Rickettsia rickettsii 1 Common Bacterial Pathogens Associated with

Rickettsia rickettsii

1. Common Bacterial Pathogens Associated with Rickettsia rickettsii infection

Common Bacterial Pathogens Associated with Rickettsia rickettsii infection • R. rickettsii causes Rocky Mountain Spotted Fever (RMSF) • RMSF = specific illness that falls within the broader class of Spotted Fever • The most common pathogens implicated in cases of R. rickettsii infection are mostly Genus Rickettsia pathogens yield infections with ~ symptoms to RMSF • Ticks = vector of infection for most pathogens associated with rickettsial infection

Some pathogens commonly associated with this infection are a part of the Rickettsia Genus
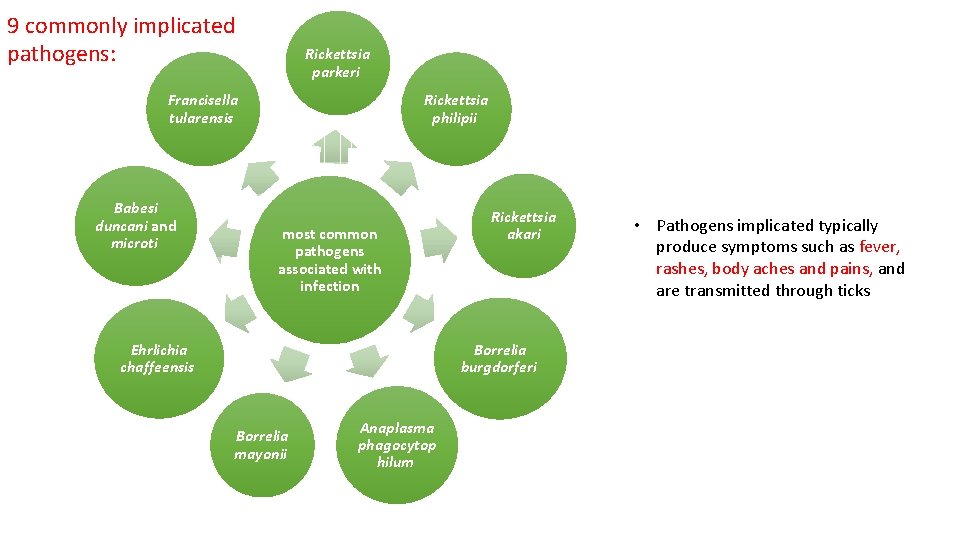
9 commonly implicated pathogens: Rickettsia parkeri Francisella tularensis Babesi duncani and microti Rickettsia philipii most common pathogens associated with infection Ehrlichia chaffeensis Rickettsia akari Borrelia burgdorferi Borrelia mayonii Anaplasma phagocytop hilum • Pathogens implicated typically produce symptoms such as fever, rashes, body aches and pains, and are transmitted through ticks

Common Bacterial Pathogens Associated with Rickettsia rickettsii Rickettsia parkeri Rickettsiosis symptoms = fever, headache, red rash, myalgia 1 All 3 yield eschar(dark scab) or ulceration at site of tick bite 2 3 Rickettsia philipii Pacific coast Tick Fever (common in children) Symptoms = fever, headache, red rash Rickettsia akari Rickettsialpox: fever, chills, headache, malaise, red rash
Common Bacterial Diseases Associated with Rickettsia rickettsii: Lyme Disease • The most common tick borne disease in America • CDC estimates ~ 300, 000 cases per year • Lyme disease is often associated with the R. rickettsii infection because it produces similar symptoms: • fever, headache, characteristic rashes • Erythema migrans “bulls-eye” rashes unique to Lyme disease

Common Bacterial Pathogens Associated with Rickettsia rickettsii (cont’d) Borrelia burgdorferi Lyme Disease & associated symptoms 4 5 6 Borrelia mayonii Lyme Disease & associated symptoms Anaplasma phagocytophilum Anaplasmosis Symptoms = fever, headache, leukopenia, low neutrophil count, liver function failure as noted by high [liver enzyme]

Common Bacterial Pathogens Associated with Rickettsia rickettsii (cont’d) Babesi duncani & microti Babesiosis Symptoms = fever, headache, chills, abnormal leukocyte count, abnormal liver function, decreased thrombocytes (platelet producing cells) 7 8 9 Ehrlichia Chaffeensis human monocyte ehrilichiosis Symptoms = fever, headache, rashes Francisella tularensis Tularemia (disruption of host tissue function; pathogen enters and disrupts macrophages) Symptoms = fever, lethargy, lymph node inflammation, skin lesion

