Rift valley fever BY Fatma Khalifa Defination It

Rift valley fever BY : Fatma Khalifa

Defination • It is an arthropode borne viral disease caused by an Arbovirus. • Characterized by Sudden onset of storms abortion among pregnant animal and high mortality of neonates. • The disease is zoonotic and human beings is highly susceptible. • The disease classified as an OIE list A disease

Aetiology • Rift Valley fever virus (RVFV), genus Phlebovirus and family Bunyaviridae. • It was isolated from infected sheep in 1930 • This family contains the most arthropodes born viruses Macrophages, Hepatocytes and Endothelial cells are the target cells • The incubation period (interval from infection to onset of symptoms) for RVF varies from two to six days.
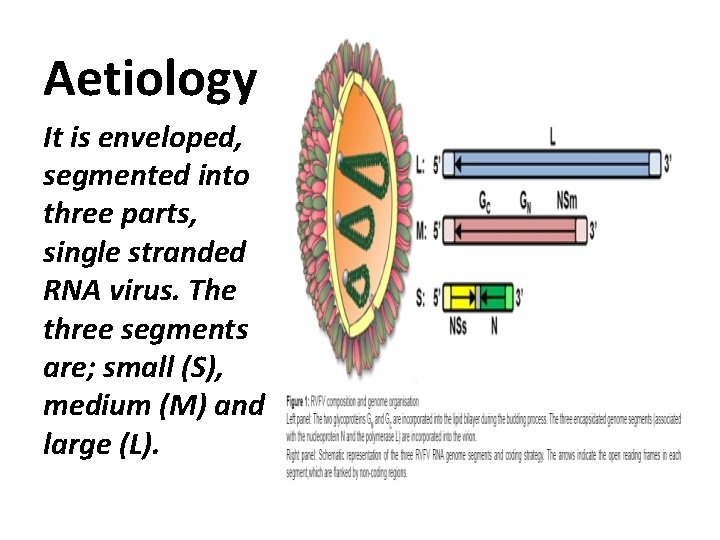
Aetiology It is enveloped, segmented into three parts, single stranded RNA virus. The three segments are; small (S), medium (M) and large (L).
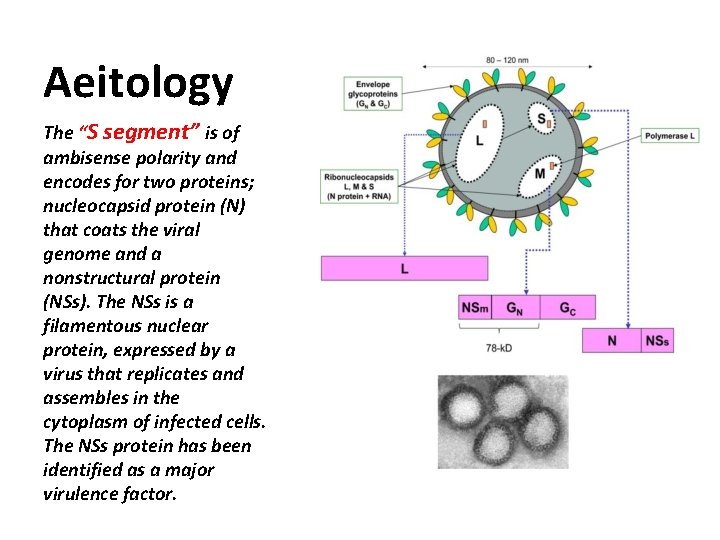
Aeitology The “S segment” is of ambisense polarity and encodes for two proteins; nucleocapsid protein (N) that coats the viral genome and a nonstructural protein (NSs). The NSs is a filamentous nuclear protein, expressed by a virus that replicates and assembles in the cytoplasm of infected cells. The NSs protein has been identified as a major virulence factor.
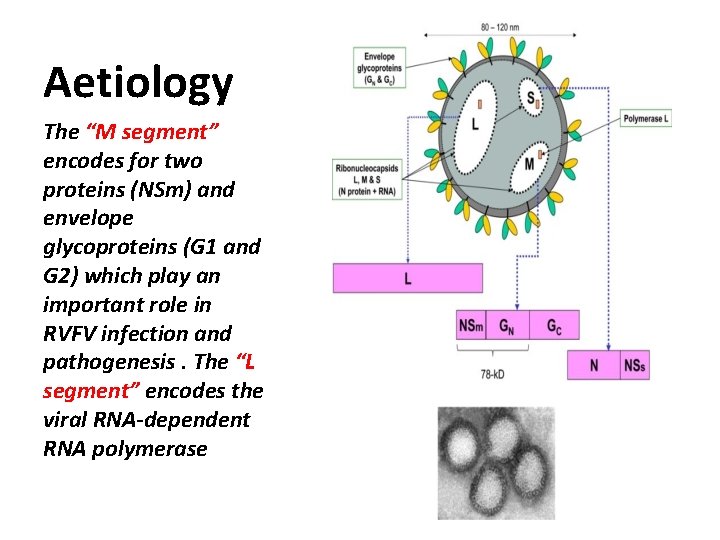
Aetiology The “M segment” encodes for two proteins (NSm) and envelope glycoproteins (G 1 and G 2) which play an important role in RVFV infection and pathogenesis. The “L segment” encodes the viral RNA-dependent RNA polymerase

