Revising the Undergraduate Laboratory Curriculum A case study

Revising the Undergraduate Laboratory Curriculum: A case study from Chemistry at the University of Leeds Dr Nimesh Mistry School of Chemistry

Reasons for change Changing staff approach towards teaching – increased focus – moving from reactive to proactive culture Application to laboratory teaching – same teaching style for 30 – 40 years – lack of evidence to support teaching and learning

Overview Reviewing a laboratory programme How to design a curriculum – Programme level aims – Year level aims – Experimental level aims

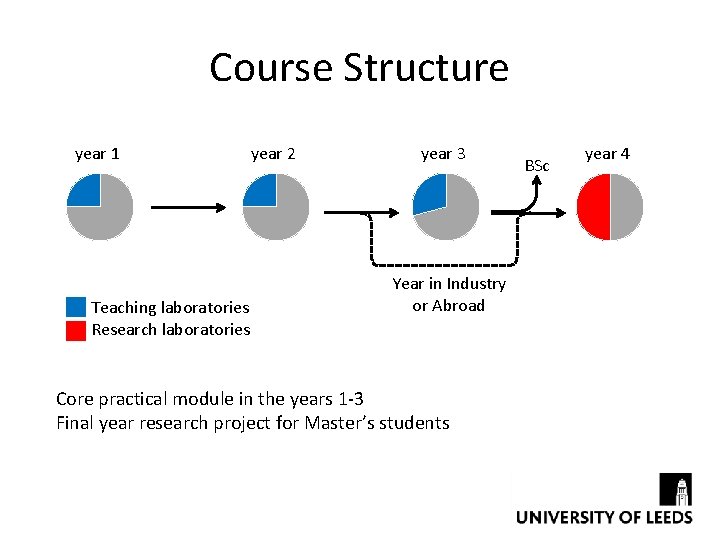
Course Structure year 1 Teaching laboratories Research laboratories year 2 year 3 Year in Industry or Abroad Core practical module in the years 1 -3 Final year research project for Master’s students BSc year 4
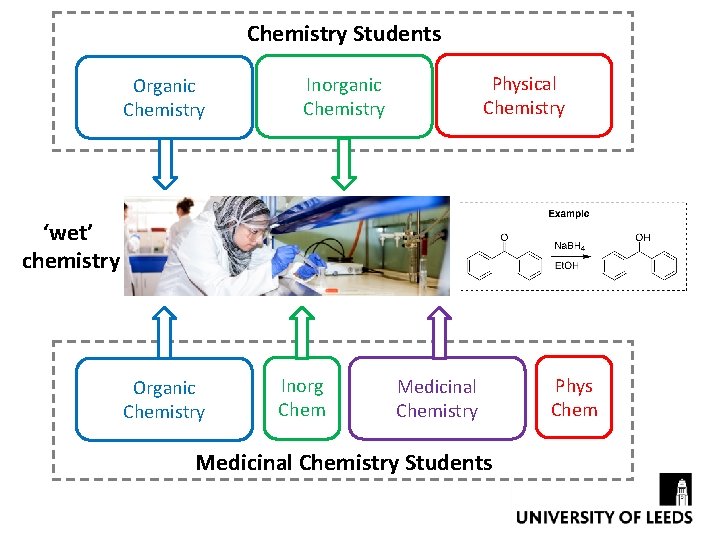
Chemistry Students Organic Chemistry Physical Chemistry Inorganic Chemistry ‘wet’ chemistry Organic Chemistry Inorg Chem Medicinal Chemistry Students Phys Chem
Change our approach to teaching Review practical teaching Implement best practice

What should students learn? technical skills application of theory data analysis transferable skills problem solving Reid N. and Shah I. , 2007, The role of laboratory work in university chemistry, Chemistry Education Research and Practice, 8, 172 -185 Benchmark Statement for Chemistry, QAA Accreditation of degree programmes, Royal Society of Chemistry

