Revision of Chapter Six Chapter Six 1 Complete




















- Slides: 21

Revision of Chapter Six

Chapter Six 1. Complete and balance the following redox equation. When properly balanced using the smallest whole-number coefficients, the coefficient of S is H 2 S + HNO 3 S + NO (acidic solution) a) 1 +1 x 2 -2 b) 2 d) 5 e) 6 H 2 S + HNO 3 → S +NO 0 oxidation H 2 S → S +2 e +1 +5 -2 x 3 c) 3 H 2 S → S +2 e + 2 H+ +2 -2 reduction 3 e+ HNO 3 → NO x 3 3 H++ 3 e+HNO 3 → NO + 2 H 2 O 3 H 2 S → 3 S + 6 e + 6 H+ x 2 6 H+ + 6 e + 2 HNO 3 → 2 NO + 4 H 2 O 3 H 2 S + 2 HNO 3→ 3 S + 2 NO + 4 H 2 O

Chapter Six 1. Complete and balance the following redox equation. When properly balanced using the smallest whole-number coefficients, the coefficient of S is H 2 S + HNO 3 S + NO (acidic solution) a) 1 2. b) 2 c) 3 d) 5 e) 6 Given the following notation for an electrochemical cell Pt(s) | H 2(g) | H+(aq) || Ag+(aq) | Ag(s), what is the balanced overall (net) cell reaction? a) 2 H+(aq) + 2 Ag+(aq) H 2(g) + 2 Ag(s) b) H 2(g) + 2 Ag(s) H+(aq) + 2 Ag+(aq) c) 2 H+(aq) + 2 Ag(s) H 2(g) + 2 Ag+(aq) d) H 2(g) + Ag+(aq) H+(aq) + Ag(s) e) H 2(g) + 2 Ag+(aq) 2 H+(aq) + 2 Ag(s) H 2(g)→ 2 H+ (aq) + 2 e+ 2 Ag+(aq) → 2 Ag (s) H 2(g) + 2 Ag+(aq)→ 2 H+(aq) + 2 Ag(s)

Chapter Six 1. Complete and balance the following redox equation. When properly balanced using the smallest whole-number coefficients, the coefficient of S is H 2 S + HNO 3 S + NO (acidic solution) a) 1 2. b) 2 c) 3 d) 5 e) 6 Given the following notation for an electrochemical cell Pt(s) | H 2(g) | H+(aq) || Ag+(aq) | Ag(s), what is the balanced overall (net) cell reaction? a) 2 H+(aq) + 2 Ag+(aq) H 2(g) + 2 Ag(s) b) H 2(g) + 2 Ag(s) H+(aq) + 2 Ag+(aq) c) 2 H+(aq) + 2 Ag(s) H 2(g) + 2 Ag+(aq) d) H 2(g) + Ag+(aq) H+(aq) + Ag(s) e) H 2(g) + 2 Ag+(aq) 2 H+(aq) + 2 Ag(s) 3. Calculate the value of E cell for the following reaction: 2 Au(s) + 3 Ca 2+(aq) 2 Au 3+(aq) + 3 Ca(s) a) -4. 37 V b) -1. 37 V c) -11. 6 V d) 1. 37 V e) 4. 37 V

Chapter Six 3. Calculate the value of E cell for the following reaction: 2 Au(s) + 3 Ca 2+(aq) 2 Au 3+(aq) + 3 Ca(s) a) -4. 37 V b) -1. 37 V c) -11. 6 V 2 Au(s)→ 2 Au+3 (aq) + 6 e E°= -1. 50 6 e + 3 Ca+2(aq) → 3 Ca(s) E°=-2. 87 d) 1. 37 V e) 4. 37 V