Transmission RVFV is an exceptional virus in that it can be vectored by many, and quite varied, arthropods depending on the specific geographic location. For example, several species of mosquito, sandflies and ticks are all capable of transmitting RVFV between hosts. mosquitoes “bridge vectors” are the primary and most important vector for this virus to livestock.

Transmission Aedes mosquitoes are the dominant mosquito vectors for enzootic transmission among animals, and for sporadic transmission among humans.
Transmission when specific features of the landscape emerge, Culex mosquit oes serve as important amplifying vectors, which transition virus circulation and transmission in animals from enzootic conditions to epizootic conditions, and subsequently also increase the risk for human epidemics.
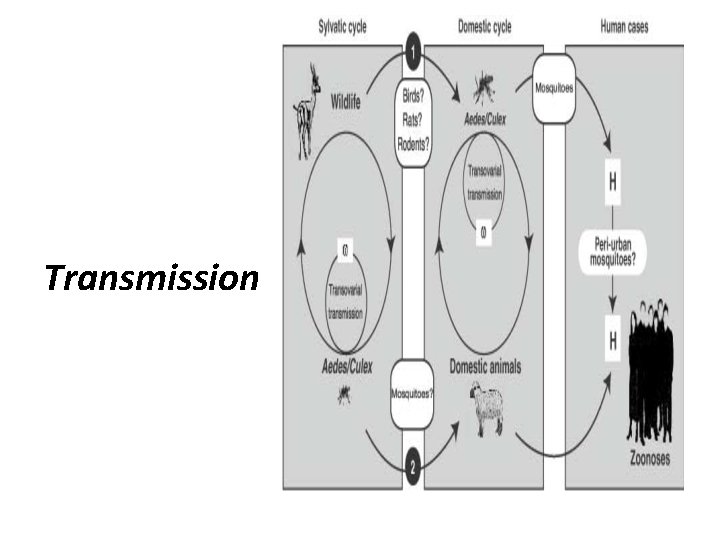
Transmission

Suseptibility • Susceptible host : • Cattle , Buffaloes , Sheep , Goat and Camels. Young animals (Lambs and Calves) usually show the most sever form of disease. “likely to be more severe in young lambs and less severe in older animals”. • Rodents are important hosts and bats to a lesser degree. • Human susceptible, following to exposure to their livestock.

Human infection • Humans infected by the RVF virus: 1 - Bitten by infected mosquitoes. 2 - Contact with blood, other body fluids or tissues during killing, skinning and cutting of infected animals. 3 - Consumption of raw milk or uncooked meat from infected animals.

Pathogensis • Mosquito suck the blood containing virus of the infected animal. • Transmit the virus to other animals on which they subsequently feed. • Some species of mosquitoes (Aedes, for example) are capable of transmitting the virus from infected female mosquitoes to offspring via its eggs.

