Rivaroxaban 2 5 mg BID Combined with Dual

Rivaroxaban 2. 5 mg BID Combined with Dual Antiplatelet Therapy for the Prevention of Death/MI/Stroke: A Patient Level Data Meta - Analysis of ATLAS-ACS-2 TIMI-51 and COMMANDER-HF C. Michael Gibson, M. S. , M. D. On Behalf of the ATLAS and COMMANDER Investigators Interventional Cardiologist Chief, Clinical Research, Beth Israel Deaconess CV Division Professor of Medicine Harvard Medical School President & CEO of Non-Profit Baim Institute & PERFUSE Study Group Founder, Editor-In-Chief www. wikidoc. org Harvard Medical School

Disclosure • Dr. Gibson has received research grant support and consulting fees in the past from all major manufacturers of antiplatelets and antithrombins • This is an educational lecture and is not intended to be an inducement to use any drug or drug in a fashion that is inconsistent with the drug or device label. Rivaroxaban is not approved for use in acute coronary syndromes in the US, but is so in many other countries • The slides were prepared by C. Michael Gibson, M. S. , M. D. and / or were under the editorial control of C. Michael Gibson, M. S. , M. D. The use of 2. 5 mg of Rivaroxaban is approved by the FDA for chronic coronary and/or peripheral artery disease when combined with aspirin. Rivaroxaban is not FDA approved for ACS. It is in many countries. Check your local label. Slide by C. Michael Gibson, M. S. , M. D. Gibson et al. CRT 2019
Disclosures Present Research/Grant Funding Angel Medical Corporation Bayer Corp. / Janssen CSL Behring Portola Pharmaceuticals Baim Institute (no salary) Patents and Stocks: None Equity: nference, Inc. Consultant Boston Clinical Research Institute Cardiovascular Research Foundation Eli Lilly Medtelligence Novo Nordisk Pharma. Mar Portola Bayer / Janssen/ J&J Wed. MD Spouse: Employee of Boston Clinical Research Institute, she has equity position Amarin Amgen Bayer/Janssen/ J&J Boehringer Ingelheim Boston Scientific Cardiovascular Research Foundation CSL Behring Chiesi DCRI Eli Lilly Impact Bio, LTD Gilead Sciences, Inc. The Medicines Company Medtelligence Med. Immune The Medicine’s Co. Merck & Co. Inc. Microport Novo Nordisk PERT Consortium Pharma. Mar Portola Sanofi Somahlution Verseon Corporation The use of 2. 5 mg of Rivaroxaban is approved by the FDA for chronic coronary and/or peripheral artery disease when combined with aspirin. Rivaroxaban is not FDA approved for ACS. It is in many countries. Check your local label. Slide by C. Michael Gibson, M. S. , M. D. Gibson et al. CRT 2019
Thrombus: Made of Both Platelets & Fibrin Slide by C. Michael Gibson, M. S. , M. D. Gibson et al. CRT 2019

Why Add A Factor Xa Inhibitor To DAPT? • Thrombin is most potent stimulant of platelets • Aspirin and thienopyridines do not block activation of the platelet by thrombin • Blocking Xa inhibits thrombin formation from both intrinsic and extrinsic coagulation pathways • Platelet activation diminishes over time after an ACS event • Thrombin generation in contrast is persistently elevated following ACS, is related to adverse outcomes, and is reduced by rivaroxaban Slide by C. Michael Gibson, M. S. , M. D. Gibson et al. CRT 2019
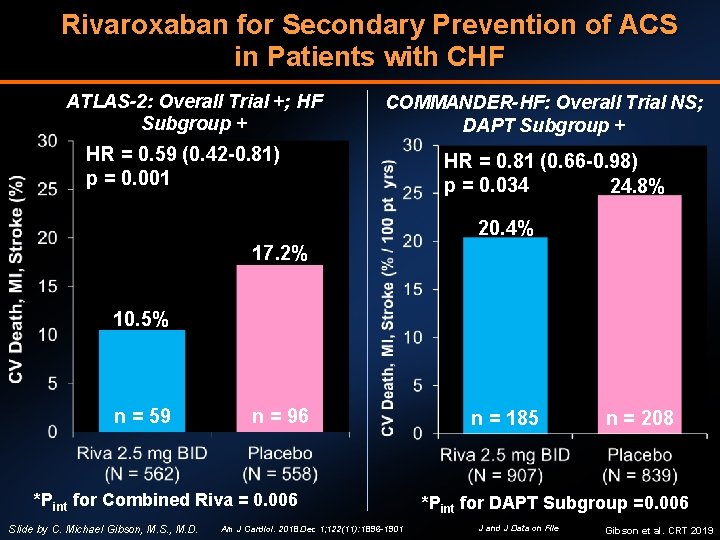
Rivaroxaban for Secondary Prevention of ACS in Patients with CHF ATLAS-2: Overall Trial +; HF Subgroup + COMMANDER-HF: Overall Trial NS; DAPT Subgroup + HR = 0. 59 (0. 42 -0. 81) p = 0. 001 HR = 0. 81 (0. 66 -0. 98) p = 0. 034 24. 8% 20. 4% 17. 2% 10. 5% n = 59 n = 96 *Pint for Combined Riva = 0. 006 Slide by C. Michael Gibson, M. S. , M. D. Am J Cardiol. 2018 Dec 1; 122(11): 1896 -1901 n = 185 n = 208 *Pint for DAPT Subgroup =0. 006 J and J Data on File Gibson et al. CRT 2019
