Revision L O to summarise biochemical tests to

Revision! L. O: to summarise biochemical tests to apply knowledge to questions

Biochemical Tests are used to indicate the presence / absence of biological molecules… • http: //www. bbc. co. uk/schools/gcsebitesize/science/add_oc r/chemicals/biologicalcompoundsact. shtml
Summary Table Biological Biochemical Molecule test Positive Result
Summary Table Biological Molecule Protein Lipid Reducing Sugar Non Reducing Sugar Biochemical test Positive Result
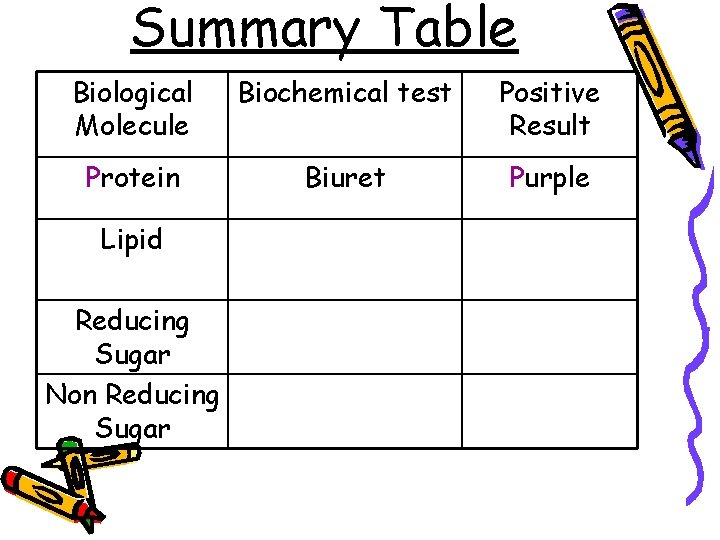
Summary Table Biological Molecule Biochemical test Positive Result Protein Biuret Purple Lipid Reducing Sugar Non Reducing Sugar
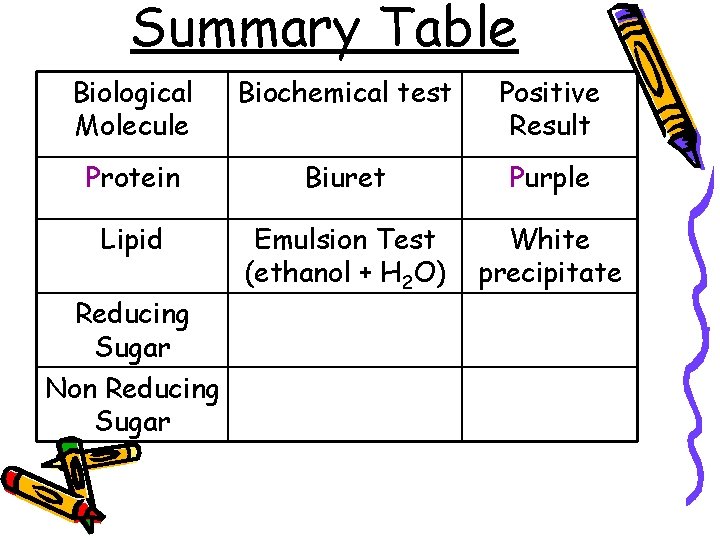
Summary Table Biological Molecule Biochemical test Positive Result Protein Biuret Purple Lipid Emulsion Test (ethanol + H 2 O) White precipitate Reducing Sugar Non Reducing Sugar
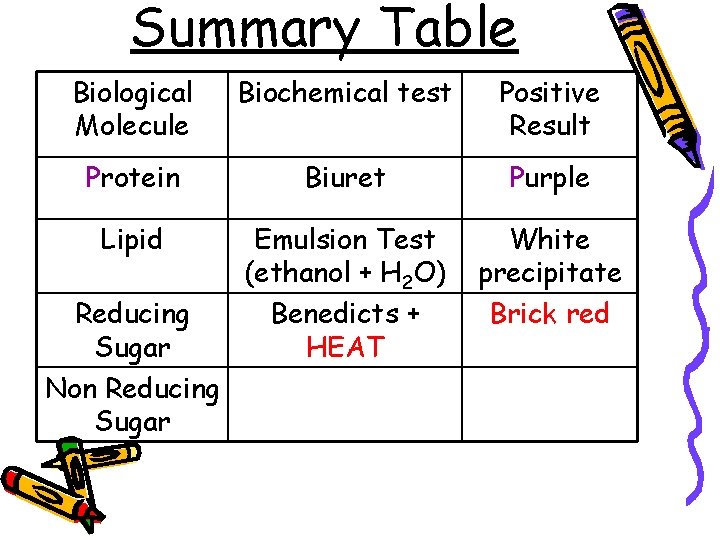
Summary Table Biological Molecule Biochemical test Positive Result Protein Biuret Purple Lipid Emulsion Test (ethanol + H 2 O) Benedicts + HEAT White precipitate Brick red Reducing Sugar Non Reducing Sugar
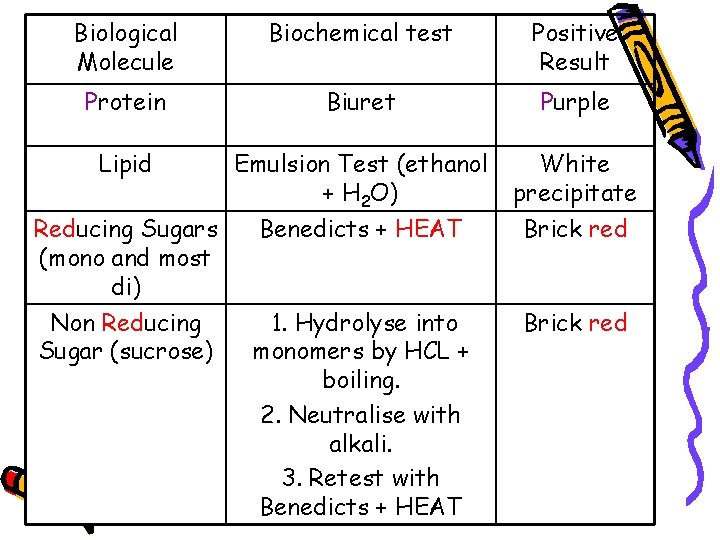
Biological Molecule Biochemical test Positive Result Protein Biuret Purple Lipid Emulsion Test (ethanol + H 2 O) White precipitate Benedicts + HEAT Brick red 1. Hydrolyse into monomers by HCL + boiling. 2. Neutralise with alkali. 3. Retest with Benedicts + HEAT Brick red Reducing Sugars (mono and most di) Non Reducing Sugar (sucrose)

Application of Knowledge • http: //www. classtools. net/education-games-php/timer

Question 1 a) A = Protein B = Lipid C = Reducing Sugar b) Hydrolyse with acid and then neutralise with alkali, re test with Benedicts c) Carbon, Hydrogen, Oxygen

Question 1 (again!) 1 a) C 12 H 22 O 11 (Because H 2 O is formed in the condensation reaction) 1 bi) Purple 1 bii) Because it’s a protein 1 c) Benedicts AND heat Brick Red

Application of Knowledge • http: //www. classtools. net/education-games-php/timer

Question 1 (again!!) 1 a) Nitrogen 1 b i) Benedicts AND heat Brick Red 1 b ii ) Substance A (glucose) 1 c i) Forming bonds, with the removal of water 1 c ii) C (protein) 1 d i) Substance D 1 d ii) Maximum amount of hydrogen atoms NO double bonds / all carbons attached to two hydrogens
- Slides: 13