How do students learn? Constructive alignment – Students learn what they are asked to do – Driven by teaching tasks – Driven by assessment Biggs, J. and Tang, C. , 2011, Teaching for quality learning at university. 4 th edition. Open University Press.
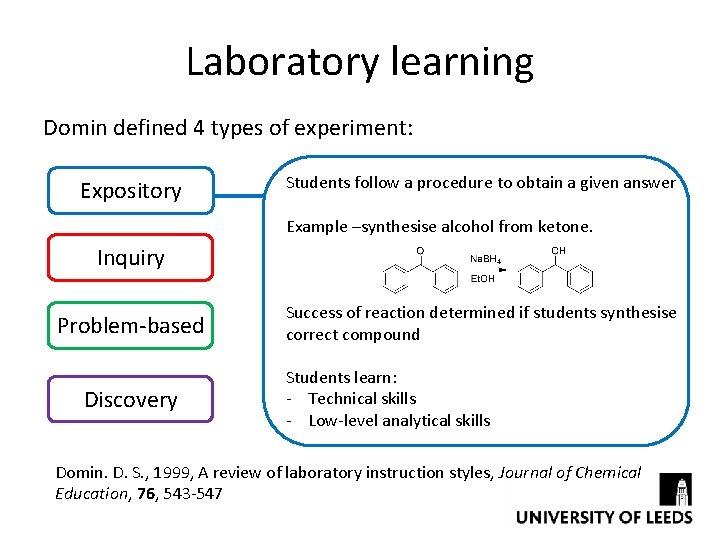
Laboratory learning Domin defined 4 types of experiment: Expository Students follow a procedure to obtain a given answer Example –synthesise alcohol from ketone. Inquiry Problem-based Discovery Success of reaction determined if students synthesise correct compound Students learn: - Technical skills - Low-level analytical skills Domin. D. S. , 1999, A review of laboratory instruction styles, Journal of Chemical Education, 76, 543 -547
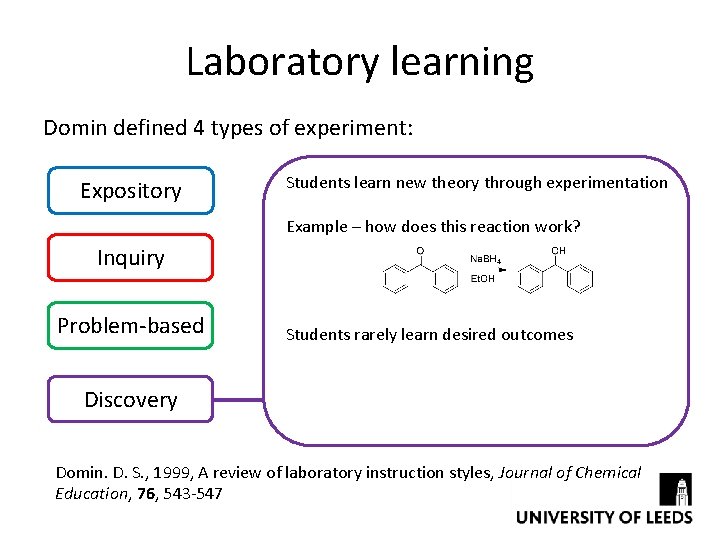
Laboratory learning Domin defined 4 types of experiment: Expository Students learn new theory through experimentation Example – how does this reaction work? Inquiry Problem-based Students rarely learn desired outcomes Discovery Domin. D. S. , 1999, A review of laboratory instruction styles, Journal of Chemical Education, 76, 543 -547
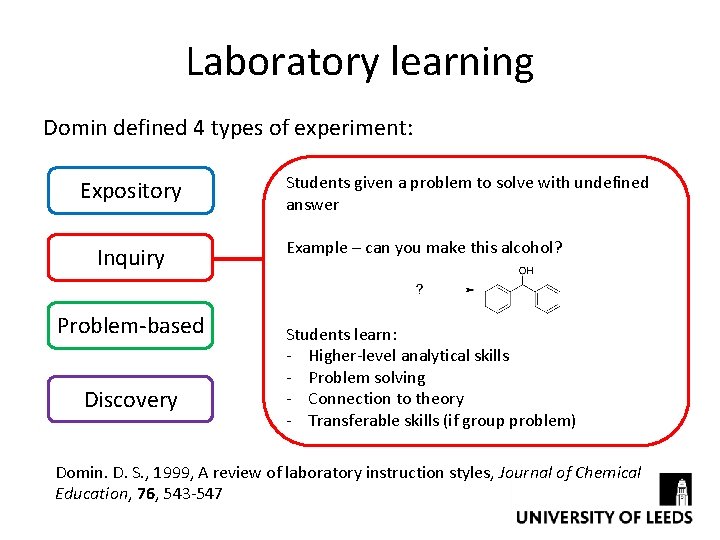
Laboratory learning Domin defined 4 types of experiment: Expository Inquiry Problem-based Discovery Students given a problem to solve with undefined answer Example – can you make this alcohol? Students learn: - Higher-level analytical skills - Problem solving - Connection to theory - Transferable skills (if group problem) Domin. D. S. , 1999, A review of laboratory instruction styles, Journal of Chemical Education, 76, 543 -547
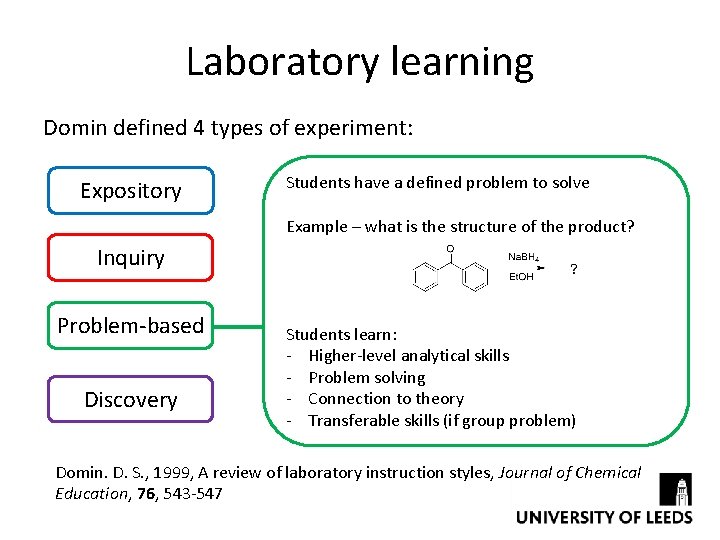
Laboratory learning Domin defined 4 types of experiment: Expository Students have a defined problem to solve Example – what is the structure of the product? Inquiry Problem-based Discovery Students learn: - Higher-level analytical skills - Problem solving - Connection to theory - Transferable skills (if group problem) Domin. D. S. , 1999, A review of laboratory instruction styles, Journal of Chemical Education, 76, 543 -547

