RIBS VI A Prospective Multicenter Registry of Bioresorbable

RIBS VI: A Prospective, Multicenter, Registry of Bioresorbable Vascular Scaffolds in Patients With Coronary Artery Bare-Metal or Drug-Eluting In-Stent Restenosis Fernando Alfonso MD, Ph. D, FESC Hospital Universitario “La Princesa” Madrid. Spain. On Behalf of the RIBS VI Investigators Javier Cuesta MD, Fernando Rivero MD, María J. Pérez-Vizcayno MD, Bruno García MD, José R. Rumoroso MD, Francisco Bosa MD, Armando Pérez de Prado MD, Mónica Masotti MD, Raúl Moreno MD, Angel Cequier MD, Hipólito Gutiérrez MD, Arturo García-Touchard MD, José R López-Mínguez MD, Javier Zueco MD.
![Disclosure Statement of Financial Interest I, [Fernando Alfonso, MD, and the coauthors], DO NOT Disclosure Statement of Financial Interest I, [Fernando Alfonso, MD, and the coauthors], DO NOT](http://slidetodoc.com/presentation_image_h2/20168f74a64a87e6b5abc731934ebf32/image-2.jpg)
Disclosure Statement of Financial Interest I, [Fernando Alfonso, MD, and the coauthors], DO NOT have a financial interest/arrangement or affiliation with one or more organizations that could be perceived as a real or apparent conflict of interest in the context of the subject of this presentation