• The geographical distribution of tick species dictates the distribution of bacterial species: • Ixodes scapularis tick spread Lyme disease through the pacific region • E. chaffeensis tick east coast • Lone star tick South and West United States Ticks are the vector for many diseases including R. rickettsii
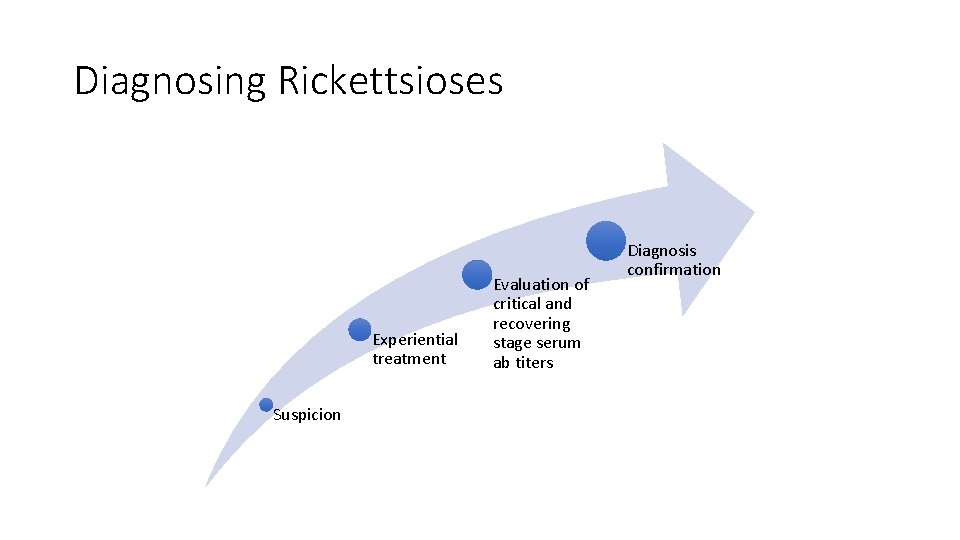
Diagnosing Rickettsioses Experiential treatment Suspicion Evaluation of critical and recovering stage serum ab titers Diagnosis confirmation

