfarin “skillfully used” - Cost - Lack of monitoring vs. lack of reversibility - Optimal dose (no dose-ranging study in intended population)

Objective of Noninferiority Assessment • The new treatment is not inferior to (“not much worse than”, not “as good as”) the standard treatment • Employed when standard placebo controlled trials are considered unethical or impractical • Therapeutic utility may be based on the ancillary advantages in safety, tolerability, cost, convenience

Operational Criterion for “As Effective” Federal Register 1995 60(14): 39180 -1 • “…for products intended to treat life threatening diseases, diseases with irreversible morbidity, and contagious diseases that pose serious health risks to others, it is essential for public health protection that a new therapy be as effective as existing, approved therapies. ” • • Point estimate of the test drug favored it over control Upper bound of 95% CI very close to showing superiority (HR <1. 05 -1. 10)

FDA Draft Noninferiority Guidance (2010) • Non-inferiority analysis - Both the “as treated” and ITT populations should be analyzed - Agreement of ITT and as treated analyses increases confidence in the trial’s results • Superiority analysis - The primary analysis should be ITT because as treated analysis are prone to bias from informative censoring
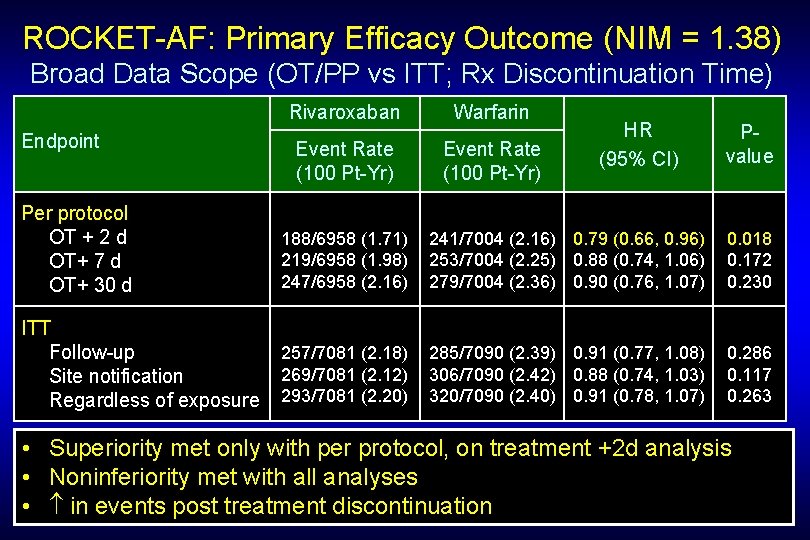
ROCKET-AF: Primary Efficacy Outcome (NIM = 1. 38) Broad Data Scope (OT/PP vs ITT; Rx Discontinuation Time) Endpoint Rivaroxaban Warfarin Event Rate (100 Pt-Yr) HR (95% CI) Pvalue Per protocol OT + 2 d OT+ 7 d OT+ 30 d 188/6958 (1. 71) 219/6958 (1. 98) 247/6958 (2. 16) 241/7004 (2. 16) 0. 79 (0. 66, 0. 96) 253/7004 (2. 25) 0. 88 (0. 74, 1. 06) 279/7004 (2. 36) 0. 90 (0. 76, 1. 07) 0. 018 0. 172 0. 230 ITT Follow-up Site notification Regardless of exposure 257/7081 (2. 18) 269/7081 (2. 12) 293/7081 (2. 20) 285/7090 (2. 39) 0. 91 (0. 77, 1. 08) 306/7090 (2. 42) 0. 88 (0. 74, 1. 03) 320/7090 (2. 40) 0. 91 (0. 78, 1. 07) 0. 286 0. 117 0. 263 • Superiority met only with per protocol, on treatment +2 d analysis • Noninferiority met with all analyses • in events post treatment discontinuation