Pathogensis • Mosquito eggs may survive up to several years in dry conditions. • During periods of high rainfall and/or flooding the eggs hatch and there is an increase in the infection of animals on which these mosquitoes feed.
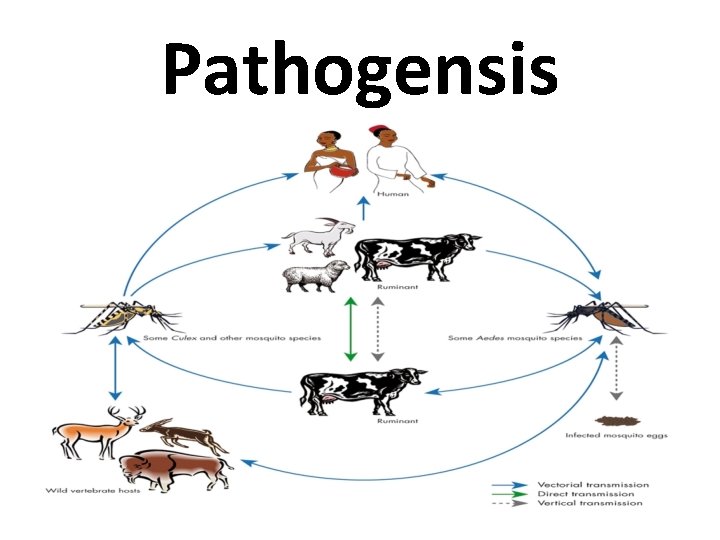
Pathogensis
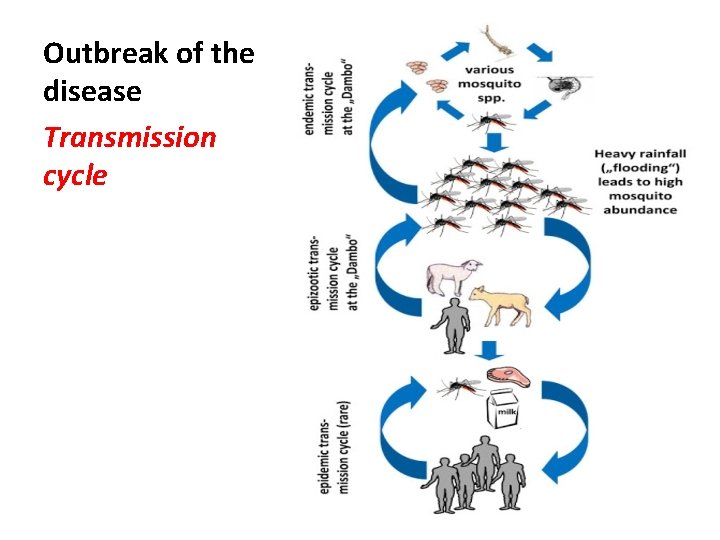
Outbreak of the disease Transmission cycle

Outbreak in Egypt • Four major epidemics have been recorded in Egypt (1977, 1978, 1993 and 2003). resulted in human and animal mortality.

Economic importance • RVF outbreaks can have significant societal consequences, including : • Massive reduction in trade and economic losses. • The virus mostly affects livestock, leading to : disease and abortion in domesticated animals, which is a vital source of income for many.

Clinical signs • The clinical signs of RVF in sheep and goats classified into four groups according to the severity of the disease. 1 - Hyperacute, 2 - Acute, 3 - Subacute, 4 - Inapparent. • Each group is considered separately. • In general, goats are less severely affected than sheep, with much lower morbidity and mortality, with fewer abortions and less severe clinical signs.

Clinical signs • Hyperacute RVF. Susceptible sheep breeds, showing : 1 - 90 -100 % abortion during periods of intense RVF virus transmission in epizootic situations. 2 - 80 -100 % of lambs under 10 days of age, “born during such periods”, die from RVF. 3 - Most deaths are sudden and occur within 12 hours after the onset of a pyrexia (40 -42°C). • Collapse and death are all that may be seen. 4 - Other lambs may be depressed, too weak to suckle or stand, and die within 24 -48 hours, without showing clinical disease other than fever, an elevated respiratory rate and prostration.

Ewe abortion

Clinical signs • Acute RVF Older lambs, “ 2 -3 weeks of age” and all. susceptible breeds, showing: 1 - High fever and an elevated respiratory rate, mucopurulent or sero-sanguineous “bloody”nasal discharges. 2 - Vomiting and often abdominal pain. 3 - Generalized lymphadenitis and gait abnormalities , animals are disinclined to move, become recumbent. 4 - Haemorrhagic diarrhoea, often develop. 5 - Abortion. 6 - Deaths occur after 24 -48 hours and may continue throughout the flock for up to ten days. Mortality rates may be from 10 to 60%. Sick and recovering animals generally show moderate to severe signs of jaundice.

Clinical signs • Subacute RVF. more frequent in adult animals. 1 - Febrile reaction of 40. 5 - 42°C and persists for “ 1 -5”days. 2 - Anorexia, nasal discharges, vomiting and other signs are seen but are generally less severe than in young animals. 3 - Abortion is a feature. 4 - Diarrhoea. colic may be less obvious, there may be 5 - Some lack of coordination of movement, animals are weak or recumbent for several days but most recover. Many will be jaundiced and may remain unthrifty and weak for several months. The mortality rates are of the order of 5 -20 percent.