Chapter Six 1. Complete and balance the following redox equation. When properly balanced using the smallest whole-number coefficients, the coefficient of S is H 2 S + HNO 3 S + NO (acidic solution) a) 1 2. b) 2 c) 3 d) 5 e) 6 Given the following notation for an electrochemical cell Pt(s) | H 2(g) | H+(aq) || Ag+(aq) | Ag(s), what is the balanced overall (net) cell reaction? a) 2 H+(aq) + 2 Ag+(aq) H 2(g) + 2 Ag(s) b) H 2(g) + 2 Ag(s) H+(aq) + 2 Ag+(aq) c) 2 H+(aq) + 2 Ag(s) H 2(g) + 2 Ag+(aq) d) H 2(g) + Ag+(aq) H+(aq) + Ag(s) e) H 2(g) + 2 Ag+(aq) 2 H+(aq) + 2 Ag(s) 3. Calculate the value of E cell for the following reaction: 2 Au(s) + 3 Ca 2+(aq) 2 Au 3+(aq) + 3 Ca(s) a) -4. 37 V b) -1. 37 V c) -11. 6 V d) 1. 37 V e) 4. 37 V

Chapter Six 4. Calculate the standard cell emf for the following cell: Mg |Mg 2+ || NO 3 - (acid soln)| NO(g) | Pt a) 3. 33 V b) 1. 41 V Mg(s)→ Mg+2 (aq) + 2 e 2 e + NO 3 -(aq) → NO(g) E°= -2. 37 E°= 0. 96 c) -1. 41 V d) 8. 46 V e) -8. 46 V

Chapter Six 4. Calculate the standard cell emf for the following cell: Mg |Mg 2+ || NO 3 - (acid soln)| NO(g) | Pt a) 3. 33 V 5. b) 1. 41 V c) -1. 41 V d) 8. 46 V e) -8. 46 V The overall reaction 2 Co 3+(aq) + 2 Cl-(aq) 2 Co 2+(aq) + Cl 2(g) has the standard cell voltage E cell= 0. 46 V. Given that Cl 2(g) + 2 e- 2 Cl-(aq), E = 1. 36 V, calculate the standard reduction potential for the following the half reaction at 25 C: Co 3+ + e- Co 2+ a) 1. 82 V b) -0. 90 V c) 0. 90 V d) -1. 82 V 2 Co 3+(aq) + 2 Cl-(aq) 2 Co 2+(aq) + Cl 2(g) 2 Cl-(aq)→ Cl 2 (aq) + 2 e 2 e +2 Co+3(aq) → 2 Co+2(s) - e) -1. 36 V

Chapter Six 4. Calculate the standard cell emf for the following cell: Mg |Mg 2+ || NO 3 - (acid soln)| NO(g) | Pt a) 3. 33 V 5. c) -1. 41 V d) 8. 46 V e) -8. 46 V The overall reaction 2 Co 3+(aq) + 2 Cl-(aq) 2 Co 2+(aq) + Cl 2(g) has the standard cell voltage E cell= 0. 46 V. Given that Cl 2(g) + 2 e- 2 Cl-(aq), E = 1. 36 V, calculate the standard reduction potential for the following the half reaction at 25 C: Co 3+ + e- Co 2+ a) 1. 82 V 6. b) 1. 41 V b) -0. 90 V c) 0. 90 V d) -1. 82 V e) -1. 36 V In the following half equation, which is the oxidizing agent? NO 3 -(aq) + 4 H+(aq) + 3 e- NO(g) + 2 H 2 O a) NO 3 - b) H+ c) e+5 -2 x 3 +1 d) NO +2 -2 NO 3 -(aq) + 4 H+(aq) + 3 e- NO(g) + 2 H 2 O e) H 2 O