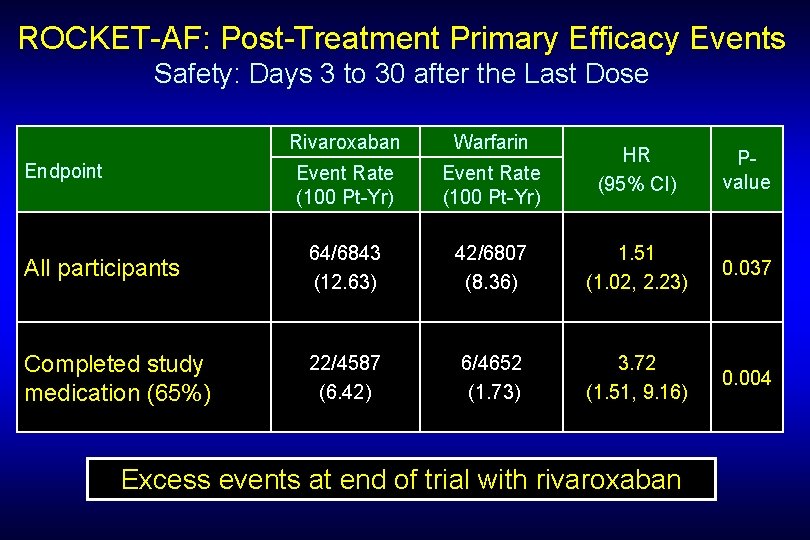
ROCKET-AF: Post-Treatment Primary Efficacy Events Safety: Days 3 to 30 after the Last Dose Rivaroxaban Warfarin Event Rate (100 Pt-Yr) All participants 64/6843 (12. 63) Completed study medication (65%) 22/4587 (6. 42) Endpoint HR (95% CI) Pvalue 42/6807 (8. 36) 1. 51 (1. 02, 2. 23) 0. 037 6/4652 (1. 73) 3. 72 (1. 51, 9. 16) 0. 004 Excess events at end of trial with rivaroxaban

ROCKET-AF Excess Events at End of Trial with Rivaroxaban • ? Rebound hypercoagulability - Warfarin-naïve patients Rx with warfarin x 30 d = 3. 78/100 Pt-Yrs Rivaroxaban patiens transitioning to warfarin = 6. 42/100 Pt-Yrs HR = 1. 7 • ? Inadequate anticoagulation (no formal transition/bridging plan) - Both under-anticoagulation as well as over-anticoagulation with R - bleeding: 113 vs 68 related to INR>3 with Riva (HR = 1. 65)
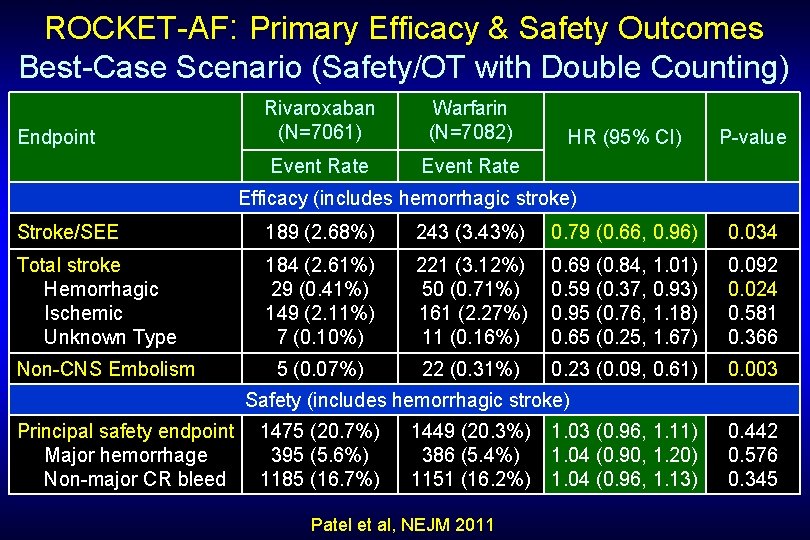
ROCKET-AF: Primary Efficacy & Safety Outcomes Best-Case Scenario (Safety/OT with Double Counting) Endpoint Rivaroxaban (N=7061) Warfarin (N=7082) Event Rate HR (95% CI) P-value Efficacy (includes hemorrhagic stroke) Stroke/SEE 189 (2. 68%) 243 (3. 43%) 0. 79 (0. 66, 0. 96) 0. 034 Total stroke Hemorrhagic Ischemic Unknown Type 184 (2. 61%) 29 (0. 41%) 149 (2. 11%) 7 (0. 10%) 221 (3. 12%) 50 (0. 71%) 161 (2. 27%) 11 (0. 16%) 0. 69 (0. 84, 1. 01) 0. 59 (0. 37, 0. 93) 0. 95 (0. 76, 1. 18) 0. 65 (0. 25, 1. 67) 0. 092 0. 024 0. 581 0. 366 5 (0. 07%) 22 (0. 31%) 0. 23 (0. 09, 0. 61) 0. 003 Non-CNS Embolism Safety (includes hemorrhagic stroke) Principal safety endpoint Major hemorrhage Non-major CR bleed 1475 (20. 7%) 395 (5. 6%) 1185 (16. 7%) 1449 (20. 3%) 386 (5. 4%) 1151 (16. 2%) Patel et al, NEJM 2011 1. 03 (0. 96, 1. 11) 1. 04 (0. 90, 1. 20) 1. 04 (0. 96, 1. 13) 0. 442 0. 576 0. 345
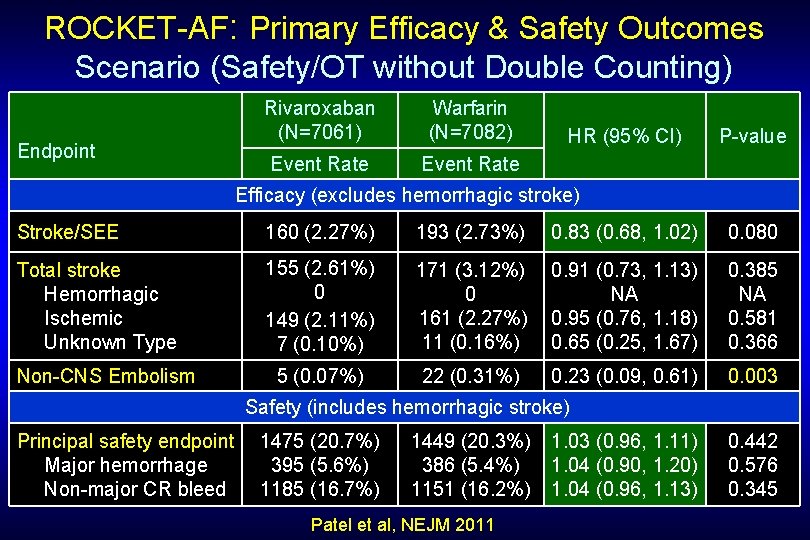