RIBS VI Background: Management of patients with in-stent restenosis (ISR) still remains a challenge. The role of bioresorbable vascular scaffolds (BVS) in these patients is unknown. However, the potencial benefits of their strong antiproliferative effects without the need of an additional permanent metal layer, make these devices an attractive strategy in this scenario.

RIBS VI Objective: To assess the efficacy of BVS in the treatment of patients with ISR. To compare BVS results with those obtained with drugeluting balloons (DEB) and everolimus-eluting stents (EES) in the RIBS IV and RIBS V RCT - Primary Endpoint: MLD at Follow-up - Clinical Endpoint: Combined (Cardiac D, MI, TVR/TLR) Clinical. Trials. gov ID: NCT 02672878
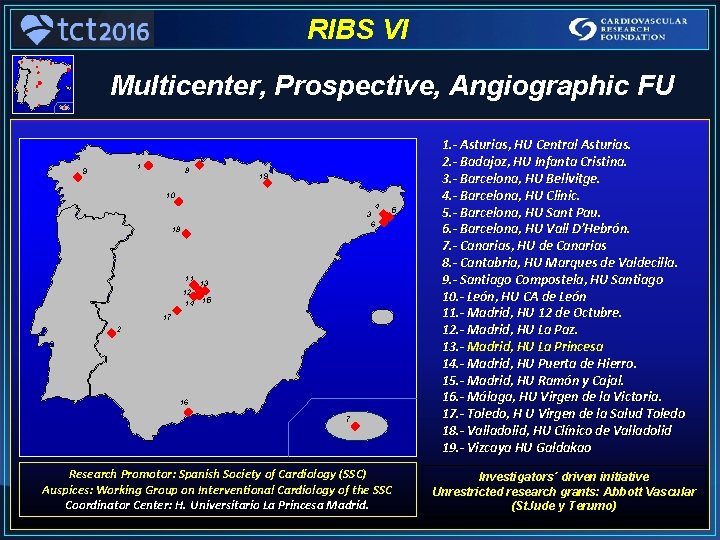
RIBS VI Multicenter, Prospective, Angiographic FU 1 9 8 19 10 4 3 6 18 11 12 14 5 13 15 17 2 16 7 Research Promotor: Spanish Society of Cardiology (SSC) Auspices: Working Group on Interventional Cardiology of the SSC Coordinator Center: H. Universitario La Princesa Madrid. 1. - Asturias, HU Central Asturias. 2. - Badajoz, HU Infanta Cristina. 3. - Barcelona, HU Bellvitge. 4. - Barcelona, HU Clinic. 5. - Barcelona, HU Sant Pau. 6. - Barcelona, HU Vall D’Hebrón. 7. - Canarias, HU de Canarias 8. - Cantabria, HU Marques de Valdecilla. 9. - Santiago Compostela, HU Santiago 10. - León, HU CA de León 11. - Madrid, HU 12 de Octubre. 12. - Madrid, HU La Paz. 13. - Madrid, HU La Princesa 14. - Madrid, HU Puerta de Hierro. 15. - Madrid, HU Ramón y Cajal. 16. - Málaga, HU Virgen de la Victoria. 17. - Toledo, H U Virgen de la Salud Toledo 18. - Valladolid, HU Clínico de Valladolid 19. - Vizcaya HU Galdakao Investigators´ driven initiative Unrestricted research grants: Abbott Vascular (St. Jude y Terumo)