Chapter Six 4. Calculate the standard cell emf for the following cell: Mg |Mg 2+ || NO 3 - (acid soln)| NO(g) | Pt a) 3. 33 V 5. d) 8. 46 V e) -8. 46 V b) -0. 90 V c) 0. 90 V d) -1. 82 V e) -1. 36 V In the following half equation, which is the oxidizing agent? NO 3 -(aq) + 4 H+(aq) + 3 e- NO(g) + 2 H 2 O a) NO 37. c) -1. 41 V The overall reaction 2 Co 3+(aq) + 2 Cl-(aq) 2 Co 2+(aq) + Cl 2(g) has the standard cell voltage E cell= 0. 46 V. Given that Cl 2(g) + 2 e- 2 Cl-(aq), E = 1. 36 V, calculate the standard reduction potential for the following the half reaction at 25 C: Co 3+ + e- Co 2+ a) 1. 82 V 6. b) 1. 41 V b) H+ c) e- d) NO e) H 2 O Which statement is true for a spontaneous redox reaction carried out at standard-state conditions? a) E red is always negative. b) E cell is always positive c) E ox is always positive d) E red is always positive

Chapter Six 8. Consider the following standard reduction potentials in acid solution: The strongest oxidation agent among those shown above is a) Fe 3+ 9. b) Fe 2+ c) Br - d) Al 3+ e) Al Determine the equilibrium constant, Keq, at 25 C for the reaction: 2 Br- (aq) + I 2(s) a) 5. 7 10 -19 b) 18. 30 c) 1. 7 1054 d) 1. 9 1018 e) 5. 7 10 -55 Br 2(l) + 2 I- (aq)

Chapter Six 9. Determine the equilibrium constant, Keq, at 25 C for the reaction: 2 Br- (aq) + I 2(s) a) 5. 7 10 -19 b) 18. 30 c) 1. 7 1054 2 Br - (aq)→ Br 2(l) + 2 e E°= -1. 07 2 e + I 2(S) → 2 I -(aq) E°=0. 53 E°= -0. 54 , n= 2, T=25+273=298, R=8. 314, F=96500 d) 1. 9 1018 e) 5. 7 10 -55 Br 2(l) + 2 I- (aq)

Chapter Six Consider the following standard reduction potentials in acid solution: 8. The strongest reducing agent among those shown above is a) Fe 3+ b) Fe 2+ c) Br - d) Al 3+ e) Al Determine the equilibrium constant, Keq, at 25 C for the reaction: 2 Br- (aq) + I 2(s) 9. a) 5. 7 10 -19 10. b) 18. 30 c) 1. 7 1054 d) 1. 9 1018 e) 5. 7 10 -55 d) 5. 1 1013 e) None of these Given the following standard reduction potentials, Ag+(aq) + e → Ag(s) Ag. CN(s) + e → Ag(s)+ CN- (aq) E°=0. 80 V E°=-0. 01 V Calculate the solubility product of Ag. CN at 25 C a) 4. 3 10 -14 b) 2. 3 1013 c) 2. 1 10 -14 Br 2(l) + 2 I- (aq)

Chapter Six 10. Given the following standard reduction potentials, Ag+(aq) + e → Ag(s) E°=0. 80 V Ag. CN(s) + e → Ag(s)+ CN- (aq) E°=-0. 01 V Calculate the solubility product of Ag. CN at 25 C a) 4. 3 10 -14 b) 2. 3 1013 c) 2. 1 10 -14 Ag+(aq) + e → Ag(s) E°=0. 80 V Ag. CN(s) + e → Ag(s)+ CN (aq) E°= - 0. 01 V Ag(s) → Ag+(aq) + e E°= - 0. 80 V Ag. CN(s) + e → Ag(s)+ CN (aq) E°= - 0. 01 V Ag. CN(s) → Ag+(aq)+ CN- (aq) d) 5. 1 1013 e) None of these E°= -0. 81 , n= 1, T=25+273=298, R=8. 314, F=96500