ROCKET-AF: Primary Efficacy & Safety Outcomes Scenario (Safety/OT without Double Counting) Endpoint Rivaroxaban (N=7061) Warfarin (N=7082) Event Rate HR (95% CI) P-value Efficacy (excludes hemorrhagic stroke) Stroke/SEE 160 (2. 27%) 193 (2. 73%) 0. 83 (0. 68, 1. 02) 0. 080 Total stroke Hemorrhagic Ischemic Unknown Type 155 (2. 61%) 0 149 (2. 11%) 7 (0. 10%) 171 (3. 12%) 0 161 (2. 27%) 11 (0. 16%) 0. 91 (0. 73, 1. 13) NA 0. 95 (0. 76, 1. 18) 0. 65 (0. 25, 1. 67) 0. 385 NA 0. 581 0. 366 5 (0. 07%) 22 (0. 31%) 0. 23 (0. 09, 0. 61) 0. 003 Non-CNS Embolism Safety (includes hemorrhagic stroke) Principal safety endpoint Major hemorrhage Non-major CR bleed 1475 (20. 7%) 395 (5. 6%) 1185 (16. 7%) 1449 (20. 3%) 386 (5. 4%) 1151 (16. 2%) Patel et al, NEJM 2011 1. 03 (0. 96, 1. 11) 1. 04 (0. 90, 1. 20) 1. 04 (0. 96, 1. 13) 0. 442 0. 576 0. 345
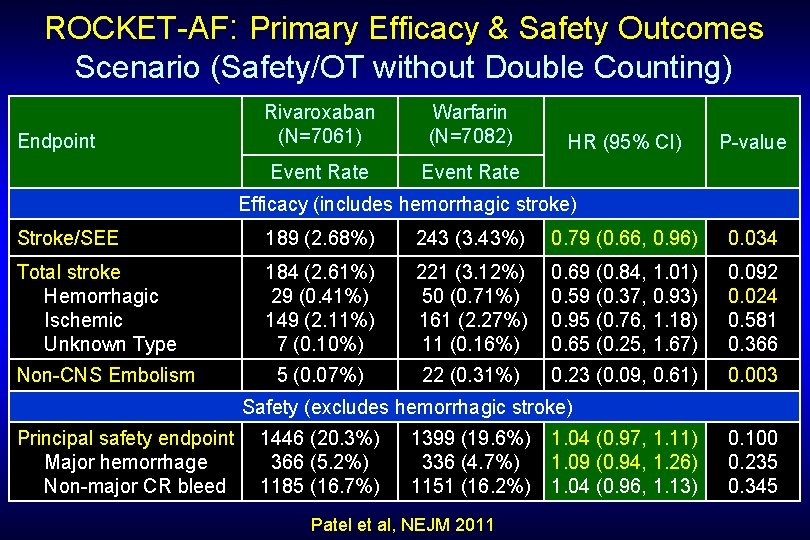
ROCKET-AF: Primary Efficacy & Safety Outcomes Scenario (Safety/OT without Double Counting) Endpoint Rivaroxaban (N=7061) Warfarin (N=7082) Event Rate HR (95% CI) P-value Efficacy (includes hemorrhagic stroke) Stroke/SEE 189 (2. 68%) 243 (3. 43%) 0. 79 (0. 66, 0. 96) 0. 034 Total stroke Hemorrhagic Ischemic Unknown Type 184 (2. 61%) 29 (0. 41%) 149 (2. 11%) 7 (0. 10%) 221 (3. 12%) 50 (0. 71%) 161 (2. 27%) 11 (0. 16%) 0. 69 (0. 84, 1. 01) 0. 59 (0. 37, 0. 93) 0. 95 (0. 76, 1. 18) 0. 65 (0. 25, 1. 67) 0. 092 0. 024 0. 581 0. 366 5 (0. 07%) 22 (0. 31%) 0. 23 (0. 09, 0. 61) 0. 003 Non-CNS Embolism Safety (excludes hemorrhagic stroke) Principal safety endpoint Major hemorrhage Non-major CR bleed 1446 (20. 3%) 366 (5. 2%) 1185 (16. 7%) 1399 (19. 6%) 336 (4. 7%) 1151 (16. 2%) Patel et al, NEJM 2011 1. 04 (0. 97, 1. 11) 1. 09 (0. 94, 1. 26) 1. 04 (0. 96, 1. 13) 0. 100 0. 235 0. 345
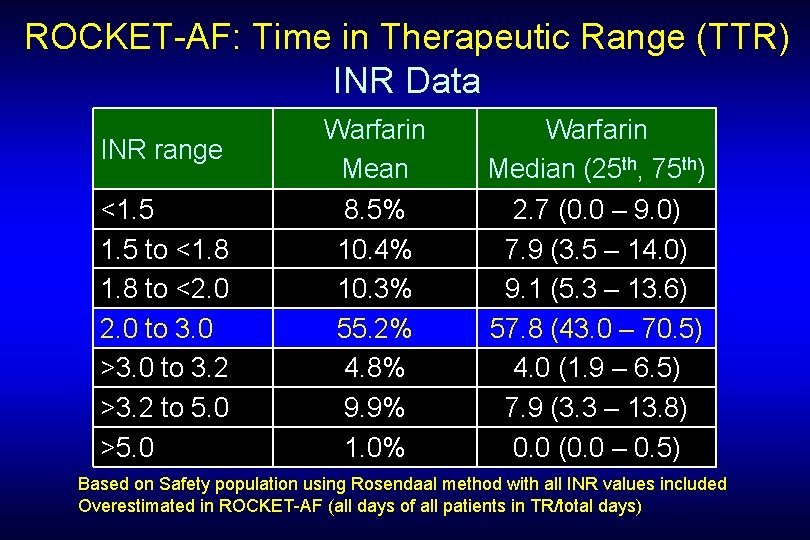
ROCKET-AF: Time in Therapeutic Range (TTR) INR Data INR range Warfarin Mean Warfarin Median (25 th, 75 th) <1. 5 to <1. 8 to <2. 0 to 3. 0 >3. 0 to 3. 2 >3. 2 to 5. 0 >5. 0 8. 5% 10. 4% 10. 3% 55. 2% 4. 8% 9. 9% 1. 0% 2. 7 (0. 0 – 9. 0) 7. 9 (3. 5 – 14. 0) 9. 1 (5. 3 – 13. 6) 57. 8 (43. 0 – 70. 5) 4. 0 (1. 9 – 6. 5) 7. 9 (3. 3 – 13. 8) 0. 0 (0. 0 – 0. 5) Based on Safety population using Rosendaal method with all INR values included Overestimated in ROCKET-AF (all days of all patients in TR/total days)
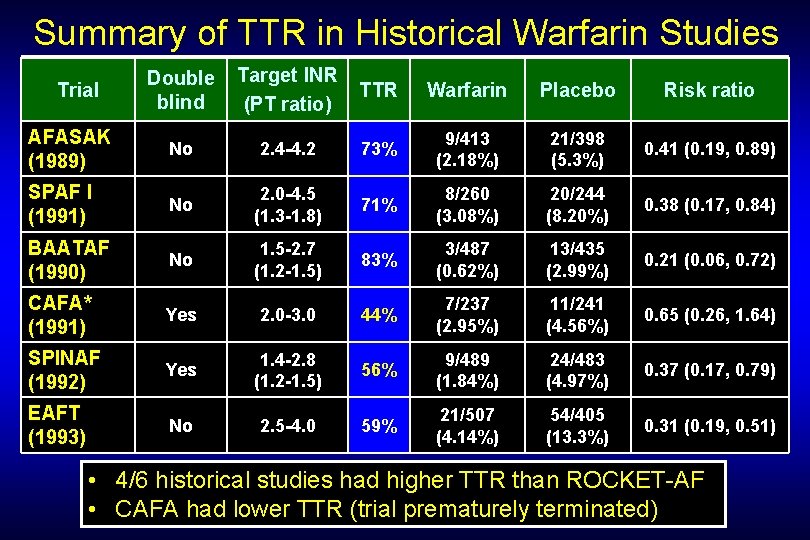