RIBS VI Study Organization Coordinating Center: Hospital Universitario La Princesa. Madrid. Steering Committee: F. Alfonso (Chairman and Principal Investigator), J. Zueco, A. Cequier, C. Morís. Clinical Events Committee: R. Hernández, M. Sabaté. Coronary Angiography Core Laboratory: (Hospital Universitario La Princesa, Madrid) J. Cuesta, M. J. Pérez-Vizcayno. Data Coordination, Safety Monitoring and Statistical Committee: J. Cuesta, M. J. Pérez-Vizcayno, Cristina Fernández. Intravascular Ultrasound and Optical Coherence Tomography Committee: F. Alfonso J. Cuesta. Sites and Investigators: In order of enrollment: 1 Hospital Universitario La Princesa, Madrid, (F. Alfonso, J. Cuesta, F. Rivero, T. Bastante, A. Benedicto, M. García. Guimaraes); 2 Hospital Universitario Vall d’Hebron, Barcelona, (B. García del Blanco); 3 Hospital Universitario de Canarias (F. Bosa); 4 Hospital Galdakao, Vizcaya (J. R. Rumoroso); 5 Complejo Asistencial Universitario de León, (A. Pérez del Prado); 6 Hospital Universitario Clinic de Barcelona, (M. Masotti); 7 Hospital Universitario Infanta Cristina, Badajoz, (J. R. López-Mínguez); 8 Hospital Universitario de Valladolid, Valladolid (H. Gutierrez); 9 Hospital Universitario de