RING OPENING POLYMERIZATION A SMART WAY TO IMPROVE










- Slides: 18

RING OPENING POLYMERIZATION - A SMART WAY TO IMPROVE CONDENSATION POLYMERIZATION Speaker: Dr. Lu, Chu-Hua Chang’s Lab 10/26/2009 Reference: Ring-Opening Polymerization Mechanisms, Catalysts, Structure, Utility (1993), edited by Daniel J. Brunelle 1

Outline ROP: ring opening polymerization Importance of ROP Nylon (polyamide) (Cyclic amide monomers) Character of ROP Monomer 1: Oxirane Monomer 2: Acetal Monomer 3: Cyclic ester Monomer 4: phosphazenes Monomer 5: siloxane Summary Conventional Radical Polymerization 2

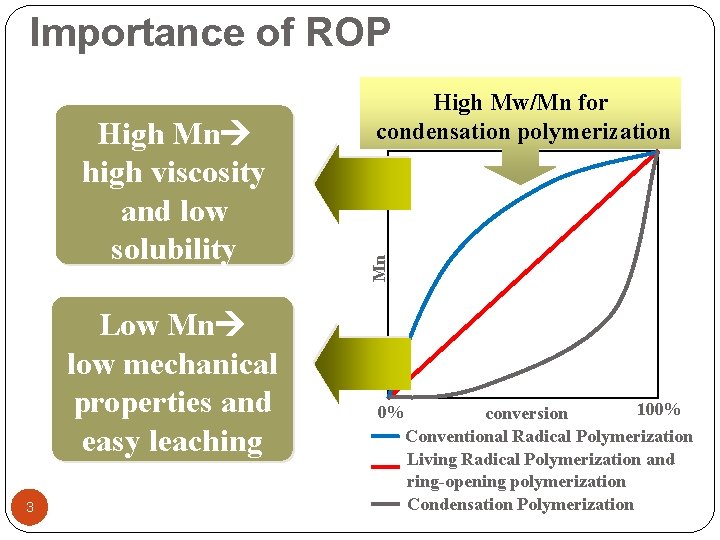
Importance of ROP Low Mn low mechanical properties and easy leaching 3 Mn High Mn high viscosity and low solubility High Mw/Mn for condensation polymerization 100% 0% conversion Conventional Radical Polymerization Living Radical Polymerization and ring-opening polymerization Condensation Polymerization

Importance of ROP Condensation Polymerization of Nylon 6, 6 ROP of Nylon 66 Bag 4 Nylon 6 Yard Pictures from wikipedia. org Nylon 66 Bed


Characteristics of ROP Using cyclic monomers Using “ionic” imitator R: alkyl chain; Z: O, S, N, P, Si, or -CONH-, -COO-,-CH=CH-, and so on. Polymerization: transformation from intra-chain bonds to inter-chain bonds and enthalpy from the ring strain of cyclic monomers. Polymerization without releasing small molecules (cf. condensation polymerization). Polymerization with low activation energy (cf. covalent bond for condensation polymerization). Some cyclic monomers are prepared by chemical modification of α , ω –difunctional monomers for condensation polymerization. Living polymerization (pseudo ionic polymerization): controllable and high molecular-mass polymers (cf. condensation polymerization (equivalent feeding ratios)). 6

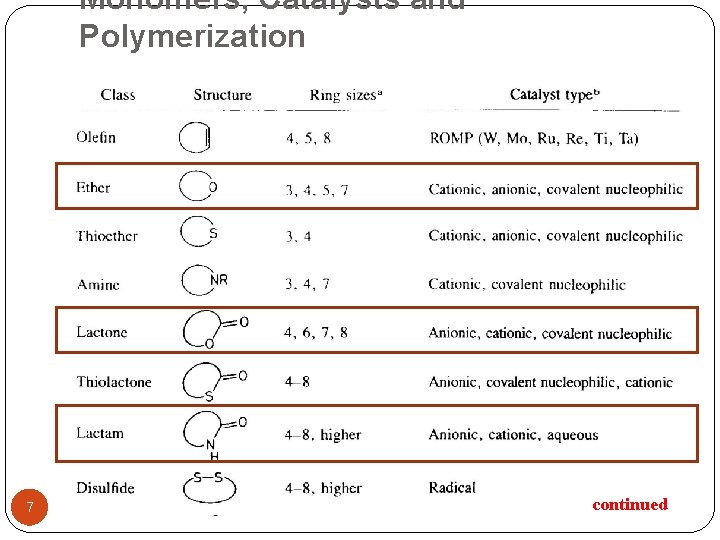
Monomers, Catalysts and Polymerization 7 continued