Summary of TTR in Historical Warfarin Studies Double blind Target INR (PT ratio) TTR Warfarin Placebo Risk ratio AFASAK (1989) No 2. 4 -4. 2 73% 9/413 (2. 18%) 21/398 (5. 3%) 0. 41 (0. 19, 0. 89) SPAF I (1991) No 2. 0 -4. 5 (1. 3 -1. 8) 71% 8/260 (3. 08%) 20/244 (8. 20%) 0. 38 (0. 17, 0. 84) BAATAF (1990) No 1. 5 -2. 7 (1. 2 -1. 5) 83% 3/487 (0. 62%) 13/435 (2. 99%) 0. 21 (0. 06, 0. 72) CAFA* (1991) Yes 2. 0 -3. 0 44% 7/237 (2. 95%) 11/241 (4. 56%) 0. 65 (0. 26, 1. 64) SPINAF (1992) Yes 1. 4 -2. 8 (1. 2 -1. 5) 56% 9/489 (1. 84%) 24/483 (4. 97%) 0. 37 (0. 17, 0. 79) EAFT (1993) No 2. 5 -4. 0 59% 21/507 (4. 14%) 54/405 (13. 3%) 0. 31 (0. 19, 0. 51) Trial • 4/6 historical studies had higher TTR than ROCKET-AF • CAFA had lower TTR (trial prematurely terminated)

Time in Therapeutic Range (TTR) Real-World Experience Study Setting Mean TTR Baker et al AC clinics Community-based Overall Rose et al VAHC AC clinics ROCKET-AF RCT 63% 51% 55% 48% (<6 m) 64% (>6 m) 55% Anticoagulation quality in ROCKET-AF reflective of “real-world” experience
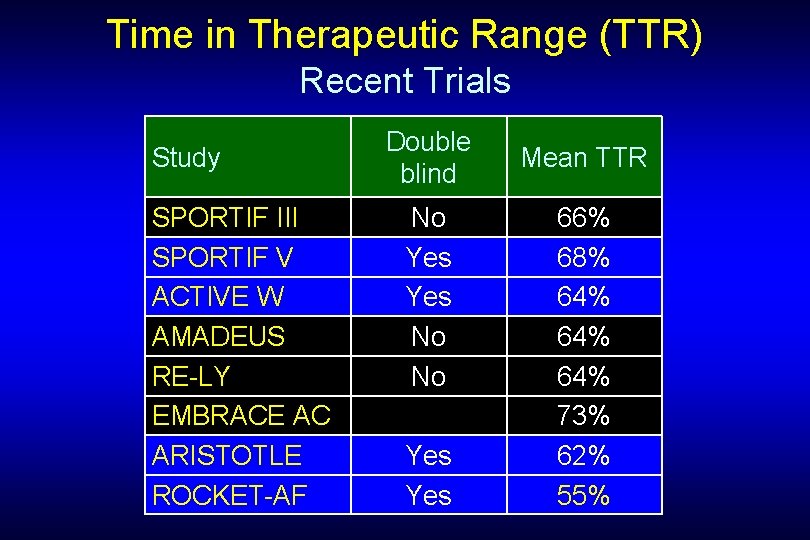
Time in Therapeutic Range (TTR) Recent Trials Study SPORTIF III SPORTIF V ACTIVE W AMADEUS RE-LY EMBRACE AC ARISTOTLE ROCKET-AF Double blind No Yes No No Yes Mean TTR 66% 68% 64% 64% 73% 62% 55%
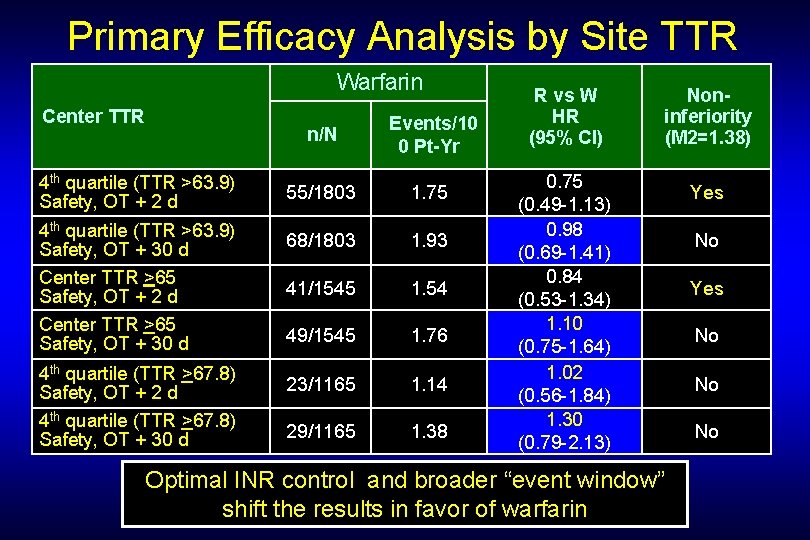
Primary Efficacy Analysis by Site TTR Warfarin Center TTR 4 th quartile (TTR >63. 9) Safety, OT + 2 d 4 th quartile (TTR >63. 9) Safety, OT + 30 d Center TTR >65 Safety, OT + 2 d Center TTR >65 Safety, OT + 30 d 4 th quartile (TTR >67. 8) Safety, OT + 2 d 4 th quartile (TTR >67. 8) Safety, OT + 30 d n/N Events/10 0 Pt-Yr 55/1803 1. 75 68/1803 1. 93 41/1545 1. 54 49/1545 1. 76 23/1165 1. 14 29/1165 1. 38 R vs W HR (95% CI) Noninferiority (M 2=1. 38) 0. 75 (0. 49 -1. 13) 0. 98 (0. 69 -1. 41) 0. 84 (0. 53 -1. 34) 1. 10 (0. 75 -1. 64) 1. 02 (0. 56 -1. 84) 1. 30 (0. 79 -2. 13) Optimal INR control and broader “event window” shift the results in favor of warfarin Yes No No No
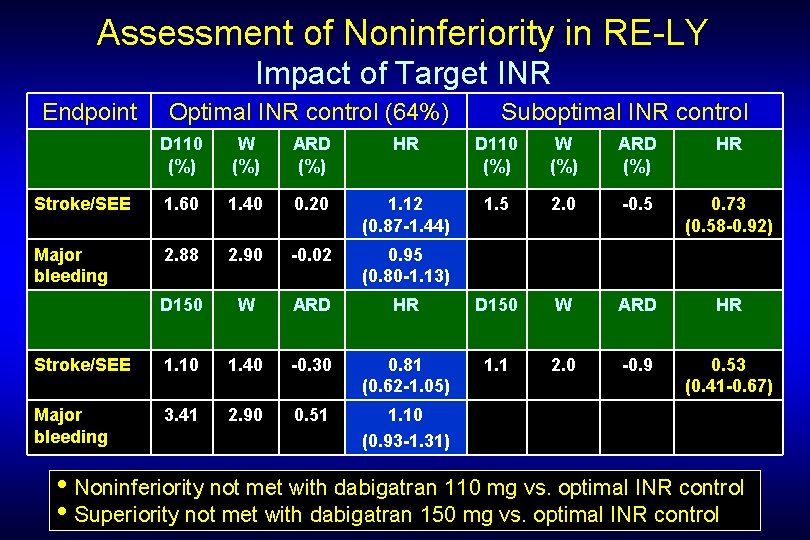
