Revolutionizing regenerative medicine by reversing cardiac damage with

“Revolutionizing regenerative medicine by reversing cardiac damage with the growth of new heart muscle cells” www. ventrinovainc. com 1

EXECUTIVE SUMMARY - COMPANY Ventri. Nova is a regenerative medicine company focused on reversing cardiac damage by activation of the intrinsic repair pathway via Cyclin A 2 to generate de novo heart muscle cells • The company’s therapeutic approach is based on the role of cyclin A 2 in mediating cardiac muscle cell mitosis and cardiac repair – A key development genetic switch whose role in cardiac tissue development was discovered by Dr. Chaudhry • Recent publication in Science Translational Medicine provides in vivo proof-ofconcept of Ventri. Nova’s initial gene therapy product “Cyclin A 2 Induces Cardiac Regeneration After Myocardial Infarction Through Cytokinesis of Adult Cardiomyocytes” SD Shapiro, AK Ranjan et al, . Science Translational Medicine, 19 February 2014 • Ventri. Nova was selected as one of 50 most innovative companies in healthcare globally to present at the “Hive” at TEDMED 2013 2

MI AND HEART FAILURE – A SIGNIFICANT UNMET MEDICAL NEED Significant unmet medical need and growing • • In 2009, 600, 000 people in the U. S. died of heart disease— 1 in 4 deaths 1 In 2012, about 715, 000 Americans had a heart attack. Of these, 525, 000 were a first heart attack and 190, 000 happen in people who have already had a heart attack 2 Coronary heart disease alone costs the United States $108. 9 billion each year. This total includes the cost of health care services, medications, and lost productivity 3 There are 5 million Americans living with congestive heart failure today. There were 1 million hospitalizations for congestive heart failure in the U. S. between 2000 and 2010 4 1. Kochanek KD, Xu JQ, Murphy SL, Miniño AM, Kung HC Deaths: final data for 2009. [PDF-2 M] National vital statistics reports. 2011; 60(3). 2. Go AS, Mozaffarian D, Roger VL, Benjamin EJ, Berry JD, et al. American Heart Association Statistics Committee and Stroke Statistics Subcommittee. Heart disease and stroke statistics— 2013 update: a report from the American Heart Association. Circulation. 2013 Jan 1; 127(1): e 6 -e 245. http: //circ. ahajournals. org/content/127/1/e 6. long 3. Heidenreich PA, Trogdon JG, Khavjou OA, et al. Forecasting the future of cardiovascular disease in the United States: a policy statement from the American Heart Association. Circulation. 2011; 123: 933 -44. Epub 2011 Jan 24. 4. American Heart Association and Hall, MJ, Leveant S, And De. Frances, CJ. Hospitalization for Congestive Heart Failure. NCHS Data Brief, October 2012 3