Types of experiment Traditional expository experiments dominate the programme year 1 year 2 year 3 Expository Problem/Inquiry based Inquiry/problem-based experiments Years 1, 2 and 3 – determine structure of unknown compound Year 3 – 3 x 1 week mini research project
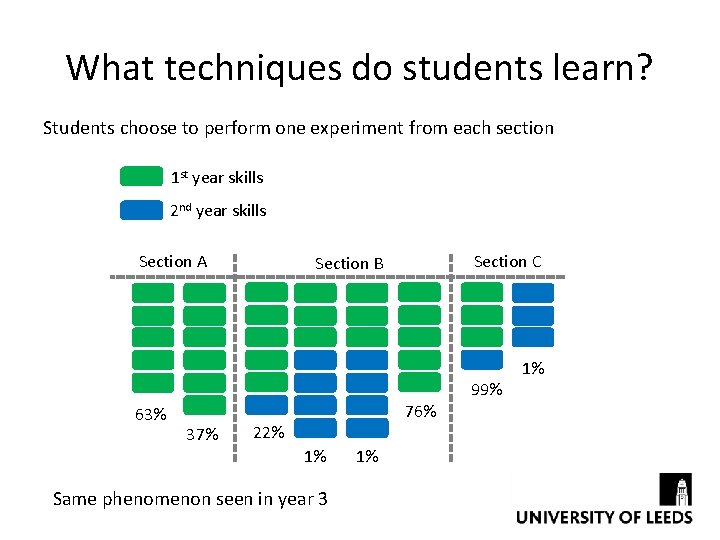
What techniques do students learn? Students choose to perform one experiment from each section 1 st year skills 2 nd year skills Section A 63% 37% Section C Section B 76% 22% 1% Same phenomenon seen in year 3 1% 99% 1%
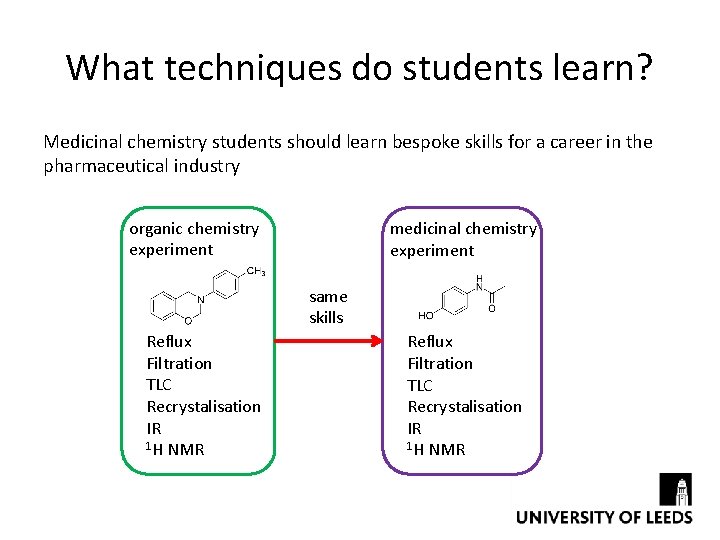
What techniques do students learn? Medicinal chemistry students should learn bespoke skills for a career in the pharmaceutical industry organic chemistry experiment medicinal chemistry experiment same skills Reflux Filtration TLC Recrystalisation IR 1 H NMR
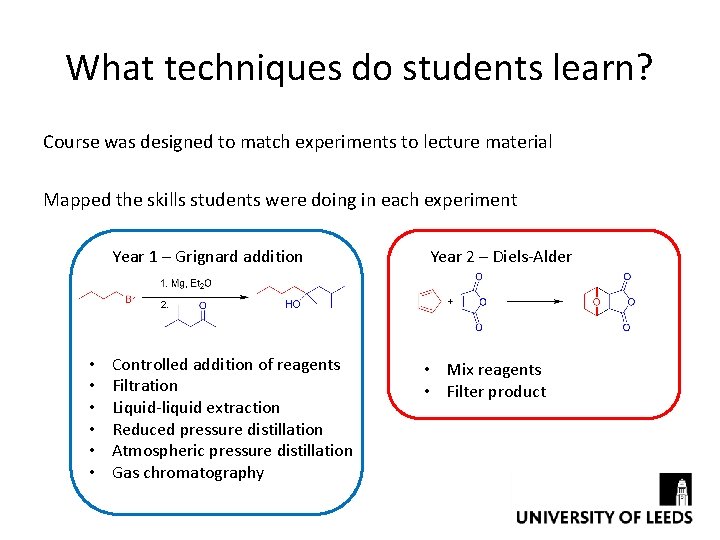
What techniques do students learn? Course was designed to match experiments to lecture material Mapped the skills students were doing in each experiment Year 1 – Grignard addition • • • Controlled addition of reagents Filtration Liquid-liquid extraction Reduced pressure distillation Atmospheric pressure distillation Gas chromatography Year 2 – Diels-Alder • Mix reagents • Filter product