Assessment of Noninferiority in RE-LY Impact of Target INR Endpoint Optimal INR control (64%) Suboptimal INR control D 110 (%) W (%) ARD (%) HR Stroke/SEE 1. 60 1. 40 0. 20 1. 12 (0. 87 -1. 44) 1. 5 2. 0 -0. 5 0. 73 (0. 58 -0. 92) Major bleeding 2. 88 2. 90 -0. 02 0. 95 (0. 80 -1. 13) D 150 W ARD HR Stroke/SEE 1. 10 1. 40 -0. 30 0. 81 (0. 62 -1. 05) 1. 1 2. 0 -0. 9 0. 53 (0. 41 -0. 67) Major bleeding 3. 41 2. 90 0. 51 1. 10 (0. 93 -1. 31) • Noninferiority not met with dabigatran 110 mg vs. optimal INR control • Superiority not met with dabigatran 150 mg vs. optimal INR control
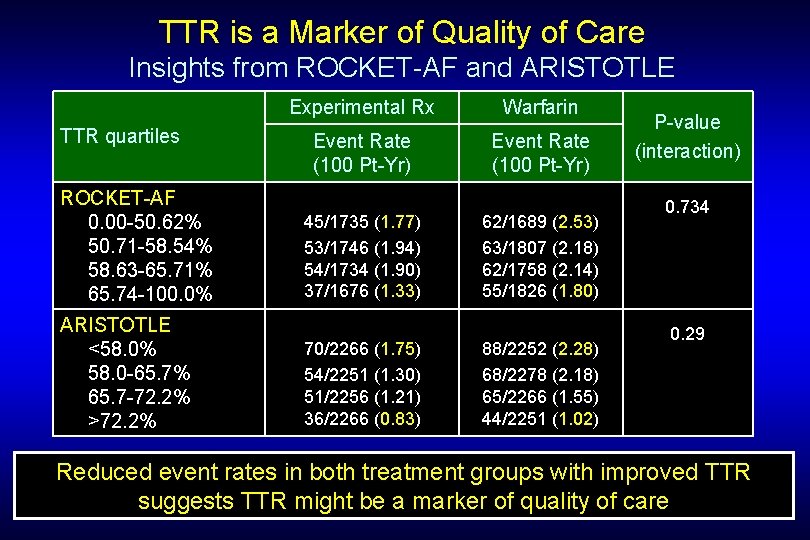
TTR is a Marker of Quality of Care Insights from ROCKET-AF and ARISTOTLE TTR quartiles Experimental Rx Warfarin Event Rate (100 Pt-Yr) ROCKET-AF 0. 00 -50. 62% 50. 71 -58. 54% 58. 63 -65. 71% 65. 74 -100. 0% 45/1735 (1. 77) 53/1746 (1. 94) 54/1734 (1. 90) 37/1676 (1. 33) 62/1689 (2. 53) 63/1807 (2. 18) 62/1758 (2. 14) 55/1826 (1. 80) ARISTOTLE <58. 0% 58. 0 -65. 7% 65. 7 -72. 2% >72. 2% 70/2266 (1. 75) 54/2251 (1. 30) 51/2256 (1. 21) 36/2266 (0. 83) 88/2252 (2. 28) 68/2278 (2. 18) 65/2266 (1. 55) 44/2251 (1. 02) P-value (interaction) 0. 734 0. 29 Reduced event rates in both treatment groups with improved TTR suggests TTR might be a marker of quality of care
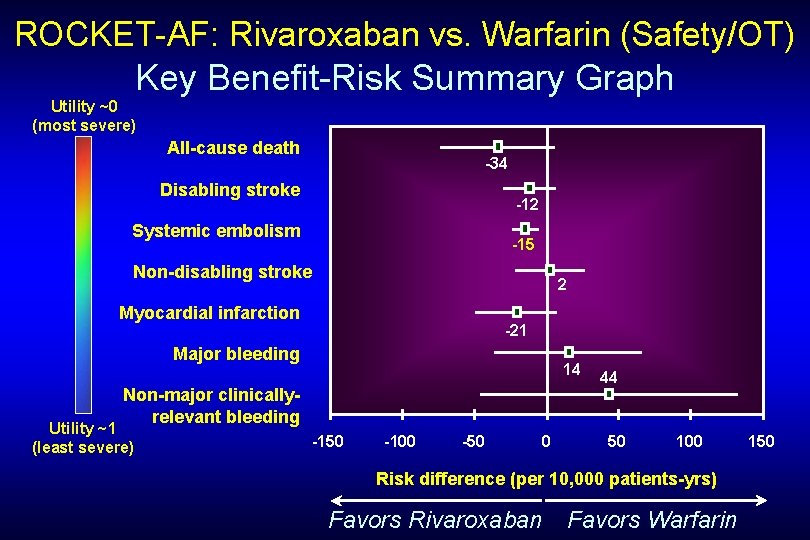
ROCKET-AF: Rivaroxaban vs. Warfarin (Safety/OT) Key Benefit-Risk Summary Graph Utility ~0 (most severe) All-cause death -34 Disabling stroke -12 Systemic embolism -15 Non-disabling stroke 2 Myocardial infarction -21 Major bleeding 14 Non-major clinicallyrelevant bleeding Utility ~1 (least severe) -150 -100 -50 0 44 50 100 Risk difference (per 10, 000 patients-yrs) Favors Rivaroxaban Favors Warfarin 150

Benefit-Risk Balance in ROCKET-AF (Safety/On Treatment) 1000 Patients Treated with Rivaroxaban instead of Warfarin Riva vs Warfarin Benefit • • 3 fewer hemorrhagic strokes 4 fewer ischemic strokes/SEE No monitoring/fixed dose Limited potential for drug/food interaction Risk • 7 excess bleeding - 3 excess blood transfusion - 5 excess Hgb drop >2 g/d. L • 6 excess SAEs leading to Rx discontinuation • Increased cost/no antidote Benefit exceeds risk, but differences are marginal (NNT>250)! What about broader event window and ITT scenario?