EXECUTIVE SUMMARY – CYCLIN A 2 TECHNOLOGY • Cyclin A 2 is the key switch that mediates cardiac cell division and tissue growth during neonatal development. The gene is turned off in the adult and therefore unavailable to promote cellular regeneration and tissue repair following an injury • Ventri. Nova’s therapeutic strategy is centered around the development of gene therapy related technologies to reactivate the cyclin A 2 gene and promote tissue repair following a heart attack • Pre-clinical work has demonstrated that modulation of the cyclin A 2 gene stimulates endogenous growth of new myocytes (heart muscle cells) and significantly enhances cardiac contractile function in both small and large animals 4
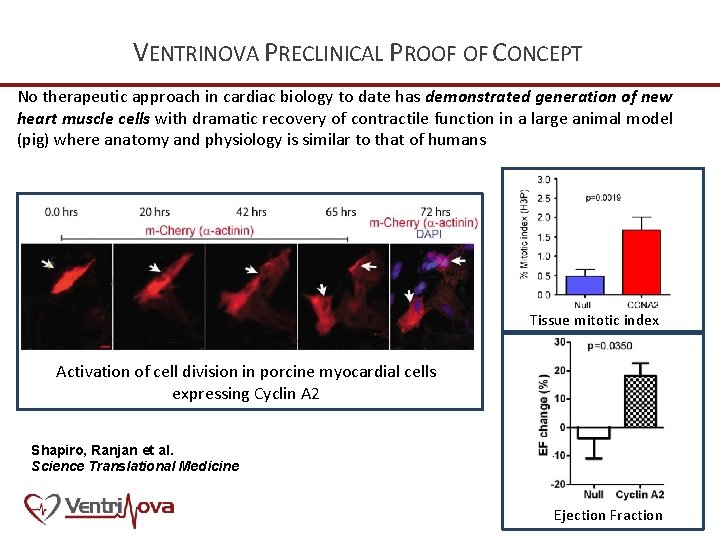
VENTRINOVA PRECLINICAL PROOF OF CONCEPT No therapeutic approach in cardiac biology to date has demonstrated generation of new heart muscle cells with dramatic recovery of contractile function in a large animal model (pig) where anatomy and physiology is similar to that of humans Tissue mitotic index Activation of cell division in porcine myocardial cells expressing Cyclin A 2 Shapiro, Ranjan et al. Science Translational Medicine Ejection Fraction
“Cyclin A 2 Induces Cardiac Regeneration After Myocardial Infarction Through Cytokinesis of Adult Cardiomyocytes” SD Shapiro, AK Ranjan et al. , Science Translational Medicine, 19 February 2014 http: //www. scientificamerican. com/article/gene-therapy-shows-promise-for-treating-heart-attack-victims/ http: //blog. tedmed. com/? p=4788 Heart cells: no longer undivided http: //www. nature. com/scibx/journal/v 7/n 9/full/scibx. 2014. 244. html http: //www. fiercebiotechresearch. com/story/gene-therapy-regenerates-damaged-tissue-pigs-after-heart-attack 6

