Risk Assessment for Air Toxics The 4 Basic

Risk Assessment for Air Toxics: The 4 Basic Steps NESCAUM Health Effects Workshop Bordentown, NJ July 30, 2008 1

The Air Toxics Risk Assessment Process 2

3

The General Four Step Risk Assessment Process Risk Assessment 1. Exposure Assessment Risk Management Statutory and legal Considerations Public Health Considerations 4. Risk Characterization Toxicity Assessment 2. Hazard ID 3. Dose-Response Assessment Social Factors Risk Management Decision Risk Management Options Economic Factors Political Considerations 4

The General Air Toxics Risk Assessment Process Planning and Scoping Exposure Assessment Toxicity Assessment Is a chemical toxic? Who is exposed? What chemicals are they exposed to? How does the exposure occur? What is the relationship between the dose of a chemical and the response that results? Risk Characterization What is the likelihood that the exposure will result in an adverse health effect? How sure are we our answers are correct? 5
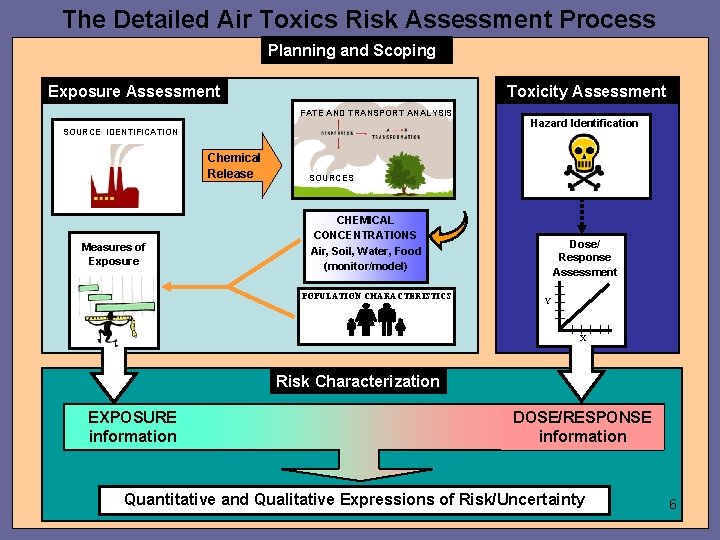
The Detailed Air Toxics Risk Assessment Process Planning and Scoping Exposure Assessment Toxicity Assessment FATE AND TRANSPORT ANALYSIS SOURCE IDENTIFICATION Chemical Release Measures of Exposure Hazard Identification SOURCES CHEMICAL CONCENTRATIONS Air, Soil, Water, Food (monitor/model) POPULATION CHARACTERISTICS Dose/ Response Assessment Y X Risk Characterization EXPOSURE information DOSE/RESPONSE information Quantitative and Qualitative Expressions of Risk/Uncertainty 6

Toxicity Assessment Focus Planning and Scoping Exposure Assessment Toxicity Assessment Hazard Identification Dose/ Response Assessment Y X Risk Characterization 7 7

Step A - Hazard Identification • Is exposure to a chemical causally linked to particular health effects? • Is the effect of practical significance? • What is the nature and strength of the evidence of causation? 8

Hazard ID can consider both…. Biological Effects • Lethality (LD 50, LC 50) • Impairment of normal biological function (e. g. , liver damage) • Heritable genetic change • Increases/decreases in species population size or range • Health/productivity of ecosystems • Etc. Non-Biological Effects • • • Reduced visibility from airborne particulates Damage to historic structures by air pollutants Climate change from global warming 9

Numerous Biologic Endpoints Cancer Non-Cancer Multiple Adverse Endpoints • Mutations • DNA damage • Etc. Uncontrolled Growth of Cells Interaction with HAP • Reproductive, developme ntal, neurological disorders • Immunologic effects • Acute effects (edema, CNS depression) • Various other systemic effects (e. g. , liver, kidney, lung damage) NASA Graphic 10

Hazard Identification Where do we get our information? Data on adverse biologic effects usually generated through… • Epidemiological studies • In-vitro assays (i. e. , test tube) Human Epidemiological Studies • In vivo biologic assays • Structure-activity relationships (SAR) Laboratory Animal Experiments 11