Clinical signs • Inapparent RVF. This occurs in older or resistant animals. 1 - Transient periods of fever, which are not detected. The fever may be accompanied by depression or a period of inappetence, which are unremarkable. 2 - Abortions may follow such infections. These infections are only detected by subsequent serological testing.

RVF in Camels • Camels do not normally show any clinical signs following RVF infections and fall into the inapparent infection group. • They have a period of viraemia. • Abortion is a common consequence of the infection, and pastoralists complain of “all their camels aborting”. • The infections can be confirmed serologically. • Deaths occur in the early post-natal period in camel foals born during RVF epizootic periods.

Post mortem lesion • widespread petechial and ecchymotic haemorrhages throughout the carcass ( parenchyma and visible in subcapsular tissues). especially noticeable on the serosal and pleural surfaces of the body cavities, and on the heart, gall bladder, kidneys, bladder and other organs. There may be some bloodstained ascitic fluid. • Generalized lymphadenopathy involving the superficial and visceral lymph nodes “oedematous with petechial haemorrhages”

Post mortem lesion Hepatic lesions develop early in the course of the disease in all RVF carcasses. • In the early stages the liver is congested, swollen and engorged with rounded edges and many scattered petechial haemorrhages. • Later, the necrosis may be small 1 -3 mm foci, which coalesce to form larger areas of necrosis (initially focal then often pan-necrosis )and these changes involve the whole of the liver.

Post mortem lesion • Liver congestion, then later a bronze to yellow colour. • Necrotic changes induce jaundice and a stage is reached when the liver has a bronze appearance as the congestion, necrotic areas and icterus develop. At later stages the liver will be completely yellow with jaundice.

Post mortem lesion Lung lesion : congestion with oedema , emphysema and subpleural haemorrhages. Alimentary tract lesion : Some level of inflammation from catarrhal to haemorrhagic and necrotic. Serosal surfaces , mucosal lining of the bowel, particularly of the abomasum and small intestine and ileo-caecal areas = aemorrhages