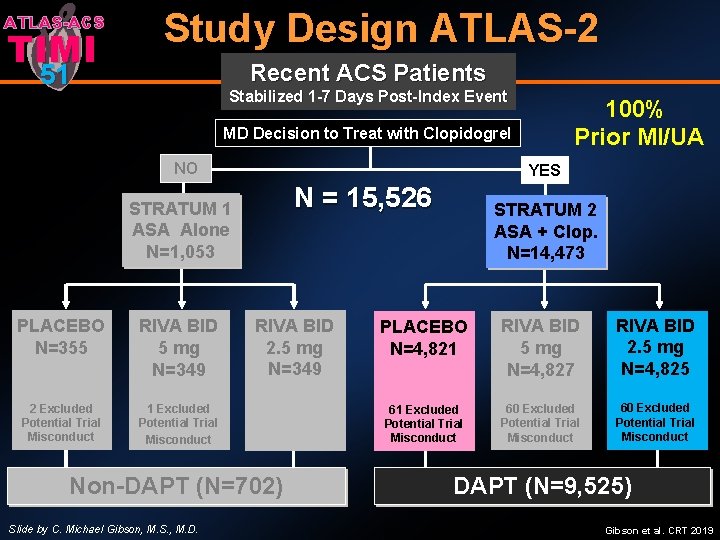
ATLAS-ACS TIMI Study Design ATLAS-2 51 Recent ACS Patients Stabilized 1 -7 Days Post-Index Event 100% Prior MI/UA MD Decision to Treat with Clopidogrel NO YES N = 15, 526 STRATUM 1 ASA Alone N=1, 053 PLACEBO N=353 N=355 5 mg (77) 10 mg (98) 2 Excluded 20 mg (78) Potential Trial Misconduct RIVA BID 5 mg N=349 RIVA BID 2. 5 mg N=349 1 Excluded Potential Trial Misconduct Non-DAPT (N=702) Slide by C. Michael Gibson, M. S. , M. D. STRATUM 2 ASA + Clop. N=14, 473 PLACEBO N=4, 821 RIVA BID 5 mg N=4, 827 RIVA BID 2. 5 mg N=4, 825 61 Excluded Potential Trial Misconduct 60 Excluded Potential Trial Misconduct DAPT (N=9, 525) Gibson et al. CRT 2019
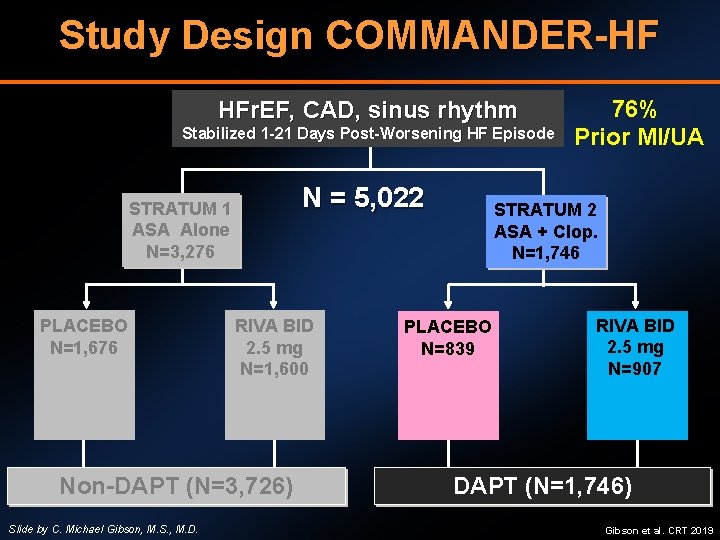
Study Design COMMANDER-HF HFr. EF, CAD, sinus rhythm Stabilized 1 -21 Days Post-Worsening HF Episode N = 5, 022 STRATUM 1 ASA Alone N=3, 276 PLACEBO N=1, 676 N=353 5 mg (77) 10 mg (98) 20 mg (78) RIVA BID 2. 5 mg N=1, 600 Non-DAPT (N=3, 726) Slide by C. Michael Gibson, M. S. , M. D. 76% Prior MI/UA STRATUM 2 ASA + Clop. N=1, 746 PLACEBO N=839 RIVA BID 2. 5 mg N=907 DAPT (N=1, 746) Gibson et al. CRT 2019
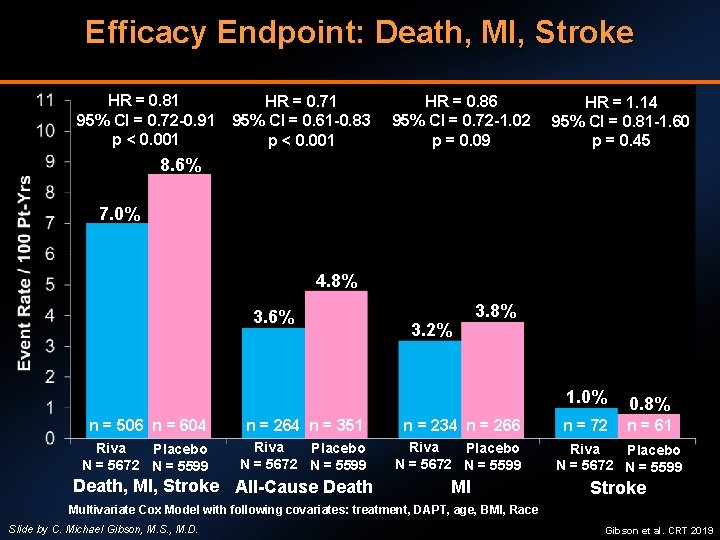
Efficacy Endpoint: Death, MI, Stroke HR = 0. 81 95% CI = 0. 72 -0. 91 p < 0. 001 HR = 0. 71 95% CI = 0. 61 -0. 83 p < 0. 001 HR = 0. 86 95% CI = 0. 72 -1. 02 p = 0. 09 HR = 1. 14 95% CI = 0. 81 -1. 60 p = 0. 45 8. 6% 7. 0% 4. 8% 3. 6% 3. 2% 3. 8% 1. 0% 0. 8% n = 72 n = 61 n = 506 n = 604 n = 264 n = 351 n = 234 n = 266 Riva Placebo N = 5672 N = 5599 MI Stroke Death, MI, Stroke All-Cause Death Multivariate Cox Model with following covariates: treatment, DAPT, age, BMI, Race Slide by C. Michael Gibson, M. S. , M. D. Gibson et al. CRT 2019