Step B – Dose/Response Assessment • Now that we’ve established that a chemical is toxic… • We need to understand how much dose gives how much response (how potent is the chemical? ) • Some of our Hazard ID information may help us answer that question 12
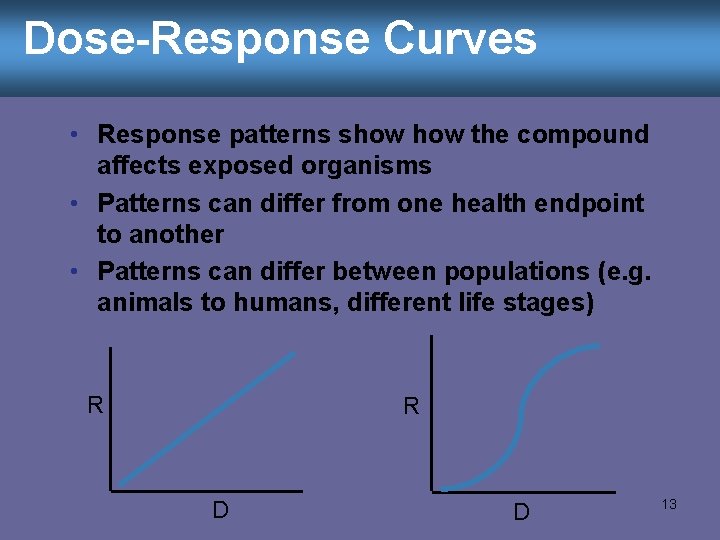
Dose-Response Curves • Response patterns show the compound affects exposed organisms • Patterns can differ from one health endpoint to another • Patterns can differ between populations (e. g. animals to humans, different life stages) R R D D 13

Dose-Response - Noncancer EPA assumes… • There is a threshold below which no observable adverse effect will occur (there is a toxicity threshold) • We usually don’t know exactly where that point is from our lab animal data, so… • We use our animal data, in conjunction with a series of uncertainty factors, to estimate a “safe” or “reference” exposure for humans 14
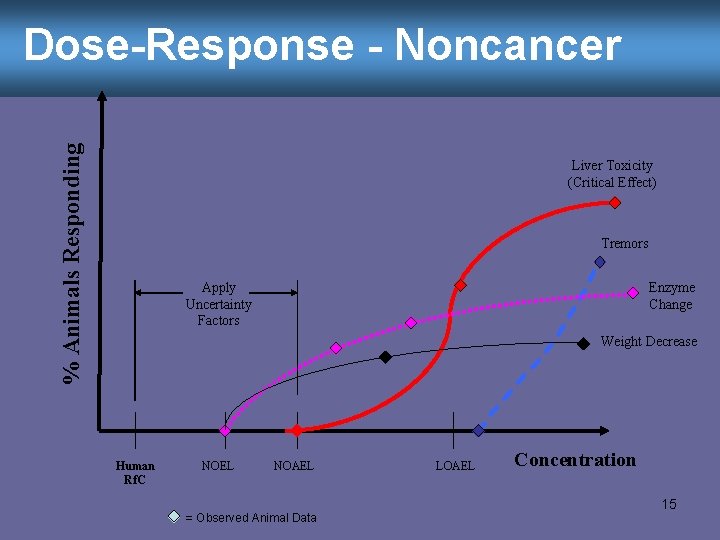
% Animals Responding Dose-Response - Noncancer Liver Toxicity (Critical Effect) Tremors Apply Uncertainty Factors Enzyme Change Weight Decrease Human Rf. C NOEL NOAEL = Observed Animal Data LOAEL Concentration 15

Inhalation RFC Uncertainty Factor Criteria • Extrapolating from animal data • Sensitive human populations • Subchronic NOAEL instead of chronic NOAEL • LOAEL used instead of NOAEL • Uncertainties in the database for the chemical UF 10, 3, or 1 10, 3, or 1 *HEC = human equivalent concentration **Rf. D (Oral) calculated similarly (usually in mg/kg-d) ***Some Rf. Cs developed in the past may have employed a modifying factor (MF) to account for overall quality of the tox database 16

Dose-Response - Cancer Unless we have data that indicates otherwise, we assume … • There is no exposure which is without cancer risk (a non-threshold response) • Even very low doses are not risk free • We know the lowest exposure from our lab animal experiments, but how do we extrapolate to the very low concentrations people are more likely to experience? 17
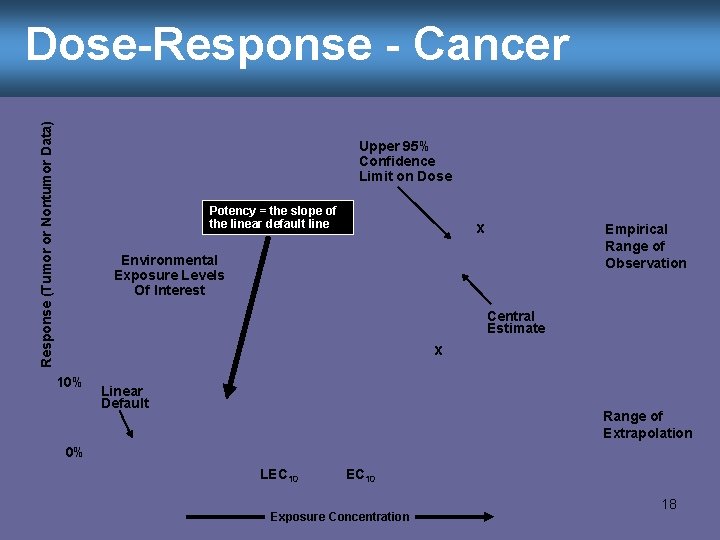
Response (Tumor or Nontumor Data) Dose-Response - Cancer Upper 95% Confidence Limit on Dose Potency = the slope of the linear default line x Empirical Range of Observation Environmental Exposure Levels Of Interest Central Estimate x 10% Linear Default Range of Extrapolation 0% LEC 10 Exposure Concentration 18