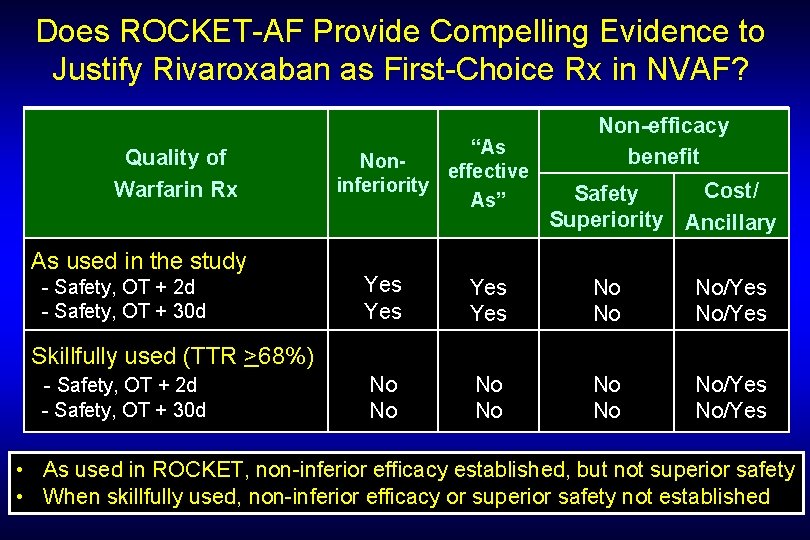
Does ROCKET-AF Provide Compelling Evidence to Justify Rivaroxaban as First-Choice Rx in NVAF? Quality of Warfarin Rx As used in the study - Safety, OT + 2 d - Safety, OT + 30 d “As Noneffective inferiority As” Non-efficacy benefit Cost/ Safety Superiority Ancillary Yes Yes No No No/Yes No No/Yes Skillfully used (TTR >68%) - Safety, OT + 2 d - Safety, OT + 30 d • As used in ROCKET, non-inferior efficacy established, but not superior safety • When skillfully used, non-inferior efficacy or superior safety not established

When Should Rivaroxaban (or Daibigatran) Not be Preferred Over Warfarin? • • • Severe renal impairment (Cr Cl <15 to 30 m. L/min) Non-adherence (dabigatran only); Non-affordability Dyspepsia, h/o GI hemorrhage (dabigatran only) Need for rapid reversibility Prosthetic heart valves Hemodynamically-significant heart valve disease Acute coronary syndromes, TIA, ischemic strokes, DVTs, PE P-gp affecting drugs (rifampim, ? quinidine); ketoconazole ? h/o MI (dabigatran only) ? dual antiplatelet therapy (only 8% took clopidogrel in RE-LY) DC Cardioversion Patients with optimal INR who value patient-doctor relationship-building monthly visits and want to know extent of Rx effect

Who Are the Ideal Candidates for Rivaroxaban? • • • History of TIA/stroke (optimal benefit-risk) Poor anticoagulant control on warfarin Patients unable, unwilling, or desiring not to undergo regular coagulation tests • • History of rare side effects from warfarin (skin necrosis) ? Increased risk of ICH (not studied in RCTs)

“The reports of my death are greatly exaggerated”

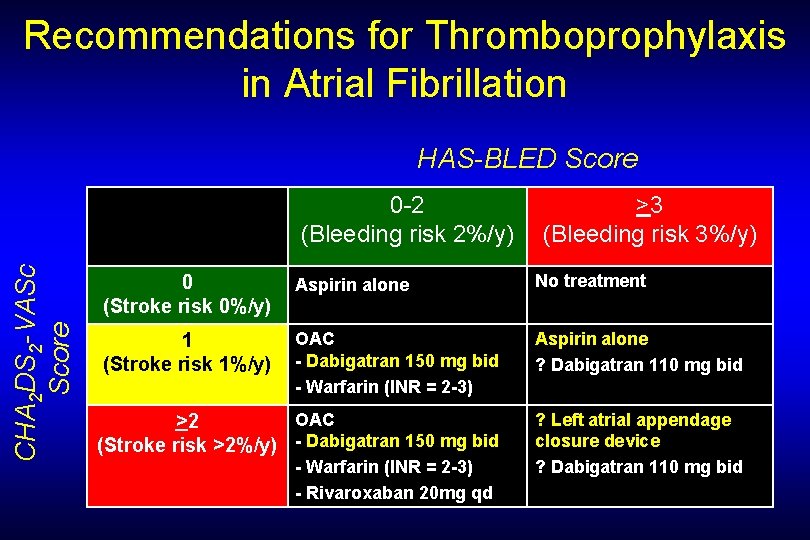
Recommendations for Thromboprophylaxis in Atrial Fibrillation HAS-BLED Score CHA 2 DS 2 -VASc Score 0 -2 (Bleeding risk 2%/y) >3 (Bleeding risk 3%/y) 0 (Stroke risk 0%/y) Aspirin alone No treatment 1 (Stroke risk 1%/y) OAC - Dabigatran 150 mg bid - Warfarin (INR = 2 -3) Aspirin alone ? Dabigatran 110 mg bid OAC >2 (Stroke risk >2%/y) - Dabigatran 150 mg bid ? Left atrial appendage closure device ? Dabigatran 110 mg bid - Warfarin (INR = 2 -3) - Rivaroxaban 20 mg qd
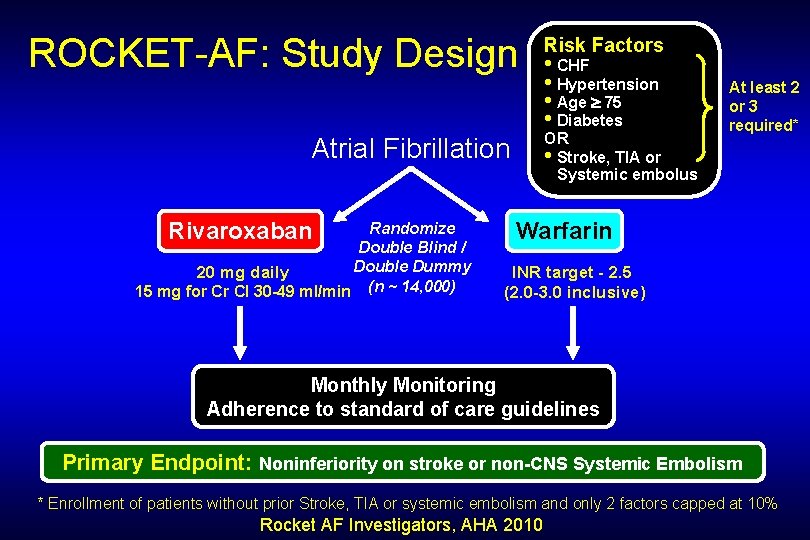