2. Samples taken back to the lab are important for diagnosis
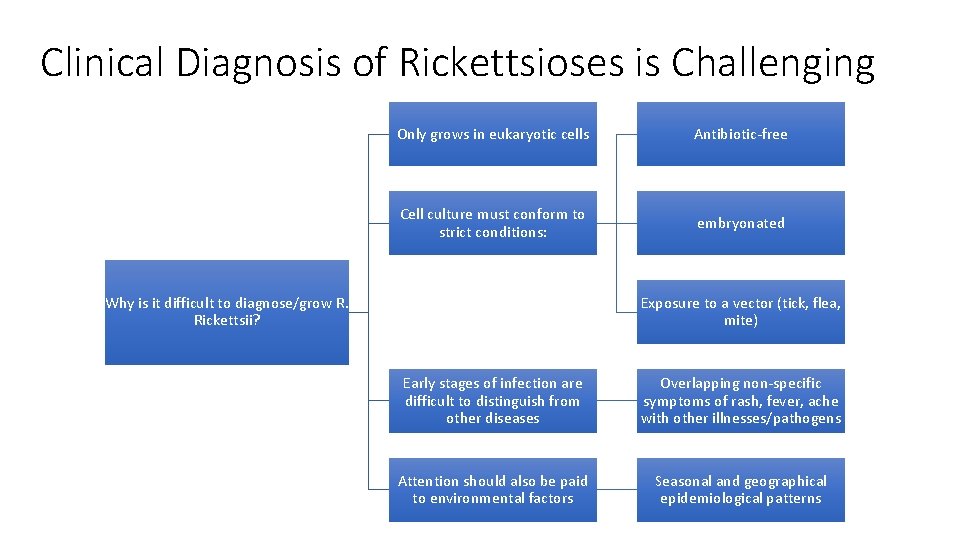
Clinical Diagnosis of Rickettsioses is Challenging Only grows in eukaryotic cells Antibiotic-free Cell culture must conform to strict conditions: embryonated Why is it difficult to diagnose/grow R. Rickettsii? Exposure to a vector (tick, flea, mite) Early stages of infection are difficult to distinguish from other diseases Overlapping non-specific symptoms of rash, fever, ache with other illnesses/pathogens Attention should also be paid to environmental factors Seasonal and geographical epidemiological patterns
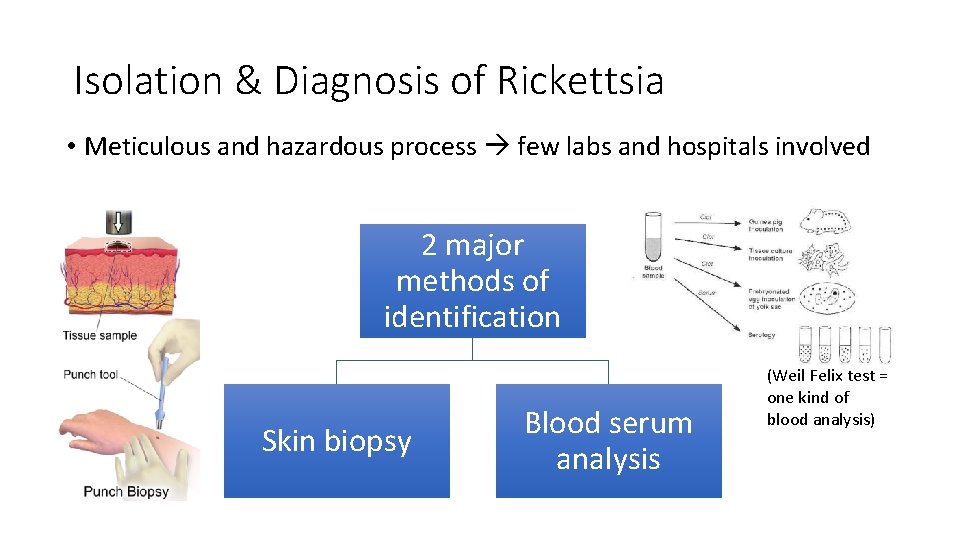
Isolation & Diagnosis of Rickettsia • Meticulous and hazardous process few labs and hospitals involved 2 major methods of identification Skin biopsy Blood serum analysis (Weil Felix test = one kind of blood analysis)
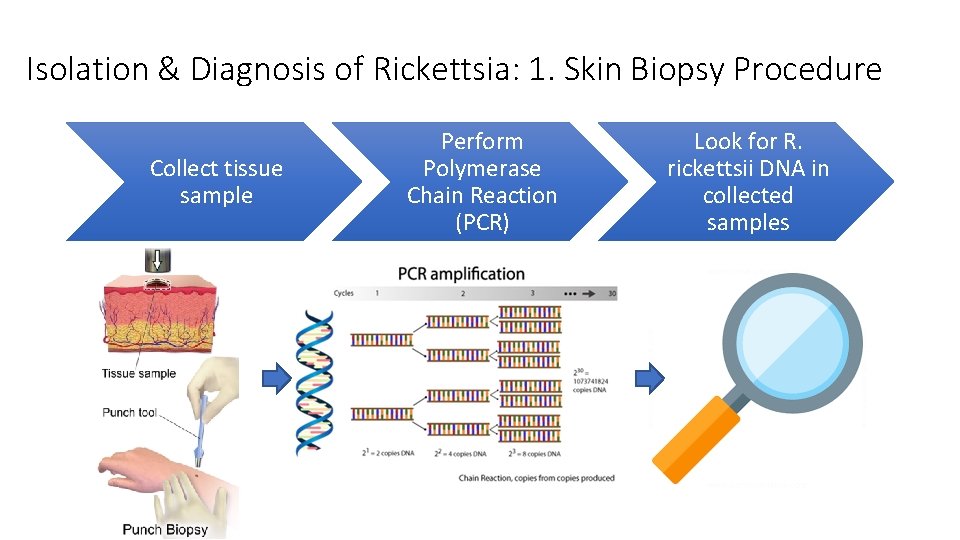
Isolation & Diagnosis of Rickettsia: 1. Skin Biopsy Procedure Collect tissue sample Perform Polymerase Chain Reaction (PCR) Look for R. rickettsii DNA in collected samples
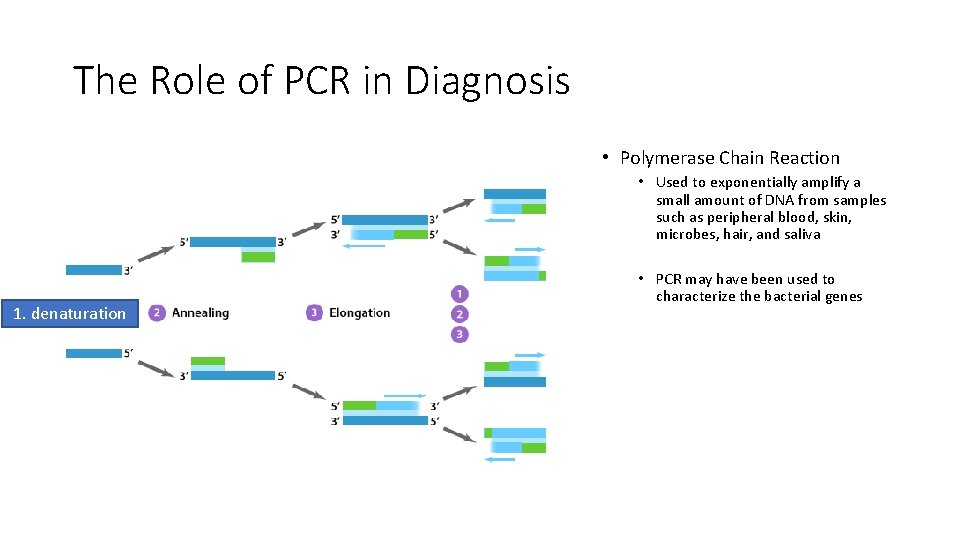
The Role of PCR in Diagnosis • Polymerase Chain Reaction • Used to exponentially amplify a small amount of DNA from samples such as peripheral blood, skin, microbes, hair, and saliva 1. denaturation • PCR may have been used to characterize the bacterial genes
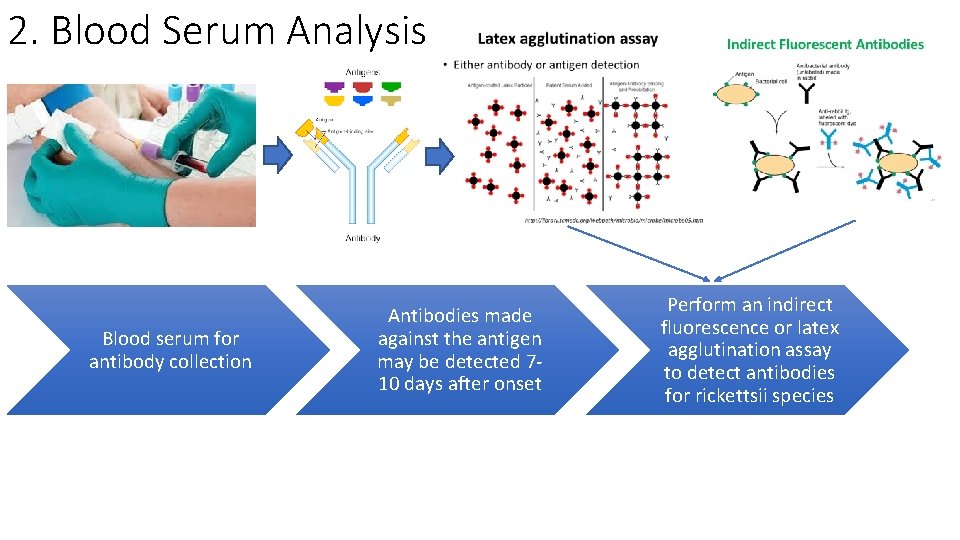
2. Blood Serum Analysis Blood serum for antibody collection Antibodies made against the antigen may be detected 710 days after onset Perform an indirect fluorescence or latex agglutination assay to detect antibodies for rickettsii species