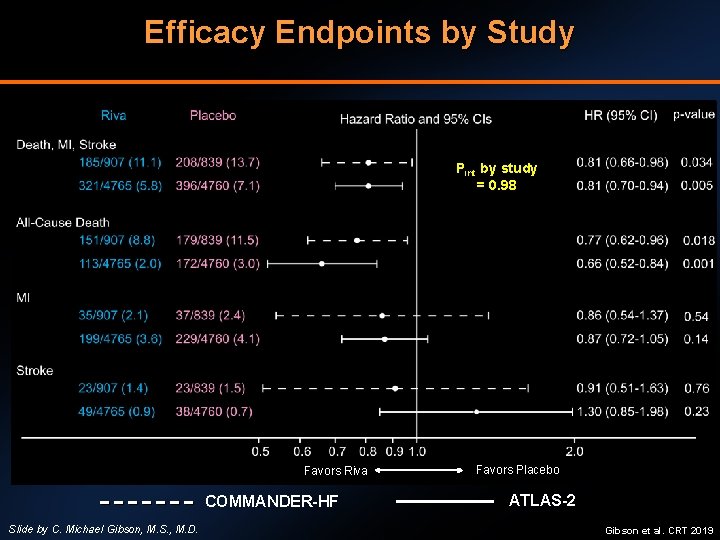
Efficacy Endpoints by Study Pint by study = 0. 98 Favors Riva COMMANDER-HF Slide by C. Michael Gibson, M. S. , M. D. Favors Placebo ATLAS-2 Gibson et al. CRT 2019
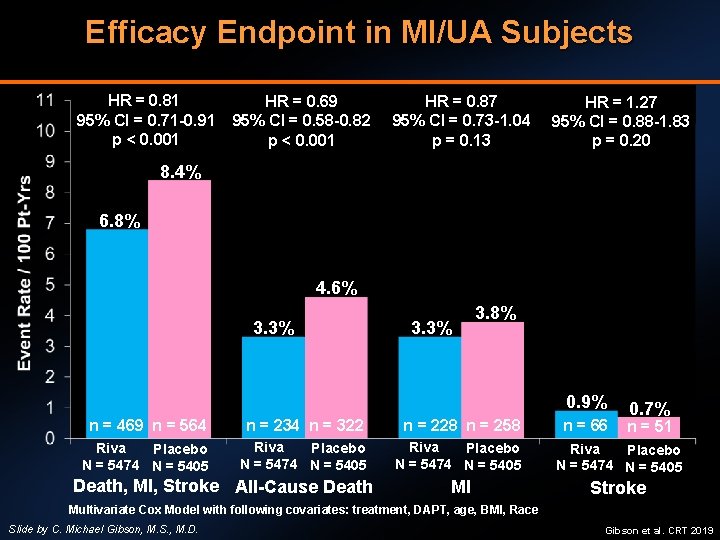
Efficacy Endpoint in MI/UA Subjects HR = 0. 81 95% CI = 0. 71 -0. 91 p < 0. 001 HR = 0. 69 95% CI = 0. 58 -0. 82 p < 0. 001 HR = 0. 87 95% CI = 0. 73 -1. 04 p = 0. 13 HR = 1. 27 95% CI = 0. 88 -1. 83 p = 0. 20 8. 4% 6. 8% 4. 6% 3. 3% 3. 8% 0. 9% n = 66 0. 7% n = 469 n = 564 n = 234 n = 322 n = 228 n = 258 Riva Placebo N = 5474 N = 5405 MI Stroke Death, MI, Stroke All-Cause Death n = 51 Multivariate Cox Model with following covariates: treatment, DAPT, age, BMI, Race Slide by C. Michael Gibson, M. S. , M. D. Gibson et al. CRT 2019
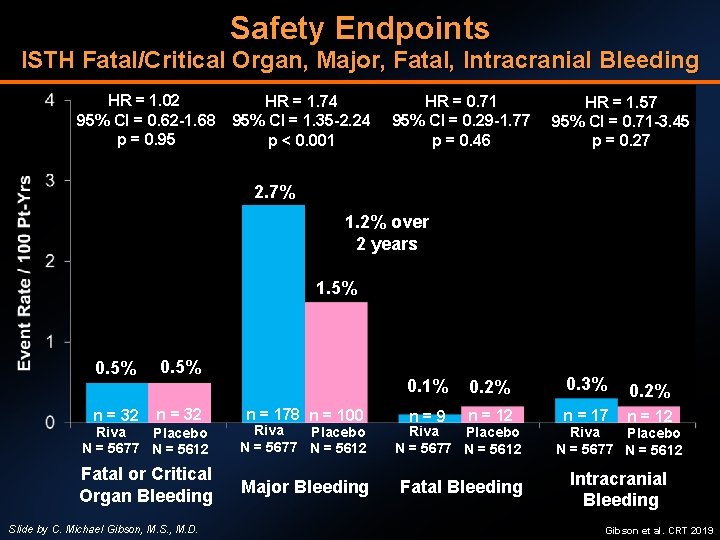
Safety Endpoints ISTH Fatal/Critical Organ, Major, Fatal, Intracranial Bleeding HR = 1. 02 95% CI = 0. 62 -1. 68 p = 0. 95 HR = 1. 74 95% CI = 1. 35 -2. 24 p < 0. 001 HR = 0. 71 95% CI = 0. 29 -1. 77 p = 0. 46 HR = 1. 57 95% CI = 0. 71 -3. 45 p = 0. 27 2. 7% 1. 2% over 2 years 1. 5% 0. 5% n = 32 n = 178 n = 100 0. 1% 0. 2% 0. 3% 0. 2% n=9 n = 12 n = 17 n = 12 Riva Placebo N = 5677 N = 5612 Fatal or Critical Organ Bleeding Major Bleeding Fatal Bleeding Intracranial Bleeding Slide by C. Michael Gibson, M. S. , M. D. Gibson et al. CRT 2019
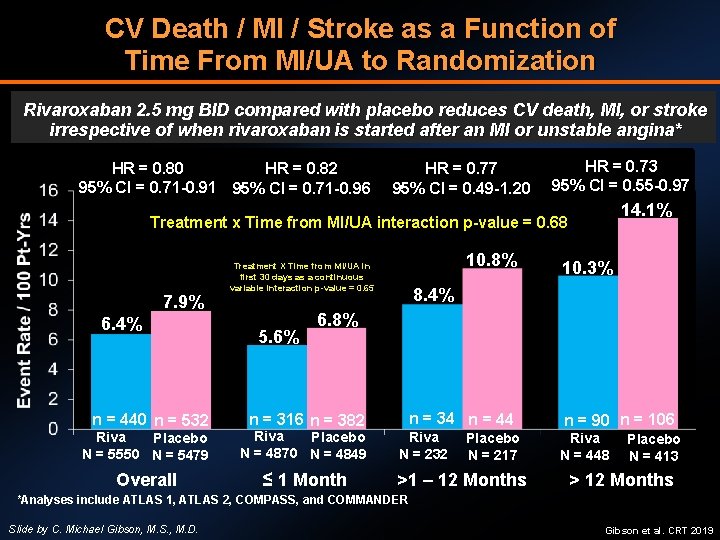
CV Death / MI / Stroke as a Function of Time From MI/UA to Randomization Rivaroxaban 2. 5 mg BID compared with placebo reduces CV death, MI, or stroke irrespective of when rivaroxaban is started after an MI or unstable angina* HR = 0. 80 HR = 0. 82 95% CI = 0. 71 -0. 91 95% CI = 0. 71 -0. 96 HR = 0. 77 95% CI = 0. 