Chapter Six Consider the following standard reduction potentials in acid solution: 8. The strongest reducing agent among those shown above is a) Fe 3+ b) Fe 2+ c) Br - d) Al 3+ e) Al Determine the equilibrium constant, Keq, at 25 C for the reaction: 2 Br- (aq) + I 2(s) 9. a) 5. 7 10 -19 10. b) 18. 30 c) 1. 7 1054 d) 1. 9 1018 e) 5. 7 10 -55 d) 5. 1 1013 e) None of these Given the following standard reduction potentials, Ag+(aq) + e → Ag(s) Ag. CN(s) + e → Ag(s)+ CN- (aq) E°=0. 80 V E°=-0. 01 V Calculate the solubility product of Ag. CN at 25 C a) 4. 3 10 -14 b) 2. 3 1013 c) 2. 1 10 -14 Br 2(l) + 2 I- (aq)

Chapter Six 11. Calculate the cell emf for the following reaction at 25 C: 2 Ag+(0. 010 M) + H 2(1 atm) 2 Ag(s) + 2 H+(p. H = 10. 0) a)1. 04 V H 2(g)→ 2 H+ (aq) + 2 e b) 1. 27 V c) 0. 92 V d) 0. 56 V E°= 0. 0 2 e+ 2 Ag+(aq) → 2 Ag (s) E°=0. 80 [H+]=10 -ph H 2(g) + 2 Ag+(aq)→ 2 H+(aq) + 2 Ag(s) E°= 0. 80 , n= 2, T=25+273=298, R=8. 314, F=96500 e) 0. 80 V

Chapter Six Calculate the cell emf for the following reaction at 25 C: 2 Ag+(0. 010 M) + H 2(1 atm) 2 Ag(s) + 2 H+(p. H = 10. 0) 11. a)1. 04 V 12. b) 1. 27 V c) 0. 92 V d) 0. 56 V e) 0. 80 V How many coulombs of charge are required to cause reduction of 0. 20 mole of Cr 3+ to Cr? a) 0. 60 C b) 3. 0 C c) 2. 9 104 C d) 5. 8 104 C e) 9. 65 104 C 3 e +Cr+3 → Cr 3 e ----- 1 mole Cr 3 x 96500 C ---- 1 mol Cr ? C ---- 0. 2 mol Cr Charge = 3 x 96500 x 0. 2 = 5. 8 x 104 C

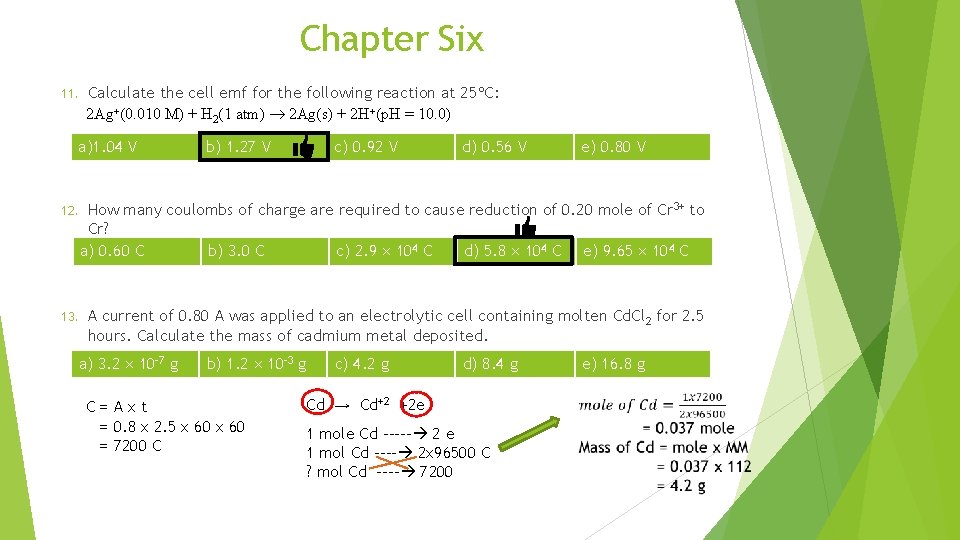
Chapter Six 11. Calculate the cell emf for the following reaction at 25 C: 2 Ag+(0. 010 M) + H 2(1 atm) 2 Ag(s) + 2 H+(p. H = 10. 0) a)1. 04 V b) 1. 27 V c) 0. 92 V d) 0. 56 V e) 0. 80 V 12. How many coulombs of charge are required to cause reduction of 0. 20 mole of Cr 3+ to Cr? a) 0. 60 C b) 3. 0 C c) 2. 9 104 C d) 5. 8 104 C e) 9. 65 104 C 13. A current of 0. 80 A was applied to an electrolytic cell containing molten Cd. Cl 2 for 2. 5 hours. Calculate the mass of cadmium metal deposited. a) 3. 2 10 -7 g b) 1. 2 10 -3 g C=Axt = 0. 8 x 2. 5 x 60 = 7200 C c) 4. 2 g d) 8. 4 g Cd → Cd+2 +2 e 1 mole Cd ----- 2 e 1 mol Cd ---- 2 x 96500 C ? mol Cd ---- 7200 e) 16. 8 g