Bellvitge, Barcelona, (A. Cequier); 10 Hospital Universitario Marqués de Valdecilla, Santander, (J. Zueco); 11 Hospital Universitario Puerta de Hierro, Majadahonda, (A. García-Touchard); 12 Hospital Universitario La Paz, Madrid, (R. Moreno); 13 Hospital Universitario 12 de Octubre, Madrid, (T. Velazquez); 14 Hospital Universitario Sant Pau, Barcelona, (V. Martí); 15 Hospitalario Universitario Central de Asturias, Oviedo, (C. Morís); 16 Hospital Ramón y Cajal, Madrid (R. Hernández); 17 Hospital Universitario Virgen de la Salud Toledo, (J. Moreu); 18 Hospital Universitario Virgen de la Victoria, Málaga, (J. M. Hernández); 19 Complejo Hospitalario Universitario de Santiago, Santiago de Compostela (R. Trillo)

RIBS VI Inclusion / Exclusion Criteria Inclusion: Informed consent Age 20 - 85 y ISR (> 50% stenosis) Angina or silent ischemia ISR amenable for BVS Exclusion: Stent Related: Stent location undefined ISR <1 Month Thrombus Vessel diameter < 2. 25 mm ISR outside the Stent General: Life expectancy < 1 y Female in childbearing age Problems FU angiography Intolerance DAT LVEF < 25%

RIBS VI Flow Diagram RIBS VI Prospective, Angio FU (BMS-ISR and DES-ISR) 141 Pts BVS 498 Pts ISR Randomization Same RIBS Centers Incl/Excl / Criteria Informed Consent April 2014 December 2015 January 2010 August 2013 309 Pts RIBS IV; 189 Pts RIBS V 249 Pts DEB EES Absorb Se. Quent Please Xience Prime (Abbott