Summary of review Too much emphasis on expository (cookbook) experiments throughout programme Chemistry theory (how the experiment works) dominates programme and experimental design • More complicated experiments in year 1 than years 2 and 3 • Avoidance of advanced techniques in years 2 and 3 • Medicinal chemistry and Chemistry programmes weren’t distinct
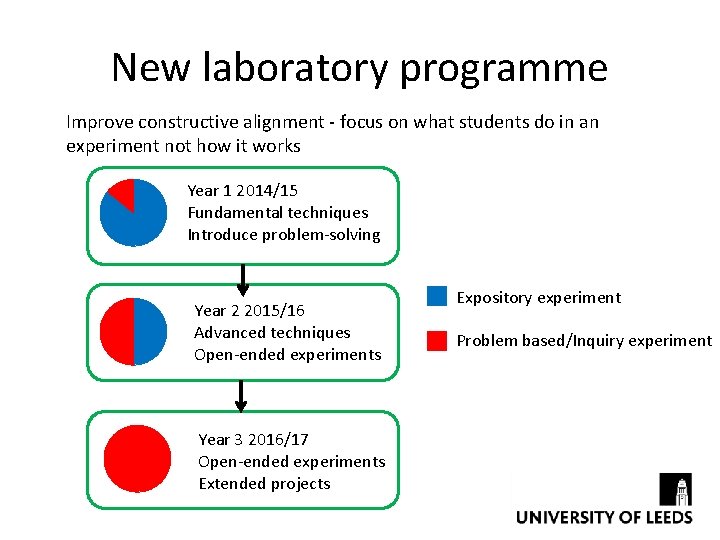
New laboratory programme Improve constructive alignment - focus on what students do in an experiment not how it works Year 1 2014/15 Fundamental techniques Introduce problem-solving Year 2 2015/16 Advanced techniques Open-ended experiments Year 3 2016/17 Open-ended experiments Extended projects Expository experiment Problem based/Inquiry experiment
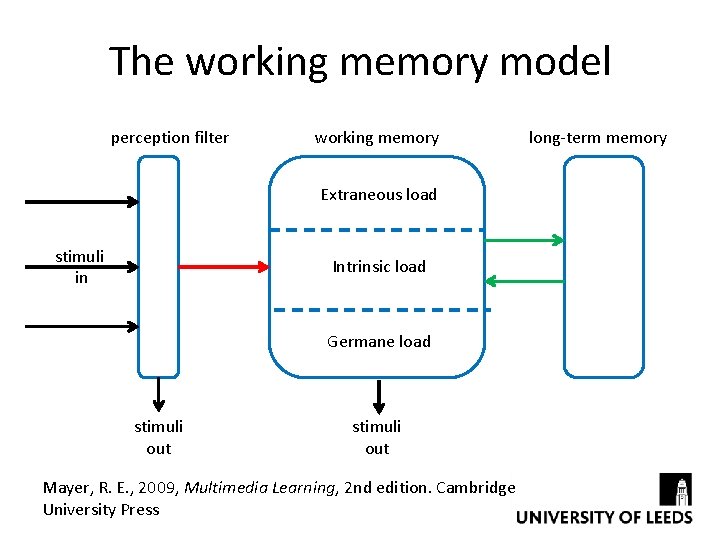
The working memory model perception filter working memory Extraneous load stimuli in Intrinsic load Germane load stimuli out Mayer, R. E. , 2009, Multimedia Learning, 2 nd edition. Cambridge University Press long-term memory