Chapter Six 11. Calculate the cell emf for the following reaction at 25 C: 2 Ag+(0. 010 M) + H 2(1 atm) 2 Ag(s) + 2 H+(p. H = 10. 0) a)1. 04 V b) 1. 27 V c) 0. 92 V d) 0. 56 V e) 0. 80 V 12. How many coulombs of charge are required to cause reduction of 0. 20 mole of Cr 3+ to Cr? a) 0. 60 C b) 3. 0 C c) 2. 9 104 C d) 5. 8 104 C e) 9. 65 104 C 13. A current of 0. 80 A was applied to an electrolytic cell containing molten Cd. Cl 2 for 2. 5 hours. Calculate the mass of cadmium metal deposited. a) 3. 2 10 -7 g 14. b) 1. 2 10 -3 g c) 4. 2 g d) 8. 4 g e) 16. 8 g A current of 2. 50 A was passed through an electrolytic cell containing molten Ca. Cl 2 for 4. 50 hours. How many moles of calcium metal should be deposited? a) 5. 83 10 -5 mol b) 0. 210 mol c) 0. 420 mol d) 0. 840 mol e) 1. 95 109 mol

Chapter Six 14. A current of 2. 50 A was passed through an electrolytic cell containing molten Ca. Cl 2 for 4. 50 hours. How many moles of calcium metal should be deposited? a) 5. 83 10 -5 mol b) 0. 210 mol C=Axt = 2. 5 x 4. 5 x 60 = 40500 C c) 0. 420 mol d) 0. 840 mol e) 1. 95 109 mol Ca → Ca+2 +2 e 1 mole Ca ----- 2 e 1 mol Ca ---- 2 x 96500 C ? mol Ca ---- 40500

Chapter Six 11. Calculate the cell emf for the following reaction at 25 C: 2 Ag+(0. 010 M) + H 2(1 atm) 2 Ag(s) + 2 H+(p. H = 10. 0) a)1. 04 V b) 1. 27 V c) 0. 92 V d) 0. 56 V e) 0. 80 V 12. How many coulombs of charge are required to cause reduction of 0. 20 mole of Cr 3+ to Cr? a) 0. 60 C b) 3. 0 C c) 2. 9 104 C d) 5. 8 104 C e) 9. 65 104 C 13. A current of 0. 80 A was applied to an electrolytic cell containing molten Cd. Cl 2 for 2. 5 hours. Calculate the mass of cadmium metal deposited. a) 3. 2 10 -7 g 14. b) 1. 2 10 -3 g c) 4. 2 g d) 8. 4 g e) 16. 8 g A current of 2. 50 A was passed through an electrolytic cell containing molten Ca. Cl 2 for 4. 50 hours. How many moles of calcium metal should be deposited? a) 5. 83 10 -5 mol b) 0. 210 mol c) 0. 420 mol d) 0. 840 mol e) 1. 95 109 mol