RiskBased Decision Making for Blood Safety PT BLC

Risk-Based Decision Making for Blood Safety PT BLC – NAC Meeting Québec, April 30, 2014 Judie Leach Bennett Chair, ABO RBDM Steering Committee Director, Centre for Innovation, CBS

Overview Alliance of Blood Operators (ABO) Risk-Based Decision Making (RBDM) Project • Impetus for Change • Change Agenda • Project Structure Phase I: RBDM Framework Design (April 1, 2013 – March 31, 2014) • Risk Management Policy Foundations • Risk-based Decision-Making Process • Decision Support: Tools and Guidelines Phase II: Feasibility Testing, Consultation, Peer Review, Building Awareness, Promoting Uptake (April 1, 2014 - March 31, 2015) Request: 2 • Input into stakeholder identification and consultation process • Input into post-project implementation

Impetus for Change • Post-1980 s blood tragedies, blood operators and regulators used varying interpretations of precautionary principle to maximize blood safety • Clear that pursuit of “zero risk” is unsustainable • Blood safety decision-making is increasingly complex: science, ethics, social values, economics, public expectations, context • Risk is inherent “vein to vein”: well-being of transfusion recipients and donors must be central to blood safety decision making • 2010 Consensus Conference recommendation: a framework to – Improve decision making – Facilitate proportional responses to risk – Ensure decisions are evidence-based – Increase trust in investment decisions – Allow for redirection of resources to improve effectiveness 3

RBDM Change Agenda Current State / Future State Stakeholder Engagement Active engagement and negotiation will fragmented achieve stakeholder trust Risk communication efforts have been inconsistent, (especially of blood recipients) in risk decision-making process. but with some pockets of excellence. Risk Intelligence Systematic, transparent assessments will lead to the Based on atimely, “zeroand risk” approach, risk intervention are identification of isolation, the right priorities to proportional risk mitigation. conducted in lacking and system perspective and Comprehensive andavailability quality data, including prioritization. patient Thereoutcome is variable and use ofhemovigilance, patient will guide decision-making and help define “acceptable risk. ” outcome and quality data. Leadership Blood operators will take an expanded leadership role in vein-to-vein blood Blood advocating operators for findtimely, themselves in arisk reactive posture, facing safety, consistent, intelligent regulatory barriers toand prudent and proportionate risk-taking. processes, pursuing innovation and continuous improvement focused on reducing risks and improving outcomes. Effectiveness and Cost Reliable information and toolswith will be readilyinformation available to balance System currently operates limited at timesrisks, and costs few and benefits in a manner which donor safetycomparisons. and patient with cost benefit analyses andoptimizes cost effectiveness outcomes. Oversight and Accountability Oversight will facilitate uptake and coordination of risk decision-making In the absence of unified oversight and clear accountability, there is processes and will foster comparison of and accountability for donor safety a lack of system-wide coherence in risk management decisions. 4 and patient outcomes under various systems.
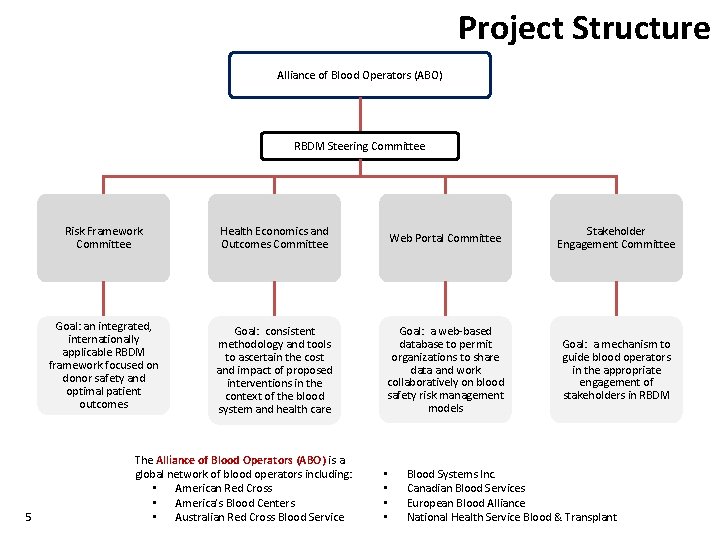
Project Structure Alliance of Blood Operators (ABO) RBDM Steering Committee 5 Risk Framework Committee Health Economics and Outcomes Committee Web Portal Committee Stakeholder Engagement Committee Goal: an integrated, internationally applicable RBDM framework focused on donor safety and optimal patient outcomes Goal: consistent methodology and tools to ascertain the cost and impact of proposed interventions in the context of the blood system and health care Goal: a web-based database to permit organizations to share data and work collaboratively on blood safety risk management models Goal: a mechanism to guide blood operators in the appropriate engagement of stakeholders in RBDM The Alliance of Blood Operators (ABO) is a global network of blood operators including: • American Red Cross • America’s Blood Centers • Australian Red Cross Blood Service • • Blood Systems Inc. Canadian Blood Services European Blood Alliance National Health Service Blood & Transplant