Information overload ‘’The conventional way of preparing students would be to encourage them to read their laboratory course manual, but these typically overload them with information to be held at the same time. Equally, if there is an incessant barrage of information, the students get completely lost in the argument and sequence of ideas. The manuals need to be re-written with simplicity in mind if it is desired that students do not use them as cookbooks’’ Johnstone A. H. and Letton K. M. , 1990, Investigation undergraduate laboratory work, Education in Chemistry, 27, 9 -11
Year 1 Introduce a non-assessed induction programme Change structure so students perform one experiment a week Changed/replaced experiments so they became progressively more difficult
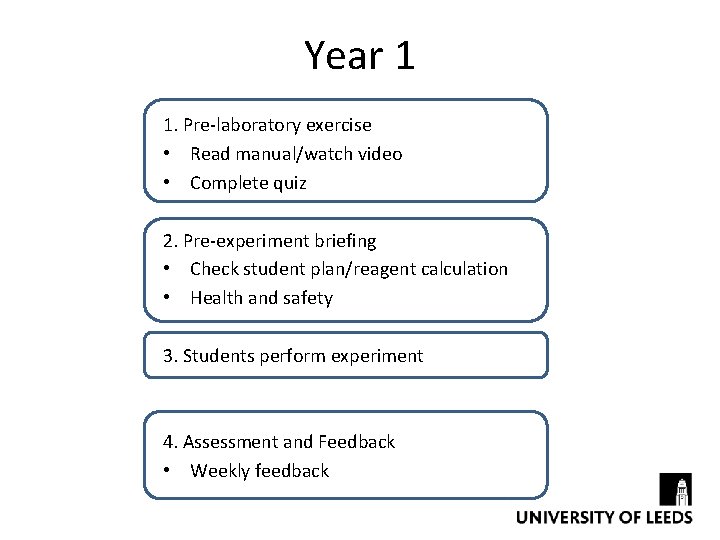
Year 1 1. Pre-laboratory exercise • Read manual/watch video • Complete quiz 2. Pre-experiment briefing • Check student plan/reagent calculation • Health and safety 3. Students perform experiment 4. Assessment and Feedback • Weekly feedback