2. Blood Serum Analysis (cont’d) • The indirect fluorescence and latex assay are better identification procedures for antibodies than the Weil-Felix test (a non specific test) based on cross-reactive antigens of Proteus vulgaris
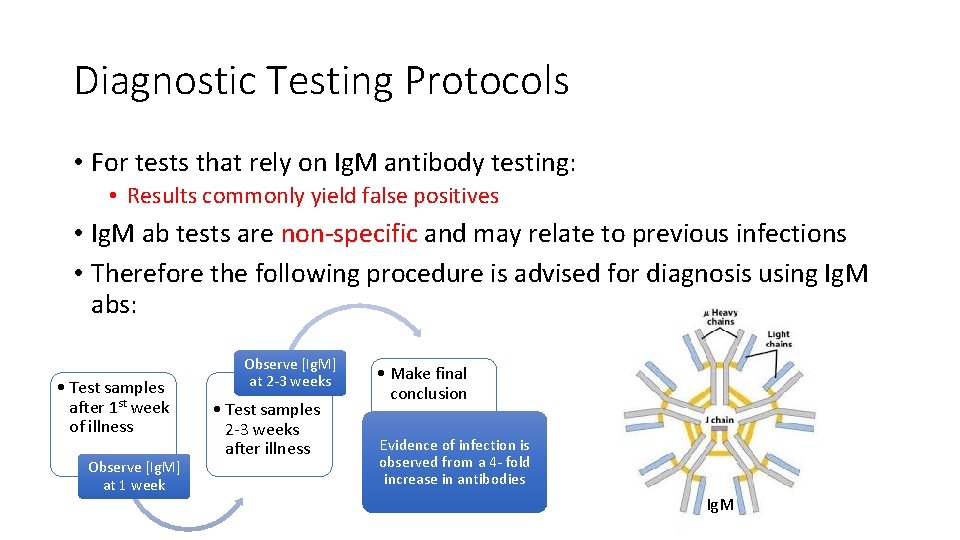
Diagnostic Testing Protocols • For tests that rely on Ig. M antibody testing: • Results commonly yield false positives • Ig. M ab tests are non-specific and may relate to previous infections • Therefore the following procedure is advised for diagnosis using Ig. M abs: • Test samples after 1 st week of illness Observe [Ig. M] at 1 week Observe [Ig. M] at 2 -3 weeks • Test samples 2 -3 weeks after illness • Make final conclusion Evidence of infection is observed from a 4 - fold increase in antibodies Ig. M