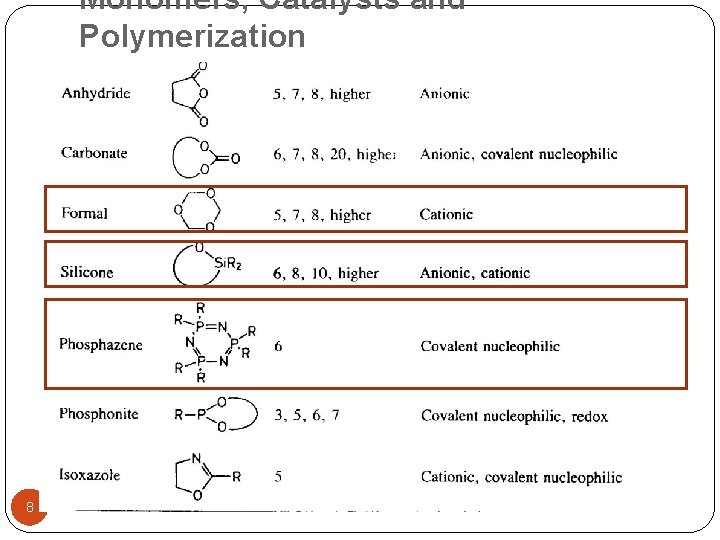
Monomers, Catalysts and Polymerization 8

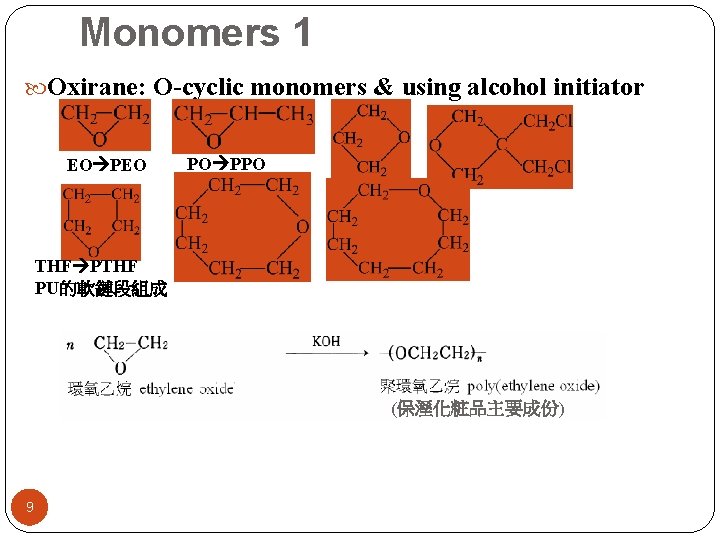
Monomers 1 Oxirane: O-cyclic monomers & using alcohol initiator EO PO PPO THF PU的軟鏈段組成 (保溼化粧品主要成份) 9

PEO (PEG) Application moosejaw. com Spenco 2 nd Skin is a hydrogel sheet, consisting of crosslinked polyethylene oxide, and containing 96% water, supported upon a net of low density polyethylene, which provides strength. The gel, which is capable of absorbing approximately its own weight of wound exudate, is permeable to water vapor and oxygen, but impermeable to water and bacteria. The dressing provides a moist environment upon the surface of the wound and provided that it is not allowed to dry out, the dressing will not adhere to the underlying tissue upon removal. 氨基酸洗面皂 全成份 Dis Water、Sodium Lauroyl Glutamate、Polyethylene Glycol 400、Potassium Laurate、1 -3 Butylene Glycol、 Methyl Paraben、Propyl Paraben、Perfume 10 Compete-cosmetic. com 成份說明 ◎ 聚乙二醇400(Polyethylene Glycol 400):界面活性劑, 防止水分過度流失。