Year 1 Pre-laboratory exercises 10% of overall grade Students cannot start if they haven’t completed the quiz Quiz only available during the week of the experiment Burewicz, A. , Miranowicz, N. , 2006, Effectiveness of multimedia laboratory instruction. Chemistry Education Research and Practice. 7(1), pp. 1 -12
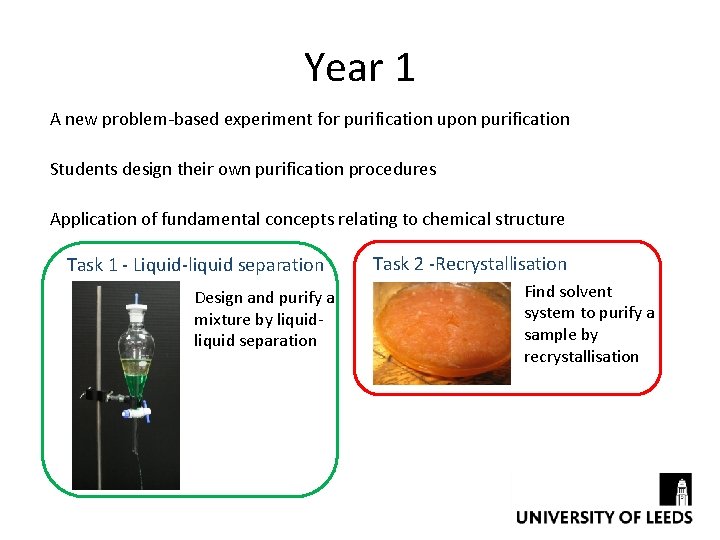
Year 1 A new problem-based experiment for purification upon purification Students design their own purification procedures Application of fundamental concepts relating to chemical structure Task 1 - Liquid-liquid separation Design and purify a mixture by liquid separation Task 2 -Recrystallisation Find solvent system to purify a sample by recrystallisation
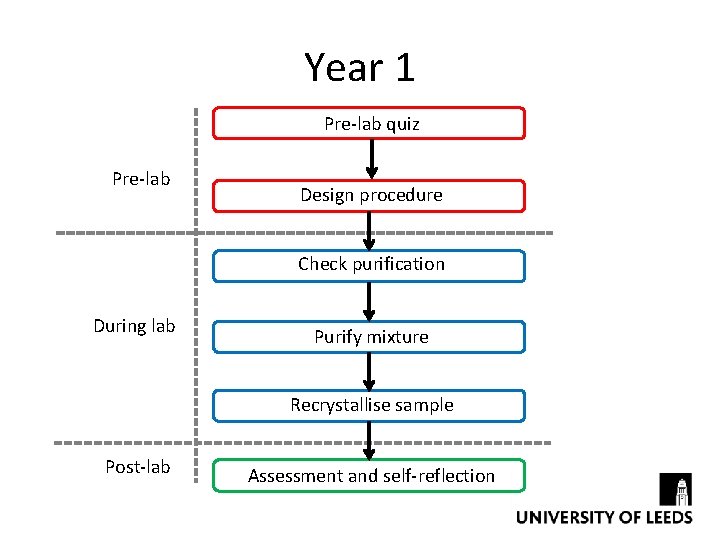
Year 1 Pre-lab quiz Pre-lab Design procedure Check purification During lab Purify mixture Recrystallise sample Post-lab Assessment and self-reflection
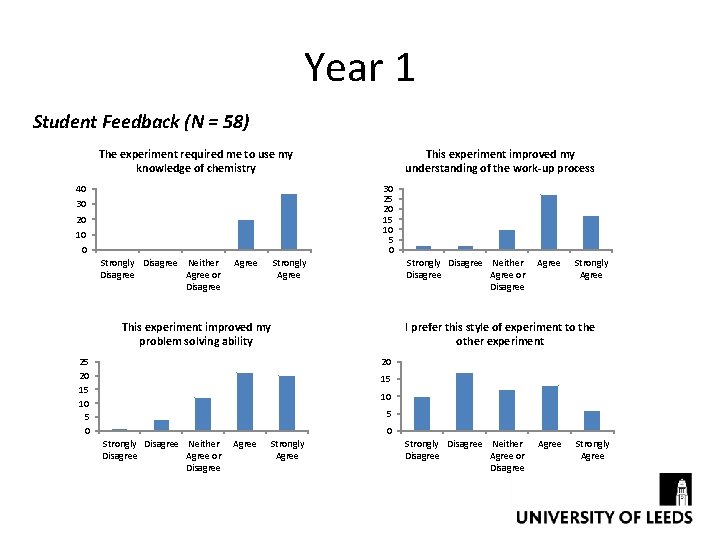
Year 1 Student Feedback (N = 58) The experiment required me to use my knowledge of chemistry 40 This experiment improved my understanding of the work-up process 30 25 20 15 10 5 0 30 20 10 0 Strongly Disagree Neither Disagree Agree or Disagree Agree Strongly Disagree Neither Disagree Agree or Disagree Strongly Agree This experiment improved my problem solving ability 25 20 15 10 5 0 Agree Strongly Agree I prefer this style of experiment to the other experiment 20 15 10 5 0 Strongly Disagree Neither Agree or Disagree Agree Strongly Agree
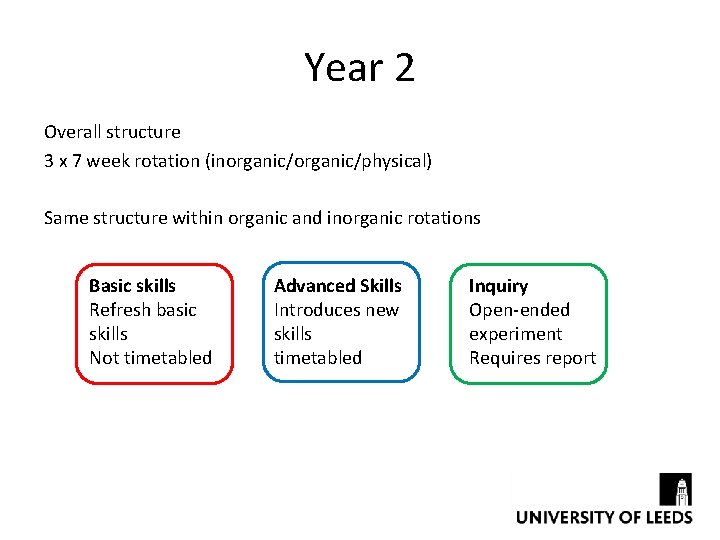
Year 2 Overall structure 3 x 7 week rotation (inorganic/physical) Same structure within organic and inorganic rotations Basic skills Refresh basic skills Not timetabled Advanced Skills Introduces new skills timetabled Inquiry Open-ended experiment Requires report
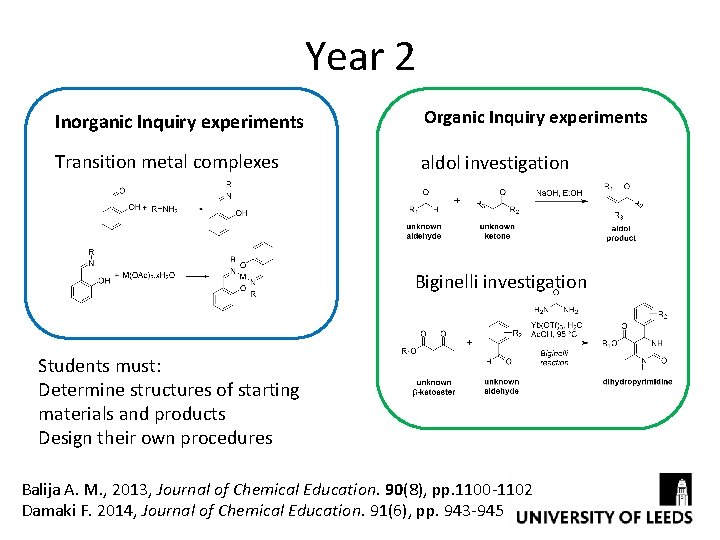
Year 2 Inorganic Inquiry experiments Organic Inquiry experiments Transition metal complexes aldol investigation Biginelli investigation Students must: Determine structures of starting materials and products Design their own procedures Balija A. M. , 2013, Journal of Chemical Education. 90(8), pp. 1100 -1102 Damaki F. 2014, Journal of Chemical Education. 91(6), pp. 943 -945