PHASE I: Risk-Based Decision Making Framework Design “To develop an integrated , internationally applicable framework, entrenched in donor safety and optimal patient outcomes, to guide major policy and operational change. ” 6

Risk-Based Decision Making Framework for Blood Safety 1. Issue Identification & Problem Formulation DRAFT Purpose: A framework for considering all relevant factors in decisions on blood safety Scope and application: Created for application by developed blood operatorsthe safety of the blood Objective 1: To optimize Risk-Based 4. 2. Communication Decisionsupply by enabling the proportional allocation of Assessment & Stakeholder Objective 2: To. Decision analyze and account for a series Making Engagement Process finite resources to mitigate the most serious of contextual, often qualitative, factors that risks, affect recognizing that the elimination of all of risk is not decision making in the management blood 3. possible Evaluation risks: social, economic, and ethical perspectives, which go beyond quantitative calculations of risk Risk Tolerability and which can alter risk tolerability Risk Expectations for Conduct of Assessments Management Policy Foundations Risk Communication & Stakeholder Engagement Risk Management Principles

Risk Management Policy Foundations What are Policy Foundations? • Stable, background elements that express the values and risk management priorities and policies of an organization, and inform the process of risk-informed decision making • Foundation for risk decision-making process, ideally available to stakeholders and the public in support of transparency • One way to establish these foundations - an overarching, organization-specific Risk Management Policy Risk Tolerability Expectations for Conduct of Assessments Risk Communication & Stakeholder Engagement 8 Risk Management Principles

Risk Management Policy Foundations Content highlights: • Risk management principles: Beneficence, Fairness, Transparency, Consultation, Practicality and Proportionality, Vigilance and Continuous Improvement • Content developed for Risk Communication & Stakeholder Engagement policies, connected to guidance documents • General expectations for evidence-based assessments, with specific recommendations for Health Risk Assessments and Health Economics & Outcomes Assessments • Risk tolerability concepts and schematic Risk Tolerability Expectations for Conduct of Assessments Risk Communication & Stakeholder Engagement 9 Risk Management Principles

Risk-Based Decision Making Process What is the RBDM process? • Four stages are intuitive and are common to many decision frameworks • Designed to ensure that all relevant information is considered in a systematic manner • Each steps describes its purpose, the decision support tools provided, and the expected outputs • Not always linear, may be iterative, involving a return to a previous step if more information is required, or if 10 new information 1. Issue Identification 4. Decision Risk Comms & Stakeholder Engagement 3. Evaluation 2. Assessment

Risk-Based Decision Making Process Content highlights: • Focus on practical templates and tools to support the decision process • “How to” guidance documents to guide the conduct of core assessments in Step 2 • Provision for the conduct of the other assessments such as feasibility, supply impact, operational impact, human resource demands • Decision tree for risk tolerability 11 questions 1. Issue Identification 4. Decision Risk Comms & Stakeholder Engagement 3. Evaluation 2. Assessment

Risk-Based Decision Making Process Content highlights: • Focus on practicality and proportionality in the allocation of effort and resources to assess and control risk • Process is scalable to be conducted with different degrees of effort and detail depending on the magnitude and tolerability of the risk in question (e. g. initial screening vs. comprehensive assessments) 12 1. Issue Identification 4. Decision Risk Comms & Stakeholder Engagement 3. Evaluation 2. Assessment