Diagnostic Testing Protocols (cont’d) The lab is necessary for identification - refrigerate serum samples at 2 -8°C - store samples in biohazardous sealed bags - if lacking lab resources use filtered paper to collect, store, transport blood samples (more convenient & cheaper) - antibiotic treatment can begin before lab confirmation - Identification & confirmation should be followed through even during nonspecific treatment of signs and symptoms to help with proceedings should infection worsen

Diagnostic Testing Protocol • Proceed with treatment during diagnosis process • Diagnosis takes time and is complicated therefore treatment must progress during confirmatory testing to prevent: • • Liver dysfunction Jaundice Splenomegaly Myocarditis Neurological conditions if the pathogen enters the nervous system: • Meningitis • Encephalitis • Retinal dysfunction

3. What tests will be performed?
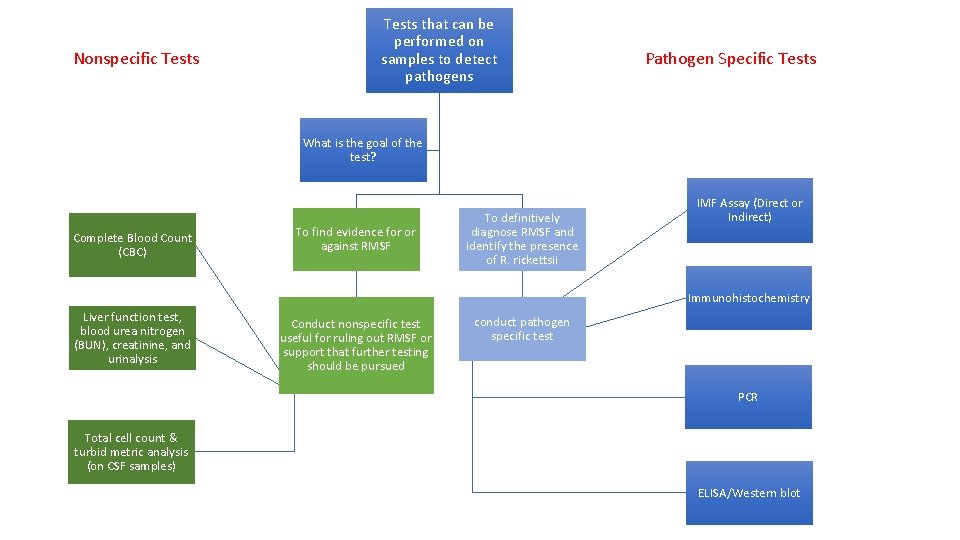
Nonspecific Tests that can be performed on samples to detect pathogens Pathogen Specific Tests What is the goal of the test? Complete Blood Count (CBC) To find evidence for or against RMSF To definitively diagnose RMSF and identify the presence of R. rickettsii IMF Assay (Direct or Indirect) Immunohistochemistry Liver function test, blood urea nitrogen (BUN), creatinine, and urinalysis Conduct nonspecific test useful for ruling out RMSF or support that further testing should be pursued conduct pathogen specific test PCR Total cell count & turbid metric analysis (on CSF samples) ELISA/Western blot
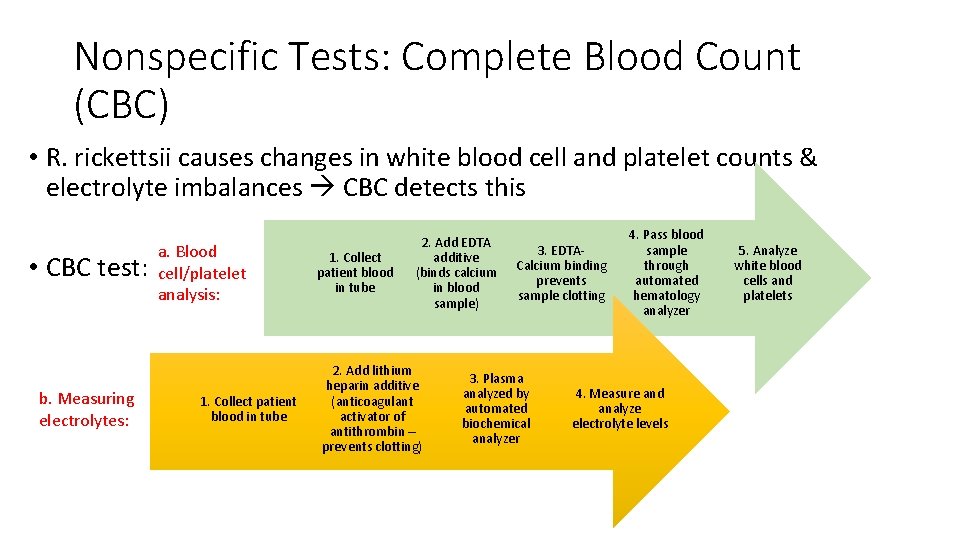
Nonspecific Tests: Complete Blood Count (CBC) • R. rickettsii causes changes in white blood cell and platelet counts & electrolyte imbalances CBC detects this • CBC test: b. Measuring electrolytes: a. Blood cell/platelet analysis: 1. Collect patient blood in tube 2. Add EDTA additive (binds calcium in blood sample) 2. Add lithium heparin additive (anticoagulant activator of antithrombin – prevents clotting) 3. EDTACalcium binding prevents sample clotting 3. Plasma analyzed by automated biochemical analyzer 4. Pass blood sample through automated hematology analyzer 4. Measure and analyze electrolyte levels 5. Analyze white blood cells and platelets