ROCKET-AF: Study Design Randomize Double Blind / Double Dummy 20 mg daily 15 mg for Cr Cl 30 -49 ml/min (n ~ 14, 000) • CHF • Hypertension • Age 75 • Diabetes OR • Stroke, TIA or Systemic embolus Atrial Fibrillation Rivaroxaban Risk Factors At least 2 or 3 required* Warfarin INR target - 2. 5 (2. 0 -3. 0 inclusive) Monthly Monitoring Adherence to standard of care guidelines Primary Endpoint: Noninferiority on stroke or non-CNS Systemic Embolism * Enrollment of patients without prior Stroke, TIA or systemic embolism and only 2 factors capped at 10% Rocket AF Investigators, AHA 2010
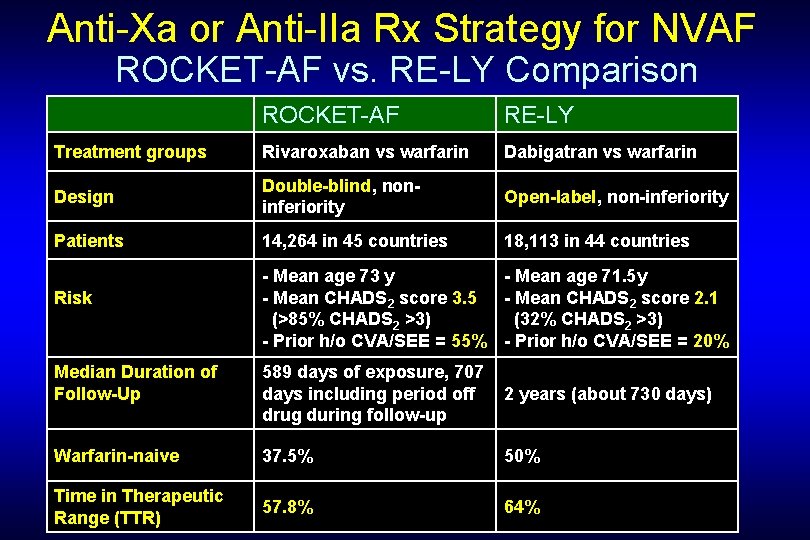
Anti-Xa or Anti-IIa Rx Strategy for NVAF ROCKET-AF vs. RE-LY Comparison ROCKET-AF RE-LY Treatment groups Rivaroxaban vs warfarin Dabigatran vs warfarin Design Double-blind, noninferiority Open-label, non-inferiority Patients 14, 264 in 45 countries 18, 113 in 44 countries Risk - Mean age 73 y - Mean age 71. 5 y - Mean CHADS 2 score 3. 5 - Mean CHADS 2 score 2. 1 (>85% CHADS 2 >3) (32% CHADS 2 >3) - Prior h/o CVA/SEE = 55% - Prior h/o CVA/SEE = 20% Median Duration of Follow-Up 589 days of exposure, 707 days including period off drug during follow-up 2 years (about 730 days) Warfarin-naive 37. 5% 50% Time in Therapeutic Range (TTR) 57. 8% 64%
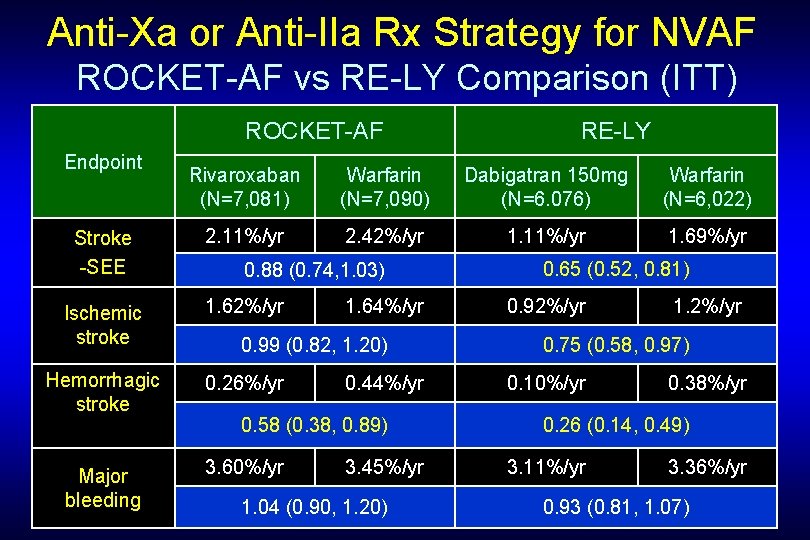
Anti-Xa or Anti-IIa Rx Strategy for NVAF ROCKET-AF vs RE-LY Comparison (ITT) ROCKET-AF Endpoint RE-LY Rivaroxaban (N=7, 081) Warfarin (N=7, 090) Dabigatran 150 mg (N=6. 076) Warfarin (N=6, 022) Stroke -SEE 2. 11%/yr 2. 42%/yr 1. 11%/yr 1. 69%/yr Ischemic stroke 1. 62%/yr Hemorrhagic stroke 0. 26%/yr Major bleeding 3. 60%/yr 0. 88 (0. 74, 1. 03) 1. 64%/yr 0. 99 (0. 82, 1. 20) 0. 44%/yr 0. 58 (0. 38, 0. 89) 3. 45%/yr 1. 04 (0. 90, 1. 20) 0. 65 (0. 52, 0. 81) 0. 92%/yr 1. 2%/yr 0. 75 (0. 58, 0. 97) 0. 10%/yr 0. 38%/yr 0. 26 (0. 14, 0. 49) 3. 11%/yr 3. 36%/yr 0. 93 (0. 81, 1. 07)
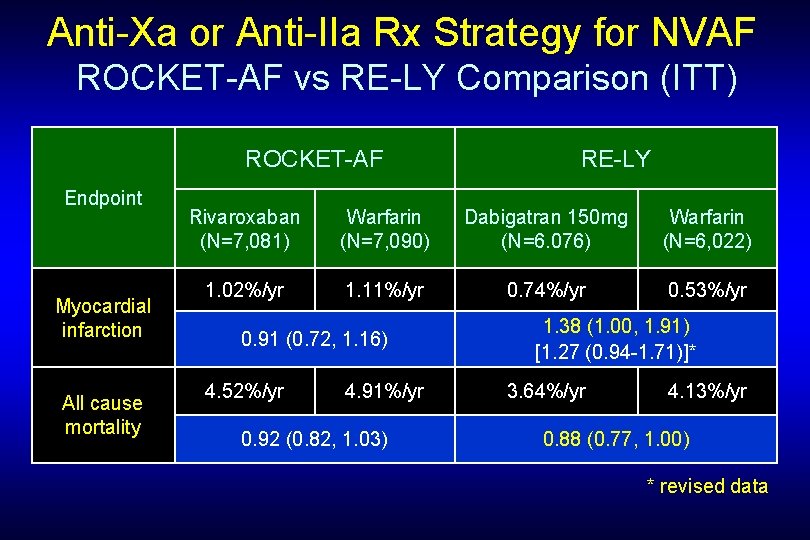
Anti-Xa or Anti-IIa Rx Strategy for NVAF ROCKET-AF vs RE-LY Comparison (ITT) ROCKET-AF Endpoint Myocardial infarction All cause mortality RE-LY Rivaroxaban (N=7, 081) Warfarin (N=7, 090) Dabigatran 150 mg (N=6. 076) Warfarin (N=6, 022) 1. 02%/yr 1. 11%/yr 0. 74%/yr 0. 53%/yr 0. 91 (0. 72, 1. 16) 4. 52%/yr 4. 91%/yr 0. 92 (0. 82, 1. 03) 1. 38 (1. 00, 1. 91) [1. 27 (0. 94 -1. 71)]* 3. 64%/yr 4. 13%/yr 0. 88 (0. 77, 1. 00) * revised data