VENTRINOVA LEAD DEVELOPMENT CANDIDATE: VN-100 Late Stage Preclinical Development VN-100, the company's lead candidate, is a viral vector-based gene therapy that induces cardiomyocyte division in adult heart tissue via in situ delivery of the cyclin A 2 gene – the principal gene regulating cell cycle activity. Ventri. Nova’s lead candidate is VN-100 • Targeted adenoviral delivery of human cyclin A 2 to cardiomyocytes through the use of a cardiomyocyte-specific promoter • Product is delivered by intra-coronary or direct injection to infarcted area • Proof of concept studies in both small and large animals models have demonstrated the efficacy of a single treatment shortly after a MI, without any noted long-term side effects 7
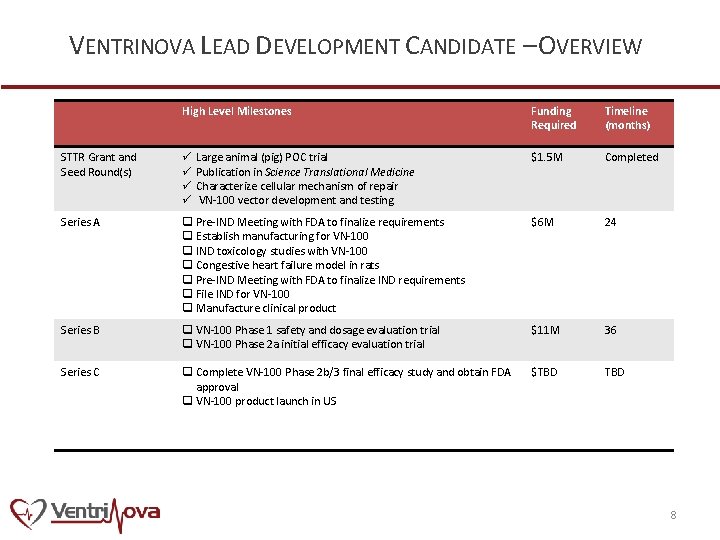
VENTRINOVA LEAD DEVELOPMENT CANDIDATE – OVERVIEW High Level Milestones Funding Required Timeline (months) STTR Grant and Seed Round(s) ü Large animal (pig) POC trial ü Publication in Science Translational Medicine ü Characterize cellular mechanism of repair ü VN-100 vector development and testing $1. 5 M Completed Series A q Pre-IND Meeting with FDA to finalize requirements q Establish manufacturing for VN-100 q IND toxicology studies with VN-100 q Congestive heart failure model in rats q Pre-IND Meeting with FDA to finalize IND requirements q File IND for VN-100 q Manufacture clinical product $6 M 24 Series B q VN-100 Phase 1 safety and dosage evaluation trial q VN-100 Phase 2 a initial efficacy evaluation trial $11 M 36 Series C q Complete VN-100 Phase 2 b/3 final efficacy study and obtain FDA approval q VN-100 product launch in US $TBD 8
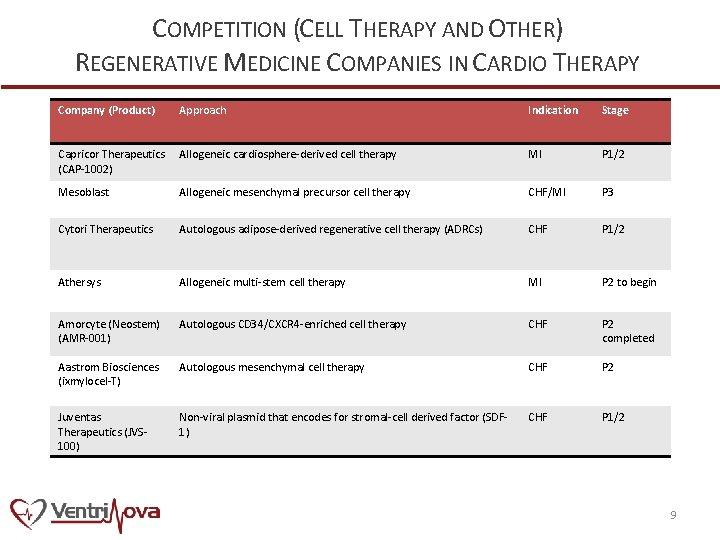
COMPETITION (CELL THERAPY AND OTHER) REGENERATIVE MEDICINE COMPANIES IN CARDIO THERAPY Company (Product) Approach Indication Stage Capricor Therapeutics (CAP-1002) Allogeneic cardiosphere-derived cell therapy MI P 1/2 Mesoblast Allogeneic mesenchymal precursor cell therapy CHF/MI P 3 Cytori Therapeutics Autologous adipose-derived regenerative cell therapy (ADRCs) CHF P 1/2 Athersys Allogeneic multi-stem cell therapy MI P 2 to begin Amorcyte (Neostem) (AMR-001) Autologous CD 34/CXCR 4 -enriched cell therapy CHF P 2 completed Aastrom Biosciences (ixmylocel-T) Autologous mesenchymal cell therapy CHF P 2 Juventas Therapeutics (JVS 100) Non-viral plasmid that encodes for stromal-cell derived factor (SDF 1) CHF P 1/2 9
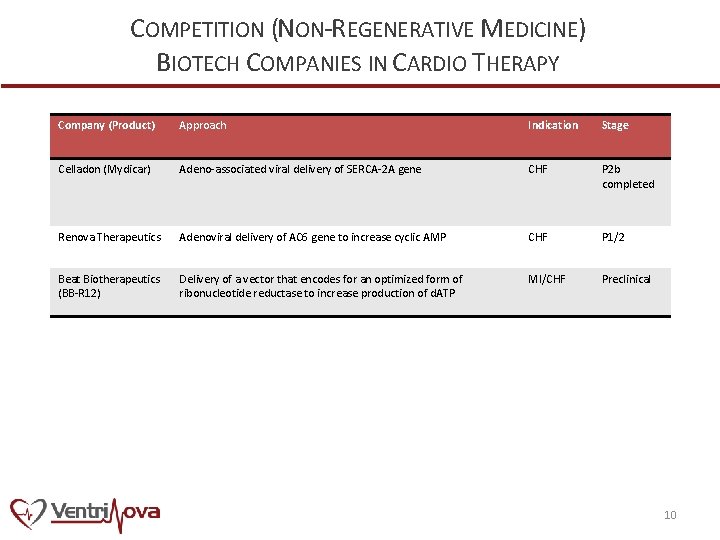
COMPETITION (NON-REGENERATIVE MEDICINE) BIOTECH COMPANIES IN CARDIO THERAPY Company (Product) Approach Indication Stage Celladon (Mydicar) Adeno-associated viral delivery of SERCA-2 A gene CHF P 2 b completed Renova Therapeutics Adenoviral delivery of AC 6 gene to increase cyclic AMP CHF P 1/2 Beat Biotherapeutics (BB-R 12) Delivery of a vector that encodes for an optimized form of ribonucleotide reductase to increase production of d. ATP MI/CHF Preclinical 10