Inhalation Unit Risk IUR (risk per ug/m 3) = Slope of the line from the point of departure to zero • The IUR is the unitless upper bound estimate of the probability of tumor formation per unit concentration of chemical • Measures of potency for ingestion are developed in a similar fashion, but are… • In units of (risk per mg/kg-d) 19

In Summary … Cancer Risk • Nonthreshold (generally) • Slope Factors Non-Cancer Hazard • Threshold (generally) • Reference Values • Inhalation Unit Risk • Oral Potency Factor • Rf. C (inhalation) • Rf. D (oral) 20

Sources of Toxicity Data There are many choices • • EPA IRIS database California Hotspots program ATSDR MRLs NCEA provisional values EPA HEAST Open literature Etc. Which value should we use? 21

Sources of Toxicity Data For air toxics risk assessments… OAQPS has developed and maintains a list of recommended chronic toxicity values for each of the HAPs • Inhalation IURs and Rf. Cs • Oral slope factors and Rf. Ds http: //www. epa. gov/ttn/atw/toxsource/summary. html 22

23
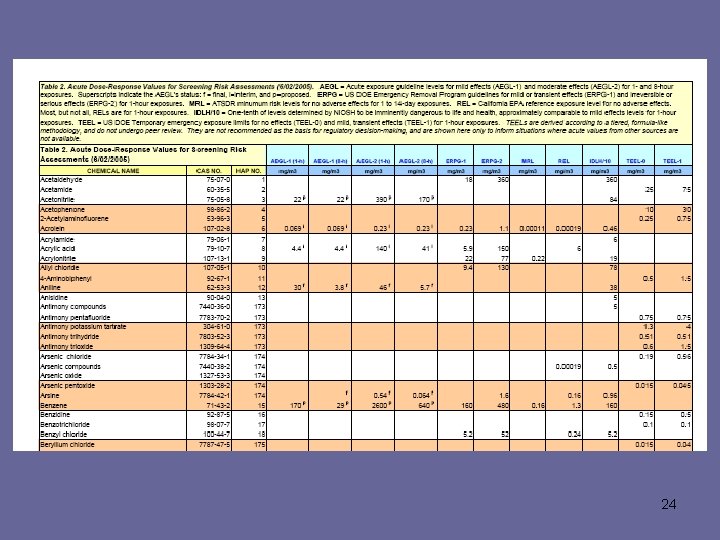
24

The General Risk Assessment Process Let’s restate this 4 -step process with a simple mathematical formula… RISK = f [(Measure of Exposure), (Measure of Toxicity)] 25

The Risk Assessment Continuum: Tiered Approaches to the Process Complete study-specific data, no assumptions; higher cost, lower uncertainty Add uncertainty/variability analysis D E IN E R O F E R More refined exposure assessment M More refined dispersion & exposure modeling N NI E E R G Simple dispersion model SC Lookup Table No data, all assumptions; lower cost, high uncertainty 26
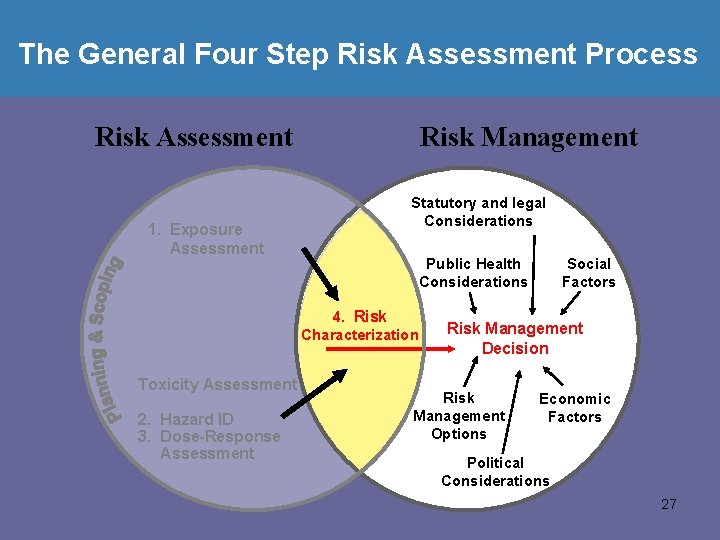
The General Four Step Risk Assessment Process Risk Assessment 1. Exposure Assessment Risk Management Statutory and legal Considerations Public Health Considerations 4. Risk Characterization Toxicity Assessment 2. Hazard ID 3. Dose-Response Assessment Social Factors Risk Management Decision Risk Management Options Economic Factors Political Considerations 27

Questions? • Contact Heidi Hales VT DEC/NESCAUM heidi. hales@state. vt. us 802 -241 -3848 28
- Slides: 28