Vascular) (B. Braun) (Abbott Vascular) 100% Angio Success 134 Pts Angio FU QCA 219 Pts 223 Pts Angio FU Mean: 257 days Mean: 270 days QCA Primary End-point (95% of Eligible) 442 Pts: 91% of Eligible 498 1 Y Clinical FU (100%) 141 9 Mo (100%); 124 (88%) 1 Y (17 Pending) Clinical. Trials. gov Identifier: NCT 01239953 & NCT 01239940
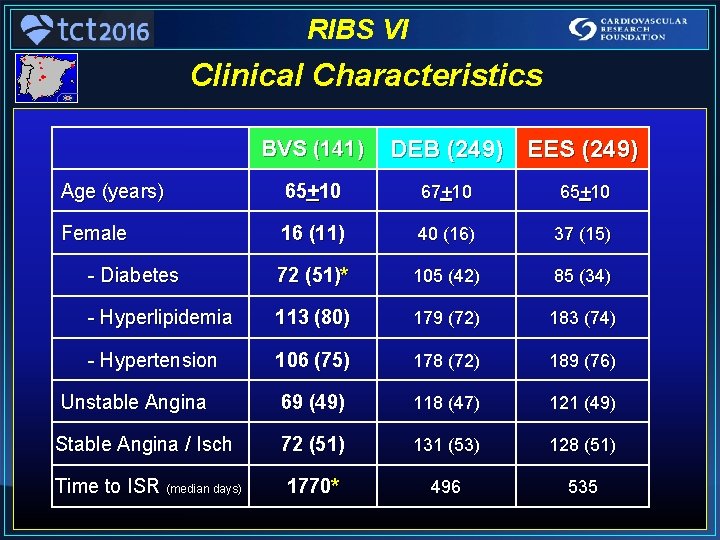
RIBS VI Clinical Characteristics BVS (141) DEB (249) EES (249) Age (years) 65+10 67+10 65+10 Female 16 (11) 40 (16) 37 (15) - Diabetes 72 (51)* 105 (42) 85 (34) - Hyperlipidemia 113 (80) 179 (72) 183 (74) - Hypertension 106 (75) 178 (72) 189 (76) Unstable Angina 69 (49) 118 (47) 121 (49) Stable Angina / Isch 72 (51) 131 (53) 128 (51) Time to ISR (median days) 1770* 496 535
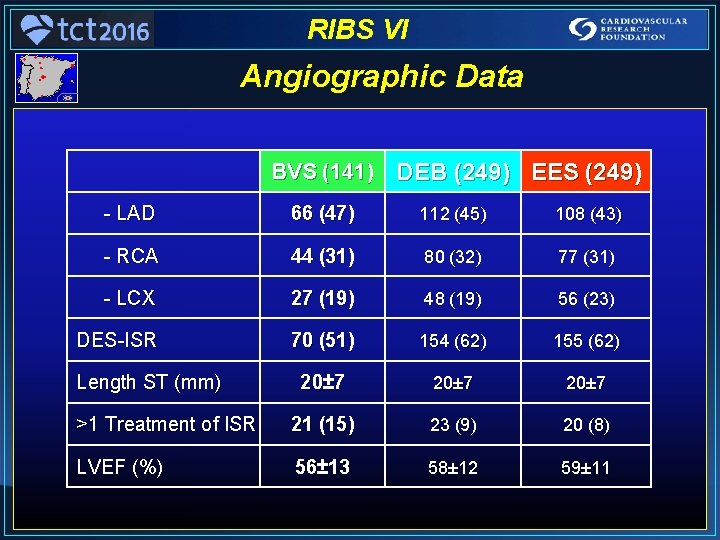
RIBS VI Angiographic Data BVS (141) DEB (249) EES (249) - LAD 66 (47) 112 (45) 108 (43) - RCA 44 (31) 80 (32) 77 (31) - LCX 27 (19) 48 (19) 56 (23) DES-ISR 70 (51) 154 (62) 155 (62) 20± 7 >1 Treatment of ISR 21 (15) 23 (9) 20 (8) LVEF (%) 56± 13 58± 12 59± 11 Length ST (mm)
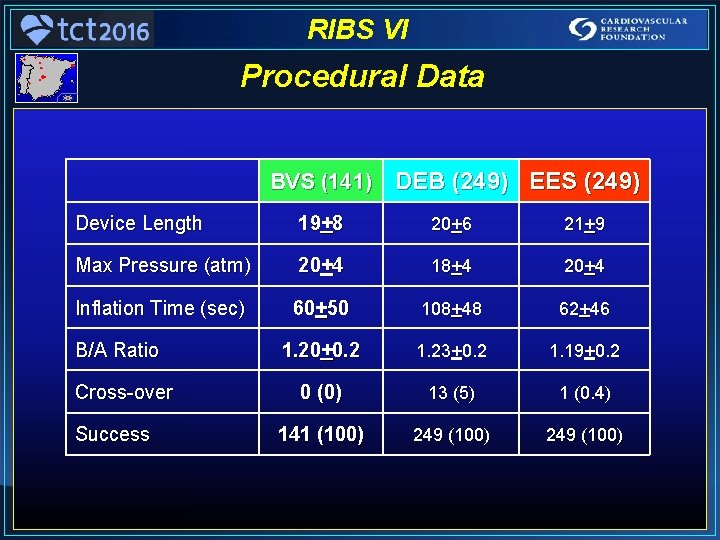
RIBS VI Procedural Data BVS (141) DEB (249) EES (249) Device Length 19+8 20+6 21+9 Max Pressure (atm) 20+4 18+4 20+4 Inflation Time (sec) 60+50 108+48 62+46 1. 20+0. 2 1. 23+0. 2 1. 19+0. 2 0 (0) 13 (5) 1 (0. 4) 141 (100) 249 (100) B/A Ratio Cross-over Success
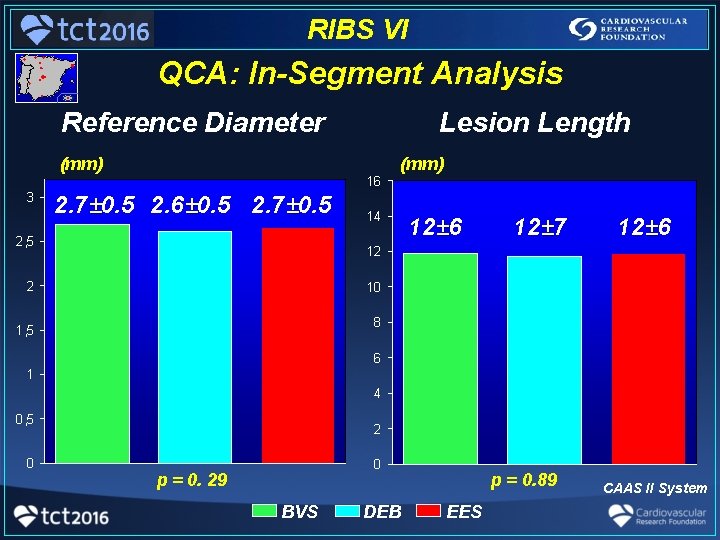