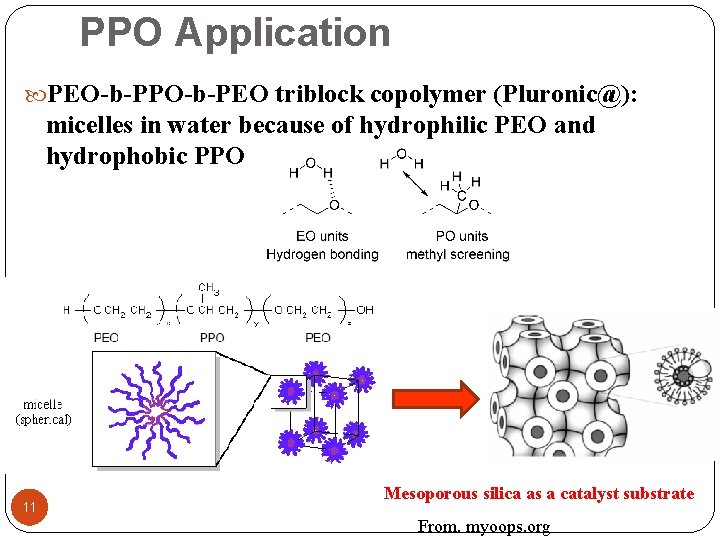
PPO Application PEO-b-PPO-b-PEO triblock copolymer (Pluronic@): micelles in water because of hydrophilic PEO and hydrophobic PPO 11 Mesoporous silica as a catalyst substrate From. myoops. org

PTHF Application PU Chemistry: diisocyanate diol Urethane linkages PU Structures: Hard domain (aggregation of urethane units via Hydrogen bonding) and soft domain (chain entanglement of alkoxide chains (PTHF) between urethane). PU Elastomers: Hard domains are used to keep the shape of elastomers and soft domains are used to extend when external force is applied. Foaming 12 From wikipedia. org

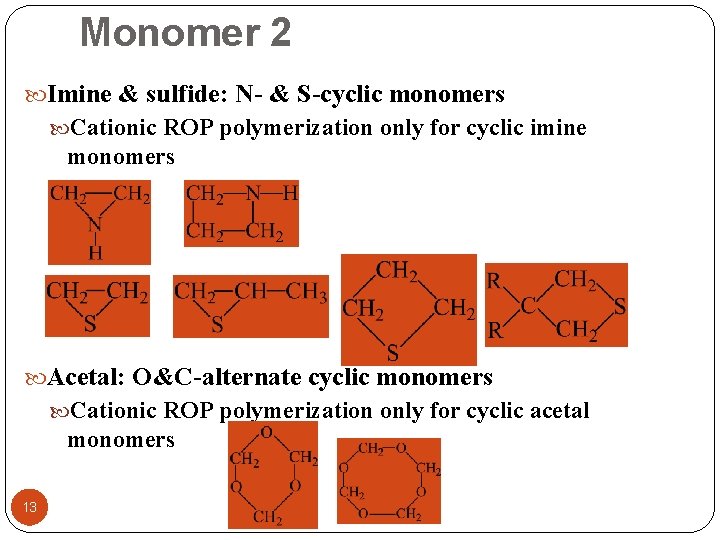
Monomer 2 Imine & sulfide: N- & S-cyclic monomers Cationic ROP polymerization only for cyclic imine monomers Acetal: O&C-alternate cyclic monomers Cationic ROP polymerization only for cyclic acetal monomers 13