Decision Support Tools Decision support tools and guidance documents in framework appendices: Appendix A: Glossary Appendix B: Decision Support Tools* Appendix C: Guidance on Risk Communication Appendix D: Guidance on Stakeholder Engagement* Appendix E: Guidance on Health Risk Assessment Appendix F: Guidance on Conducting Health Economics & Outcomes Assessments* • Appendix G: Guidance on Conducting Contextual Assessments* • Appendix H: Complete Case Study • • • Goals: Uniquely tailored to blood safety, added value, integrated, common lexicon, skill-building 13

Decision Support Tools (Appendix B) Templates, Checklists, Decision Tree 14 14

Guidance on Stakeholder Engagement (Appendix D) 15 15

Guidance on Conducting Health Economics & Outcomes Assessments (Appendix F) 16 16

Guidance on Conducting Contextual Assessments (Appendix G) Social concern and risk perception • Studies the level of awareness and concern about an issue in society, often indicated by elevated media attention to the issue, and the tone of public discussion of the issue Equity • Evaluates concerns with the equity of risk distribution, or the impacts of a risk management measure Trust • Evaluates impact on trust in the management of blood safety, and in the blood operator Jurisdictional differences • Lessons from other jurisdictions • May involve concerns stemming from another jurisdiction adopting different risk control expectations or different risk control measures Legal • May involve specific considerations such as the potential for liability 17 17

PHASE II: Feasibility Testing, Consultation, Peer Review, Building Awareness, Promoting Uptake 18

Feasibility Testing & Peer Review • Scenario-based exercises testing all aspects of framework (webinar and face -to-face) • Cost-utility assessment of widely adopted interventions (data collection challenges) • Evaluation of health economics tools in novel threat situation • Peer review: external evaluation by risk experts early in Phase II and formal peer review late in Phase II • Input with which to refine 19 framework and tools Sample Review Questions Ø Are the framework and tools usable and practical? Ø Are they scalable for both complex and simple risk issues? Ø Do the guidance documents strike the right balance, e. g. technical content versus “how-to” guidelines? Are they understandable / accessible? Ø How much expertise must a user have in the various disciplines described in the framework? Ø Do the framework and tools support a logical process and lead to a decision? Ø Do they help determine priorities for allocation of resources?

Stakeholder Engagement & Consultation E-consultation • Using an electronic consultation methodology called Choicebook • A database of stakeholders identified on Phase I will participate in the Choicebook consultation June 24: Choicebook launch August 12: Final report Face-to-Face consultation June 10: PLUS (Platform of Plasma Protein Users) December 2013 to October, 2014: Speakers’ Bureau Schedule Regulator consultation • One-on-one meetings with country regulators (consultation package developed) • Articulating our unique contribution to regulatory dialogue • Blood Regulators Network 20

Presentation Schedule 21 Event Advisory Committee on Blood and Tissue Safety and Availability AABB Board Quebec Ministry of Health Biovigilance Committee ARC Holland Lab EBA Board meeting Date/time December 2013 January 14, 2014 January 30, 2014 March 2014 European Commission - National Competent Authorities April 10, 2014 April 11 -13, 2014 April 27 - 28, 2014 May 1 -4, 2014 May 20, 2014 May 31 -June 5, 2014 June 10, 2014 June 26, 2014 August 6 -7, 2014 September 3 -5, 2014 September 23 -24, ‘ 14 October 25 -28, 2014 ABO Member meetings AABB National Blood Foundation Leadership Forum CSTM Annual Conference National Blood Authority ISBT International Congress PLUS Meeting Comité consultatif national de médecine transfusionnelle ABC, Interim Meeting European Conference on Donor Health and Management International Plasma Fractionators Association AABB Annual Meeting Education Session AABB Annual Meeting ABO booth April 10, 2014

Industry Newsletters • ABC Newsletter • EBA Newsletter • AABB News Communications Quarterly Communiqués • Sent to database of stakeholders ABO website [under development] • Risk portal Qs and As • 22 Ongoing development Contact: judie. leach. bennett@blood. ca steph. kelly@blood. ca

Publication Strategy 23 23

Next Steps PT / NAC Input • Stakeholder consultation and peer review Looking ahead… Post-Project Implementation (April 1, 2015) • Facilitating uptake, coordination, risk data sharing • Continuous improvement of framework and tools • Monitoring impacts • Integrating risk approaches with other system players 24
- Slides: 24