Post mortem lesion • Spleen lesion : may or may not be enlarged with subcapsular haemorrhages. • Heart lesion : Show subepicardial and endocardial haemorrhages. Similar pathology in foetus, with autolysis
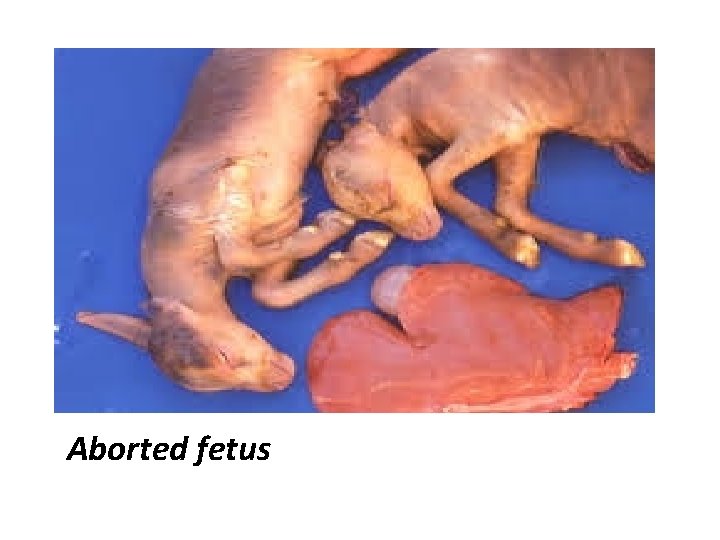
Aborted fetus
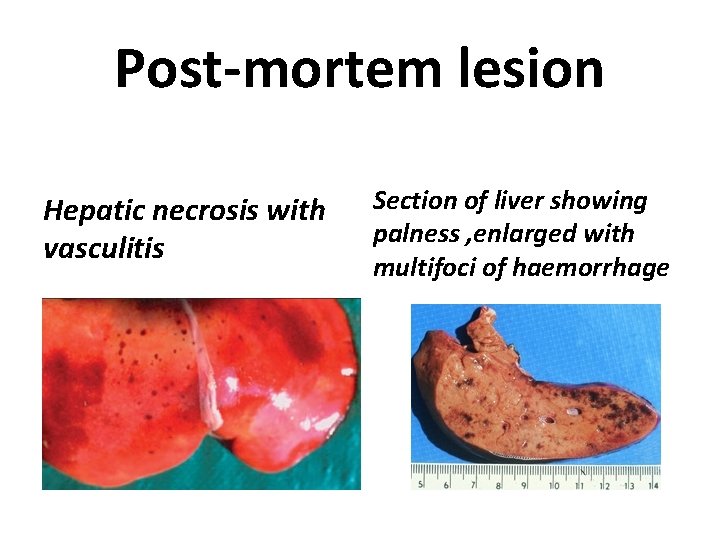
Post-mortem lesion Hepatic necrosis with vasculitis Section of liver showing palness , enlarged with multifoci of haemorrhage
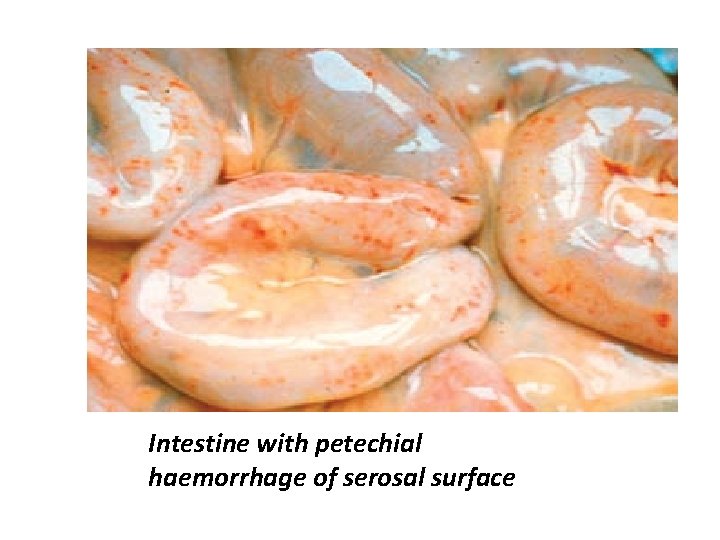
Intestine with petechial haemorrhage of serosal surface
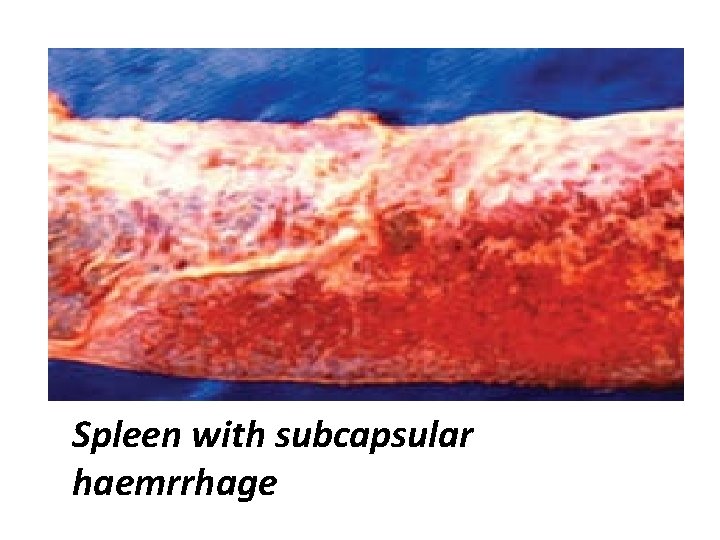
Spleen with subcapsular haemrrhage

Diagnosis • Case history. • Clinical signs. • Post-mortem lesion. • Laboratory diagnosis : a- isolation and identification of the virus. b- inoculation in mice. c- PCR for detection of virus. • Differential diagnosis from Brucellosis , Blue tongue , Toxoplasma , Listeriosis …….

Clinical pathology • Leucopenia which is most obvious in the early period of the infection. • High blood levels of enzymes associated with liver damage e. g. glutamic dehydrogenase (GLDH). • Thrombocytopenia • A viraemia persists over the period of the biphasic temperature reaction and possibly also after this has declined.

Treatment NO specific treatment for RVF

Control and prevention • • Notification. Isolation to infected animals. Restrication of animal movement. Application of quaratine measurs. Proper disposal of all sources of infection. Proper disinfection. Control the insect vector. Vaccination using Inactivated or Living attenuated tissue culture.

Vaccination • Lambs &Calves Ø Lambs 2 -3 months and calves 3 -4 months of age from vaccinated dams are vaccinated. Ø Lambs and calves from non vaccinated dams are vaccinated at first weeks of life.

Vaccination • Inactivated vaccine gives immunity for 4 -6 months. • Attenuated vaccine gives immunity for 1 -2 years. • Attenuated vaccine may cause abortion or fetal anomalies (it should be used only in enzootic areas). • Colostral immunity is 5 months.
- Slides: 40