49 -1. 20 HR = 0. 73 95% CI = 0. 55 -0. 97 14. 1% Treatment x Time from MI/UA interaction p-value = 0. 68 7. 9% 6. 4% n = 440 n = 532 10. 8% Treatment X Time from MI/UA in first 30 days as a continuous variable interaction p-value = 0. 65 5. 6% 10. 3% 8. 4% 6. 8% n = 34 n = 44 n = 316 n = 382 Riva Placebo N = 5550 N = 5479 Riva Placebo N = 4870 N = 4849 Overall ≤ 1 Month Riva N = 232 Placebo N = 217 >1 – 12 Months n = 90 n = 106 Riva N = 448 Placebo N = 413 > 12 Months *Analyses include ATLAS 1, ATLAS 2, COMPASS, and COMMANDER Slide by C. Michael Gibson, M. S. , M. D. Gibson et al. CRT 2019

LIMITATIONS • Post-hoc analysis without pre-specified outcomes • Patients in COMMANDER were temporally remote from ACS, while patients in ATLAS-2 were immediate post-ACS • Exact duration of time in days from MI/UA to randomization in COMMANDER not always known (known with monthly duration) • The blended rate of MI/UA was 96%, but this was mostly driven by ATLAS-2 patients as rate of MI was 76% in COMMANDER Slide by C. Michael Gibson, M. S. , M. D. Gibson et al. CRT 2019

CONCLUSIONS • Among post ACS or CHF patients on DAPT, the addition of rivaroxaban 2. 5 mg BID reduces Death/MI/Stroke and all-cause mortality • The results were homogenous in two studies: ATLAS 2 and COMMANDER • When analysis limited to patients with history of ACS, findings hold true, irrespective of when rivaroxaban is started after MI/UA • No increase in fatal or critical organ bleeding, although increase in major bleeding over 2 years of exposure • Rivaroxaban 2. 5 mg BID is FDA-approved for chronic CAD and/or PAD • Check local label for approval in ACS population as it varies by country, not FDA approved at this time Slide by C. Michael Gibson, M. S. , M. D. Gibson et al. CRT 2019
- Slides: 15