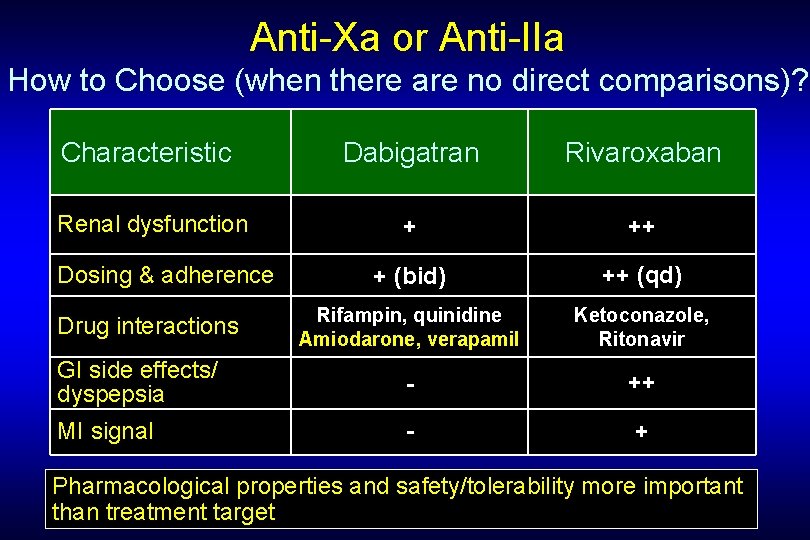
Anti-Xa or Anti-IIa How to Choose (when there are no direct comparisons)? Characteristic Dabigatran Rivaroxaban Renal dysfunction + ++ + (bid) ++ (qd) Rifampin, quinidine Amiodarone, verapamil Ketoconazole, Ritonavir GI side effects/ dyspepsia - ++ MI signal - + Dosing & adherence Drug interactions Pharmacological properties and safety/tolerability more important than treatment target
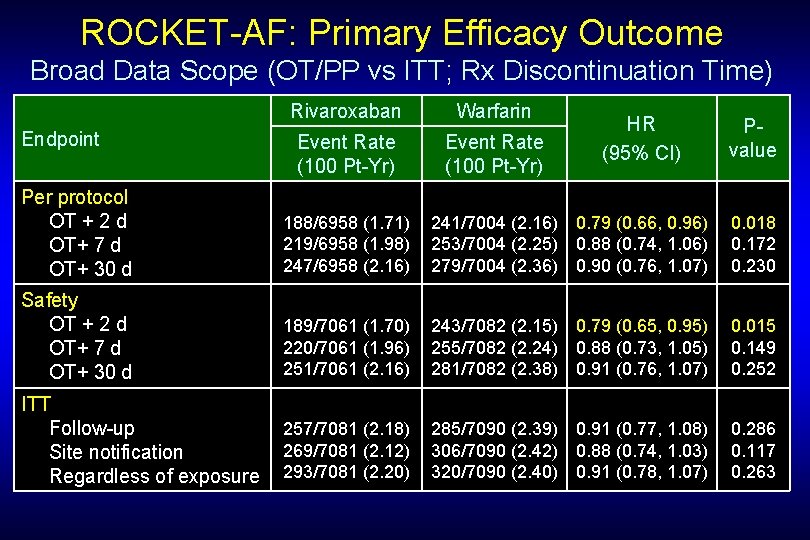
ROCKET-AF: Primary Efficacy Outcome Broad Data Scope (OT/PP vs ITT; Rx Discontinuation Time) Endpoint Rivaroxaban Warfarin Event Rate (100 Pt-Yr) HR (95% CI) Pvalue Per protocol OT + 2 d OT+ 7 d OT+ 30 d 188/6958 (1. 71) 219/6958 (1. 98) 247/6958 (2. 16) 241/7004 (2. 16) 0. 79 (0. 66, 0. 96) 253/7004 (2. 25) 0. 88 (0. 74, 1. 06) 279/7004 (2. 36) 0. 90 (0. 76, 1. 07) 0. 018 0. 172 0. 230 Safety OT + 2 d OT+ 7 d OT+ 30 d 189/7061 (1. 70) 220/7061 (1. 96) 251/7061 (2. 16) 243/7082 (2. 15) 0. 79 (0. 65, 0. 95) 255/7082 (2. 24) 0. 88 (0. 73, 1. 05) 281/7082 (2. 38) 0. 91 (0. 76, 1. 07) 0. 015 0. 149 0. 252 ITT Follow-up Site notification Regardless of exposure 257/7081 (2. 18) 269/7081 (2. 12) 293/7081 (2. 20) 285/7090 (2. 39) 0. 91 (0. 77, 1. 08) 306/7090 (2. 42) 0. 88 (0. 74, 1. 03) 320/7090 (2. 40) 0. 91 (0. 78, 1. 07) 0. 286 0. 117 0. 263
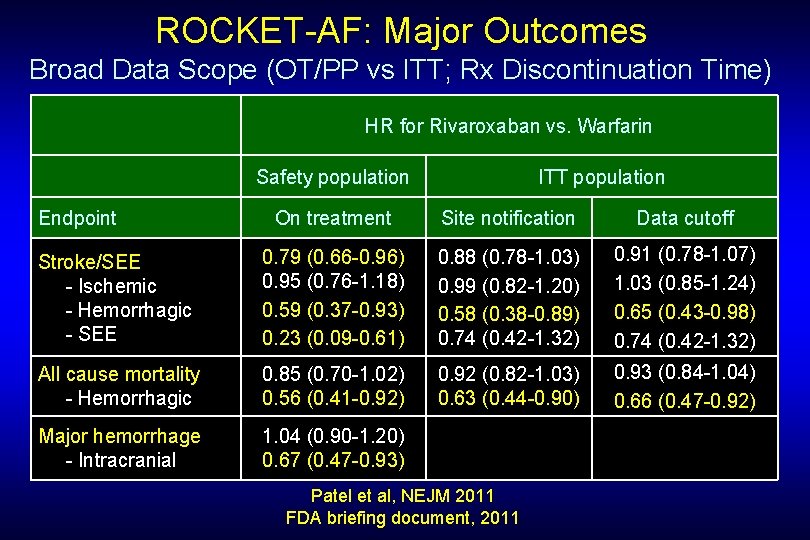
ROCKET-AF: Major Outcomes Broad Data Scope (OT/PP vs ITT; Rx Discontinuation Time) HR for Rivaroxaban vs. Warfarin Safety population Endpoint ITT population On treatment Site notification Data cutoff Stroke/SEE - Ischemic - Hemorrhagic - SEE 0. 79 (0. 66 -0. 96) 0. 95 (0. 76 -1. 18) 0. 59 (0. 37 -0. 93) 0. 23 (0. 09 -0. 61) 0. 88 (0. 78 -1. 03) 0. 99 (0. 82 -1. 20) 0. 58 (0. 38 -0. 89) 0. 74 (0. 42 -1. 32) 0. 91 (0. 78 -1. 07) 1. 03 (0. 85 -1. 24) 0. 65 (0. 43 -0. 98) 0. 74 (0. 42 -1. 32) All cause mortality - Hemorrhagic 0. 85 (0. 70 -1. 02) 0. 56 (0. 41 -0. 92) 0. 92 (0. 82 -1. 03) 0. 63 (0. 44 -0. 90) 0. 93 (0. 84 -1. 04) 0. 66 (0. 47 -0. 92) Major hemorrhage - Intracranial 1. 04 (0. 90 -1. 20) 0. 67 (0. 47 -0. 93) Patel et al, NEJM 2011 FDA briefing document, 2011
- Slides: 37