RIBS VI QCA: In-Segment Analysis Reference Diameter Lesion Length (mm) 16 3 2. 7± 0. 5 2. 6± 0. 5 2. 7± 0. 5 2, 5 14 12± 6 12± 7 12± 6 12 2 10 8 1, 5 6 1 4 0, 5 0 2 0 p = 0. 29 BVS DEB p = 0. 89 EES CAAS II System
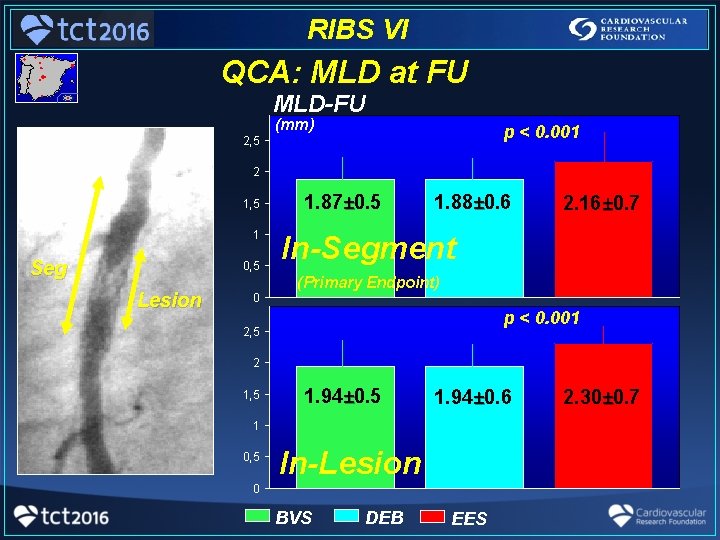
RIBS VI QCA: MLD at FU MLD-FU (mm) p < 0. 001 2, 5 2 1, 5 1 Seg 0, 5 Lesion 0 1. 87± 0. 5 1. 88± 0. 6 2. 16± 0. 7 In-Segment (Primary Endpoint) p < 0. 001 2, 5 2 1, 5 1. 94± 0. 6 1 0, 5 In-Lesion 0 BVS DEB EES 2. 30± 0. 7
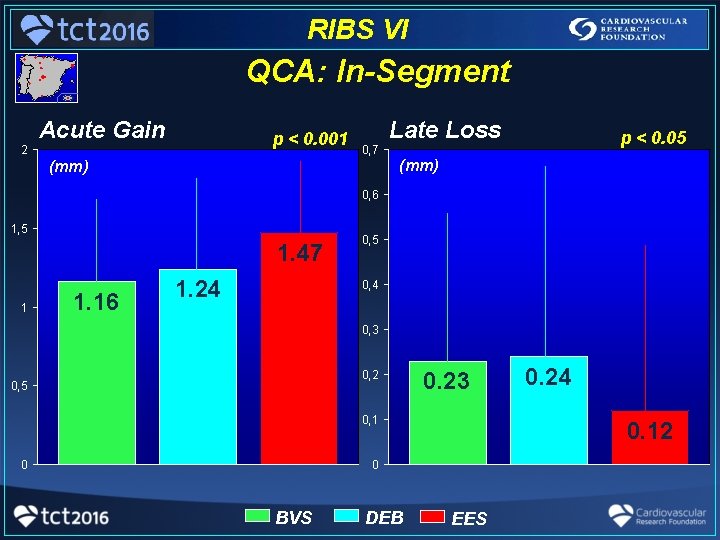
RIBS VI QCA: In-Segment 2 Acute Gain p < 0. 001 Late Loss p < 0. 05 0, 7 (mm) 0, 6 1, 5 1. 47 1 1. 16 1. 24 0, 5 0, 4 0, 3 0, 2 0, 5 0. 23 0, 1 0 0. 12 0 BVS DEB 0. 24 EES
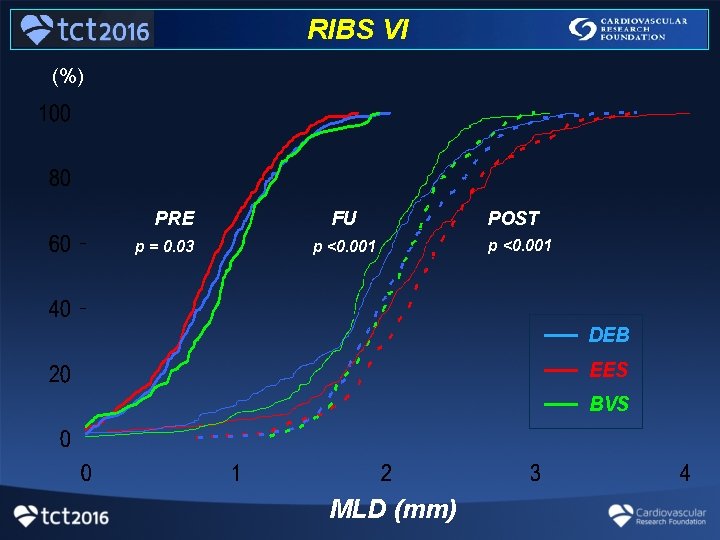
RIBS VI (%) PRE p = 0. 03 FU p <0. 001 POST p <0. 001 __ __ __ MLD (mm) DEB EES BVS
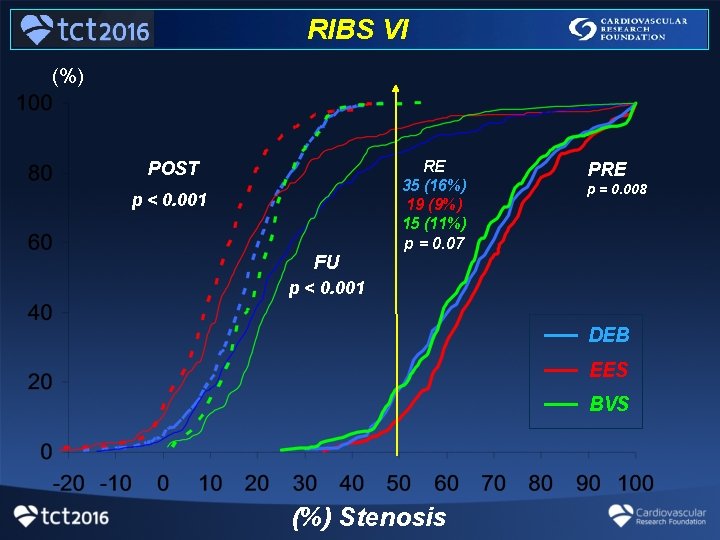
RIBS VI (%) RE 35 (16%) 19 (9%) 15 (11%) p = 0. 07 POST p < 0. 001 PRE p = 0. 008 FU p < 0. 001 __ __ __ (%) Stenosis DEB EES BVS

RIBS VI On multivariate analysis, after adjusting for all potential confounders: - MLD at FU was significantly smaller - % DS at FU was significantly larger after BVS compared with EES treatment
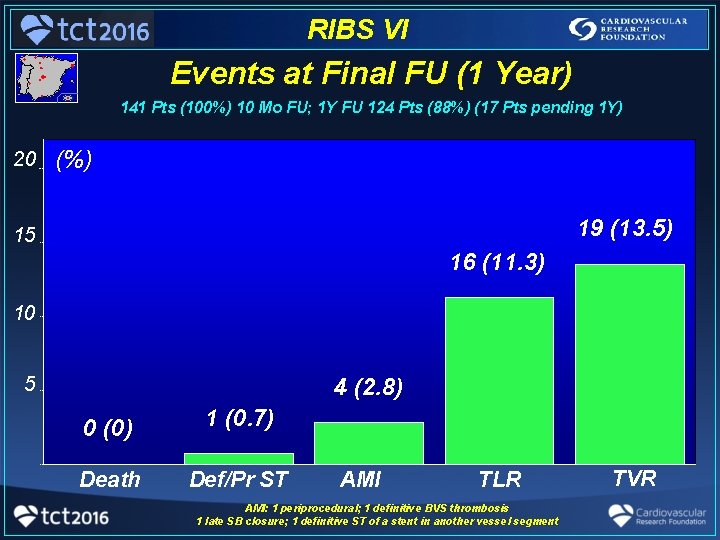
RIBS VI Events at Final FU (1 Year) 141 Pts (100%) 10 Mo FU; 1 Y FU 124 Pts (88%) (17 Pts pending 1 Y) 20 15 20 (%) 19 (13. 5) 15 16 (11. 3) 10 5 10 4 (2. 8) 5 0 (0) 1 (0. 7) Death Def/Pr ST 0 AMI TLR AMI: 1 periprocedural; 1 definitive BVS thrombosis 1 late SB closure; 1 definitive ST of a stent in another vessel segment TVR