EXECUTIVE SUMMARY – SERIES A FINANCING • Ventri. Nova’s cash position as of 5/1/14 from financing to date: $400, 000 Previous financing includes: • $1 million convertible debt investment from Broadview Ventures • $500, 000 in NIH grant financing • Ventri. Nova seeks to raise Series A financing of $6 million to achieve the following important milestones and file an IND for its first product (VN-100) with the FDA in the next 24 months: • • • Complete Pre-IND meeting with the FDA Establish c. GMP manufacturing for VN-100 with a CMO Complete IND-enabling toxicology studies of VN-100 File an IND for VN-100 in MI Complete rat study of VN-100 in CHF model Raise Series B financing for clinical trials 11
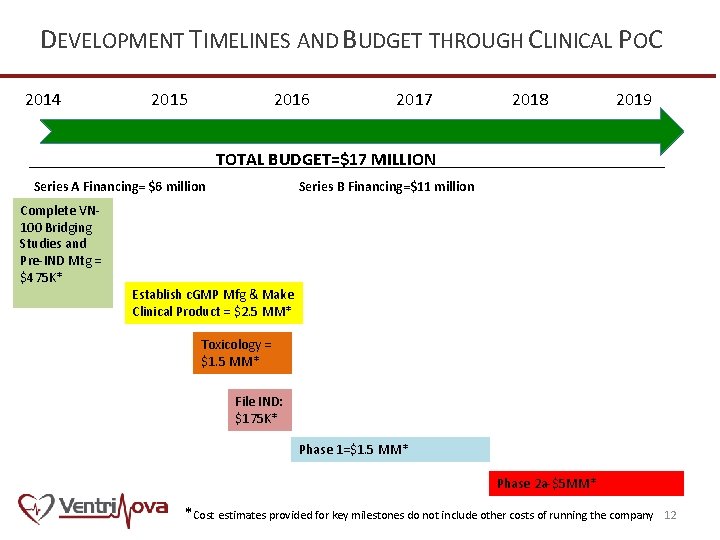
DEVELOPMENT TIMELINES AND BUDGET THROUGH CLINICAL POC 2014 2016 2015 2017 2018 2019 TOTAL BUDGET=$17 MILLION ____________________________________ Series A Financing= $6 million Complete VN 100 Bridging Studies and Pre-IND Mtg = $475 K* Series B Financing=$11 million Establish c. GMP Mfg & Make Clinical Product = $2. 5 MM* Toxicology = $1. 5 MM* File IND: $175 K* Phase 1=$1. 5 MM* Phase 2 a-$5 MM* *Cost estimates provided for key milestones do not include other costs of running the company 12