Polyacetal Application Polyoxymethylene (POM): engineering thermoplastic, high stiffness, low friction and excellent dimensional stability. Five engineering thermoplastic: Polyamide (PA, i. e. , Nylon 6), POM, polycarbonate (PC, CD substrate), polyphenylene oxide (PPO), Polybutylene terephthalate (PBT). 14 Used for guitar picks From wikipedia. org

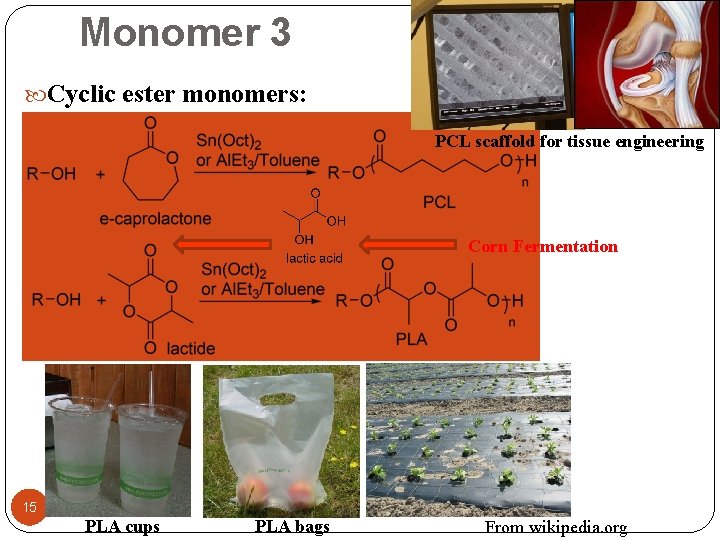
Monomer 3 Cyclic ester monomers: PCL scaffold for tissue engineering Corn Fermentation 15 PLA cups PLA bags From wikipedia. org

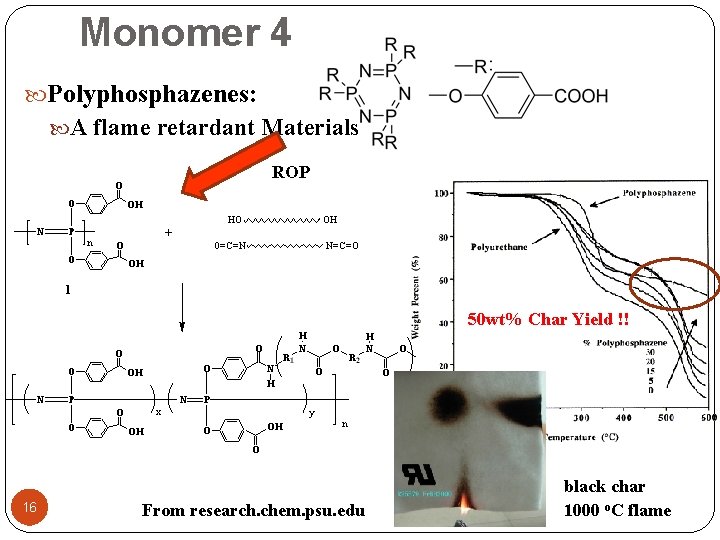
Monomer 4 Polyphosphazenes: A flame retardant Materials ROP 50 wt% Char Yield !! 16 From research. chem. psu. edu black char 1000 o. C flame

Monomer 5 Silicon (矽利康) Silicon Oil: Polydimethylsiloxane (PDMS) Viscosity of PDMS depends on Mn !! 17 Silicon Oil Lubrication Silicon Ruber Hose

Summary Polymerization without new bonds: cyclic monomers to linear polymer. Ionic initiator or using metal catalyst (coordination) The ring opening polymerization is developed to improve the condensation polymerization by combination of radical polymerization behavior--- initiation. Common ROP polymers: Nylon 6, PEO, PPO, PTHF, POM, PCL, PLA, Polyphosphazenes, PDMS. A chemist should remember the chemical names and the chemical structures of common ROP polymers. 18 Thanks for Your attention