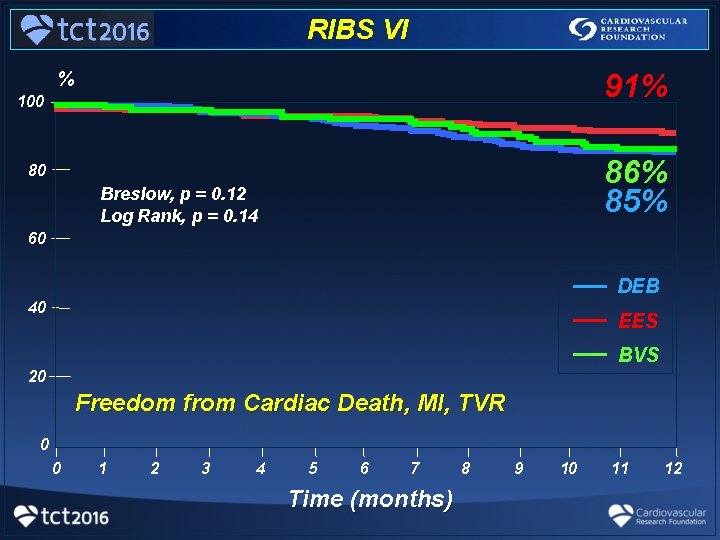
RIBS VI % 91% 100 86% 85% 80 Breslow, p = 0. 12 Log Rank, p = 0. 14 60 __ __ __ 40 DEB EES BVS 20 Freedom from Cardiac Death, MI, TVR 0 0 1 2 3 4 5 6 7 Time (months) 8 9 10 11 12
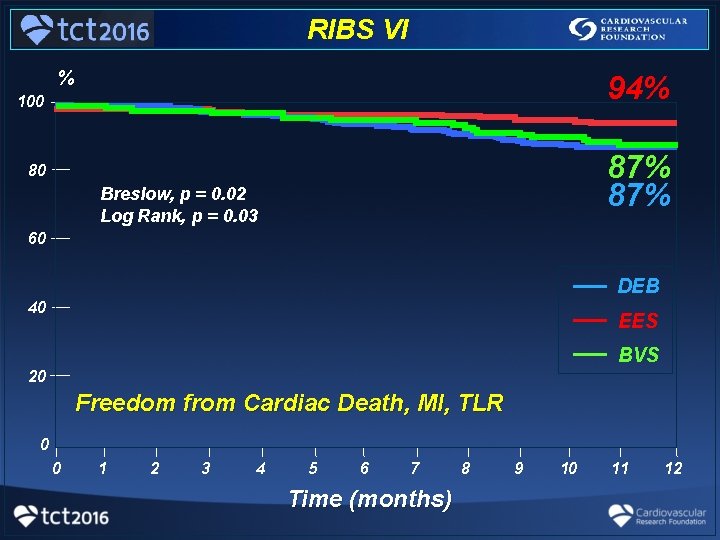
RIBS VI % 94% 100 87% 80 Breslow, p = 0. 02 Log Rank, p = 0. 03 60 __ __ __ 40 DEB EES BVS 20 Freedom from Cardiac Death, MI, TLR 0 0 1 2 3 4 5 6 7 Time (months) 8 9 10 11 12
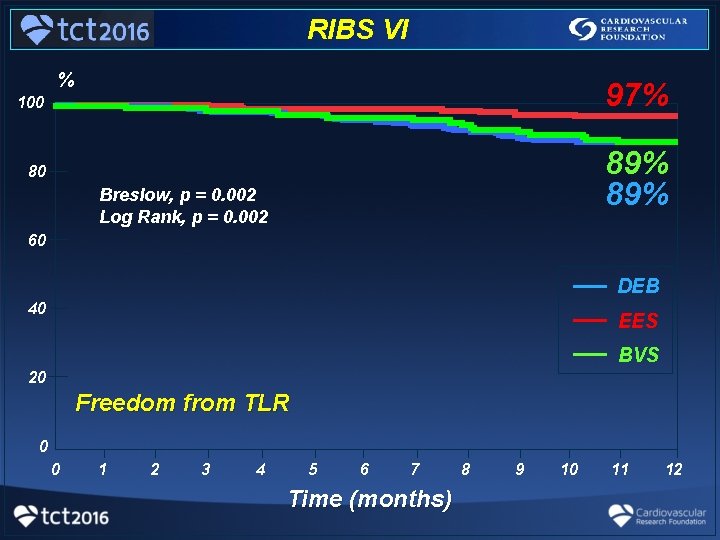
RIBS VI % 97% 100 89% 80 Breslow, p = 0. 002 Log Rank, p = 0. 002 60 __ __ __ 40 DEB EES BVS 20 Freedom from TLR 0 0 1 2 3 4 5 6 7 Time (months) 8 9 10 11 12

RIBS VI Conclusions: BVS are safe and effective in the treatment of selected patients with ISR BVS provide favorable late angiographic (restenosis rate 11%) and clinical results (TLR 11%) in these patients The acute and late angiographic findings of BVS appear to be similar to those obtained with DEB (“leave nothing behind strategy”) but poorer than those seen after EES implantation (caution required as historical controls from RCT were used) Further studies with long-term follow-up will be required to elucidate the relative value of BVS vs other well-established therapeutic strategies in this challenging setting
- Slides: 22