VENTRINOVA IP SUMMARY Issued Patents Filing Date 5/18/2004 Compositions and Methods for Treating and Preventing Heart Tissue Degeneration and Uses Thereof United States 11/4/2005 Compositions and Methods for Treating and Preventing Heart Tissue Degeneration and Uses Thereof United States Pending Patents Filing Date 5/18/2004 Compositions and Methods for Treating and Preventing Heart Tissue Degeneration and Uses Thereof EPO 5/18/2004 Compositions and Methods for Treating and Preventing Heart Tissue Degeneration and Uses Thereof Canada 5/18/2004 Compositions and Methods for Treating and Preventing Heart Tissue Degeneration and Uses Thereof Hong Kong 12/18/2009 Compositions and Methods for Treating and Preventing Heart Tissue Degeneration and Uses Thereof United States Confidential - Not for Distribution 13

THE TEAM Hina Chaudhry, MD – Founder, CSO and Chairman • Associate Professor of Medicine and Director of Cardiovascular Regenerative Medicine, Mount Sinai School of Medicine, New York • B. S. degrees in both Chemistry and Biology from MIT and MD with Honors from Harvard. • Recipient of multiple awards - AHA Clinical Scientist Award, Irving Scholar Award from Columbia University and TEDMED 2013 Innovation Scholar. Howard B. Johnson – CEO • Bioscience investor and entrepreneur with over 30 years experience in operations, finance, investor relations and venture capital. • Began career as investment banker working on large product development financings for leading biotechs: Genentech, Amgen, Centocor and Genzyme. • Founding investor and board member Acorda Therapeutics. • More recently, President and CFO of oncology drug development company that advanced from filing an IND to filing an NDA for its lead product for acute myelogenous leukemia. Managed regulatory affairs, manufacturing, business development and pre-commercial operations, as well as finance, investor relations, legal, human resources and IT. Raised $140 million in financing. Christopher de Souza, Ph. D – Broadview Venture Advisor • Shares responsibility for the day-to-day operations of Broadview Ventures, including business development strategy, identification and screening of new investment opportunities, negotiation of partnering terms and deal structure, as well as portfolio company board involvement. • Leverages 20 years of experience in biopharmaceutical R&D, business development and corporate strategy at Novartis and Skye. Pharma. 14

SCIENTIFIC ADVISORY BOARD Debra Wolgemuth, Ph. D. • Professor, Department of Genetics & Development and Obstetrics & Gynecology, The Center for Reproductive Sciences, and The Institute of Human Nutrition, Columbia University • Co-inventor of the use of cyclin A 2 modulation in cardiovascular disease. • Internationally recognized investigator in developmental biology and genetics. • Discovered mammalian cyclin A 1 and its role in reproductive tissues/diseases and oncology. Judith Swain, M. D. • Executive Director, Singapore Institute for Clinical Sciences and the Lien Ying Chow Professor of Medicine at the Yong Loo Lin School of Medicine, National University of Singapore. • Previously - Dean for Translational Medicine at the University of California, San Diego. • Chair of the Department of Medicine at Stanford University and Arthur Bloomfield and George E. Becker Professorships; Herbert C. Rorer Professor of Medical Sciences and Director of Cardiovascular Medicine, University of Pennsylvania. Roxana Mehran, M. D. , FACC, FACP, FCCP, FESC, FAHA, FSCAI • Professor of Medicine (Cardiology) and Health Evidence and Policy, Director of Interventional Cardiovascular Research and Clinical Trials at Mount Sinai School of Medicine, New York. • Internationally recognized for her work in multicenter, multinational clinical trials, specializing in complex data analysis. • Previously Associate Professor of Medicine and Director of Outcomes Research, Data Coordinating and Analysis at the Center of Interventional Vascular Therapies at Columbia University. 15
- Slides: 15