Year 2 Introduce practical skills relevant to Medicinal Chemistry course Computational drug design Industry and academic expertise Inhibition assay Interdisciplinary working
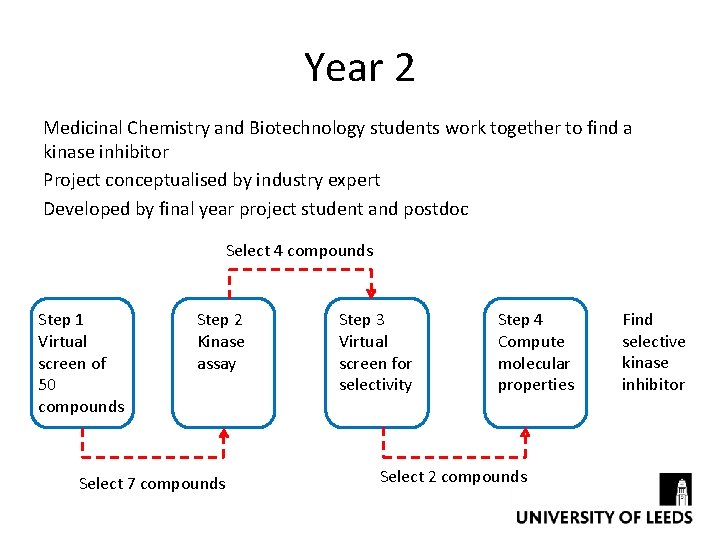
Year 2 Medicinal Chemistry and Biotechnology students work together to find a kinase inhibitor Project conceptualised by industry expert Developed by final year project student and postdoc Select 4 compounds Step 1 Virtual screen of 50 compounds Step 2 Kinase assay Select 7 compounds Step 3 Virtual screen for selectivity Step 4 Compute molecular properties Select 2 compounds Find selective kinase inhibitor