Nonspecific Tests: Liver function test, blood urea nitrogen (BUN), creatinine, and urinalysis • R. Rickettsii disrupts liver and kidney function: assess the function with these tests • Similar procedure to electrolyte analysis but liver enzymes and urea are measured not electrolytes 1. Add Sodium Citrate to Blood Sample (anticoagulant binds calcium, but with less strength than EDTA) 2. Plasma Extraction from sample 3. Tissue factor and excess calcium added (coagulation promoters) 4. Coagulation (limited by the Sodium citrate amount) occurs 5. Time it takes for coagulation to occur is measured analyzed 6. Make observations and conclusions about liver enzyme functioning/urea, etc.

Nonspecific Tests: Total cell count & turbid metric analysis (on CSF samples) • R. rickettsii infection alters the cells present and opacity in CSF, so testing CSF samples can find support for or against R. rickettsii being the underlying pathogen • Procedure: • 1. Obtain CSF sample through lumbar puncture (also can be obtained from skull base) run sample through automated cell counter determine what cells are present • 2. Visually analyze the opacity of the CSF and also use a turbidometer (turbid indicates presence of bacterial pathogens)

Pathogen Specific Tests: PCR • Enzymatic assay that allows you to amplify a small amount of DNA collected from peripheral blood, skin, microbes, hair and/or saliva • DNA amplification requires template DNA, primers, nucleotides, and DNA polymerase to be added into a thermal cycler for amplification to proceed:
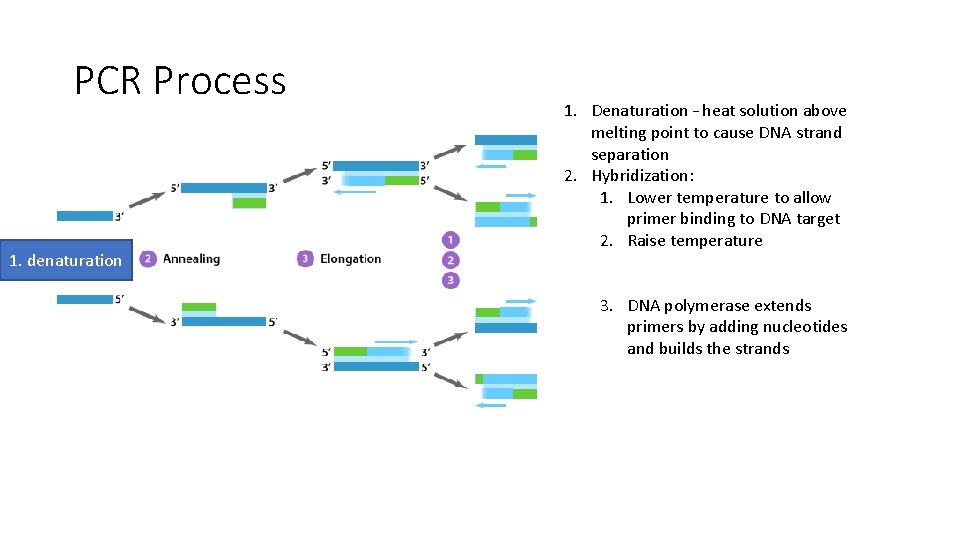
PCR Process 1. denaturation 1. Denaturation – heat solution above melting point to cause DNA strand separation 2. Hybridization: 1. Lower temperature to allow primer binding to DNA target 2. Raise temperature 3. DNA polymerase extends primers by adding nucleotides and builds the strands

Pathogen Specific Tests: (IHC) Immunohistochemistry • IHC shows distribution & localization of biomarkers in a cell or tissue • Targets ab/ag of interest through immunological, anatomical, and biochemical techniques to identify specific tissue components 1 IHC Procedure 2 3 • Dehydrate tissue sample and fix in formalin before embedding in paraffin • perform antigen retrieval recovers any ag altered by fixation • apply chromogen to illustrate the ab/ag • counterstain to illustrate tissue structure • use a secondary ab with an enzyme that is bound to a primary ab to bind the antigen • dehydrate sections

Pathogen Specific Test: Immunofluorescence assay • Immunofluorescence assay (IFA) is a cell imaging technique – Use antibodies to label antigen with fluorescent (FITC) dye • FITC dye used to visualize target antigen under fluorescent microscope • IFA used to detect Ig. G and Ig. M abs (2 pair serum sample) • Goal of IFA: detect if a 4 -fold increase in antibody titers (Elevated Ig. G and Ig. M antibody levels) has occurred sign that R. rickettsii could be present

Pathogen Specific Test: Immunofluorescence assay (cont’d) • There are 2 IFA methods i. e. direct v. indirect – allows determination of whether fluorophore is combined to primary or secondary ab • Collect blood samples for IFA analysis during the acute (early infection) and late infection phases(2 -4 weeks later) • Compare titer levels looking for 4 fold increase • Considered important for confirming if an infection is present or not
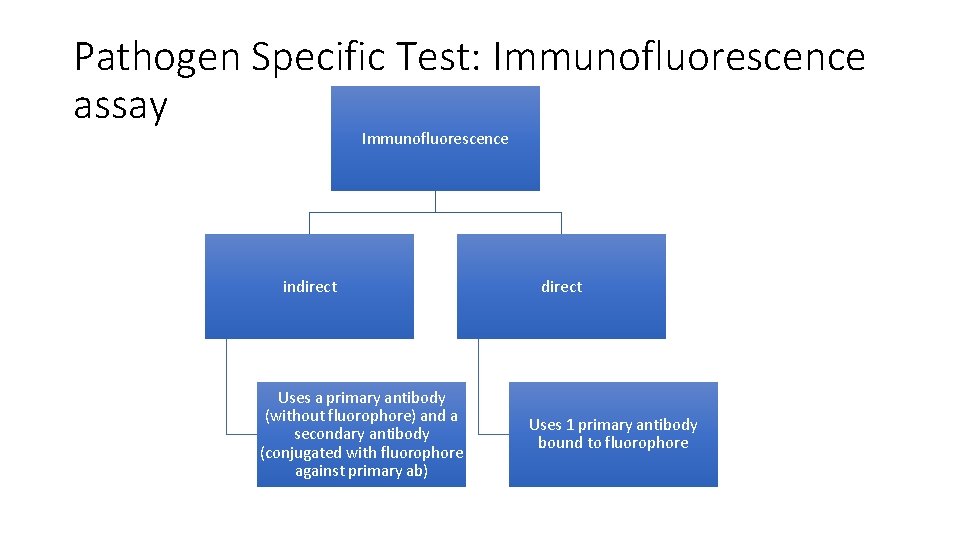
Pathogen Specific Test: Immunofluorescence assay Immunofluorescence indirect Uses a primary antibody (without fluorophore) and a secondary antibody (conjugated with fluorophore against primary ab) direct Uses 1 primary antibody bound to fluorophore