Year 3 Current year 3 • 3 x 7 week rotation (inorganic/physical) • 3 -4 weeks of core experiments • 3 -4 weeks of mini project Proposal for year 3 (at the moment) • Longer projects to develop research skills • Inquiry experiments for semester 1 • 10 week project in semester 2 • Choose between physical, inorganic, organic or non-laboratory based

Year 3 Learn Chemistry – open resources for teaching chemistry by the Royal Society of Chemistry Funding call to develop context/problem-based resources Successful in obtaining funding for C/PBL-project

Year 3 Teach/apply concepts of process and green chemistry • Cost • Hazards • Scalability Develop transferable skills • Time management • Teamwork • Communication Could be performed at different institutions • Low cost • Flexible – length, group size
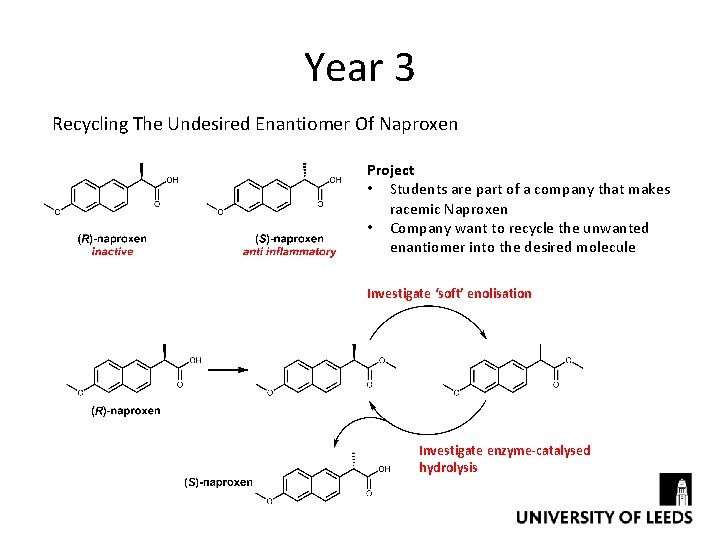
Year 3 Recycling The Undesired Enantiomer Of Naproxen Project • Students are part of a company that makes racemic Naproxen • Company want to recycle the unwanted enantiomer into the desired molecule Investigate ‘soft’ enolisation Investigate enzyme-catalysed hydrolysis

Year 3 Naproxen Resources • • • Module booklet Teacher’s guide Lecture presentation Skills videos Workshop slides, handout and answers Laboratory manual Sarah Narramore @kaytaralee Lecture and workshop resources George Burslem @Synthesist 88 Experimental development

Future plans More C/PBL-project development for year 3 • Lab-based • Non-lab based (educational/communication) Improve teaching practice • Demonstrator training Improve assessment • Practical exam?

Acknowledgements School of Chemistry management committee Development team • Stephen Gorman • Chris Pask • Sarah Narramore • George Burslem • Oliver Morrissey • Jennifer Miles Dr Samantha Pugh Royal Society of Chemistry (funding)
- Slides: 38