Pathogen Specific Test: ELISA/Western Blot • 2 tier test : usually used to diagnose Lyme disease • First: ELISA Second: Western Blot • 1. ELISA serum test performed to detect antibody presence in blood 2. Western blot performed to see if test is positive for infection (looking for Ig. M and Ig. G antibodies)
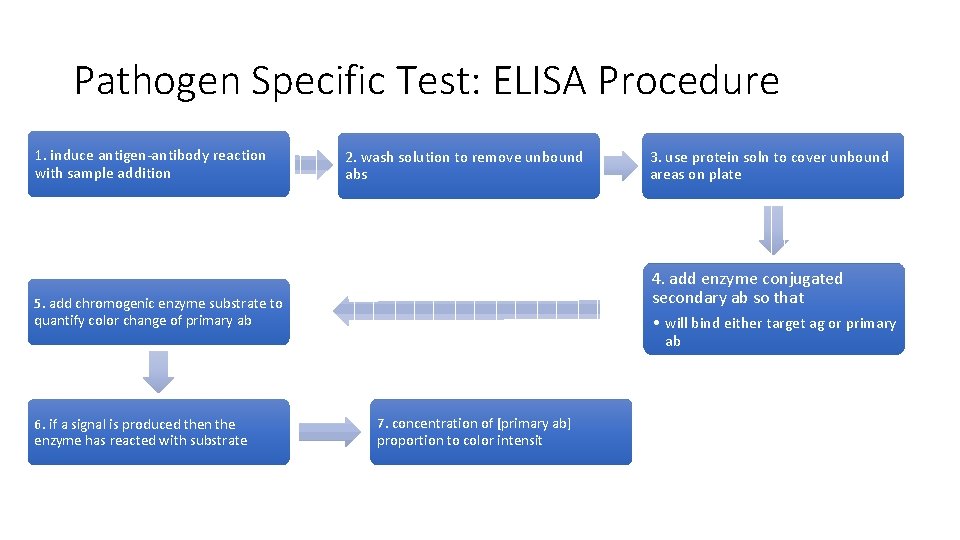
Pathogen Specific Test: ELISA Procedure 1. induce antigen-antibody reaction with sample addition 2. wash solution to remove unbound abs 4. add enzyme conjugated secondary ab so that 5. add chromogenic enzyme substrate to quantify color change of primary ab 6. if a signal is produced then the enzyme has reacted with substrate 3. use protein soln to cover unbound areas on plate • will bind either target ag or primary ab 7. concentration of [primary ab] proportion to color intensit

Pathogen Specific Test: Western Blot Procedure • 1. Run protein mixture through gel electrophoresis • Size based separation occurs • 2. Electrophoretically transfer proteins to membrane • Negatively charged proteins transfer from gel to membrane • 3. Wash to remove unbound ab • (Nonfat dried milk solution blocks nonspecific ab binding to membrane) • 4. Film band becomes visible band consists of bound ab to target protein • Thickness correlates to amount of protein present

4. Expected Results

Expected Test Results CBC Results - Low platelet count low sodium abnormal liver function (characterized by aminotransferase levels) moderate leukocytosis (immune response against R. rickettsii) Thrombocytopenia : endothelial cell attack causes decreased platelet circulation hyponatremia (low blood sodium) hypovolemia (low blood volume) decreased tissue perfusion (ADH release response causes further hyponatremia)

Expected Test Results Liver function Results elevated ALT and AST signifying liver damage caused by R. rickettsii Increased clotting time international normalized ratio/partial thromboplastin time (INR/PTT) should be increased damaged kidneys should demonstrate reduced function decrease in elimination of waste products elevated BUN

Expected Test Results • 1. Blood Smear Microscopy results • Reveal morulae (clusters of cells in host monocyte vacuoles) for ehrlichial or anaplasma infection • Insensitive diagnostic • 2. ELISA results • Results can’t be used effectively to monitor increases of decreases in antibody titer • 3. Indirect Immunofluorescence Assay (IFA) results • Ig. G and Ig. M antibodies from blood samples collected early (acute) and late (convalescent) in the disease are used for diagnosis. • increased Ig. M titers by the end of the first week of illness. • Ig. G antibody generally do not appear until 7 -10 days after the onset of illness. • Testing two sequential serum is more important in confirming acute infection with rickettsial agents - antibody titers may persist in some patients for years after the original exposure. • Limited by timeframe (need at least a week to develop abs) • Limited by false negatives produced

Expected Test Results • 4. Nucleic Acid Detection results • PCR provides diagnosis able to differentiate between different species of Rickettsia with assay for 2 target R. rickettsii genes – 17 k. Da-protein-encoding gene & omp. A outer membrane protein gene • time-consuming process • 5. Immunohistochemical Staining (IHC) results • immunostaining via skin biopsy of rash prior to therapy or within the first 48 hours after antibiotic therapy initiation • Detection of spotted fever group antigens found in endothelial cells within foci of vasculitis or perivascular inflammatory infiltrates; via biopsy specimen • used to confirm Rocky Mountain spotted fever in otherwise unexplained deaths • documents presence of organism before antibiotic treatment • 6. Culture results • BSL-3 (bio security level) agent • Culture and confirm R. rickettsii with PCR technique
- Slides: 40