Revised 2182014 Biochemistry I Dr Loren Williams Chapter

Revised 2/18/2014 Biochemistry I Dr. Loren Williams Chapter 4 Amino Acids
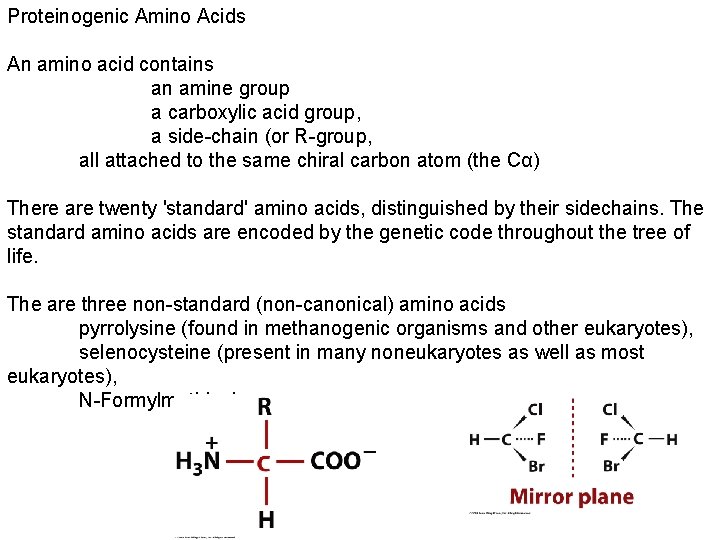
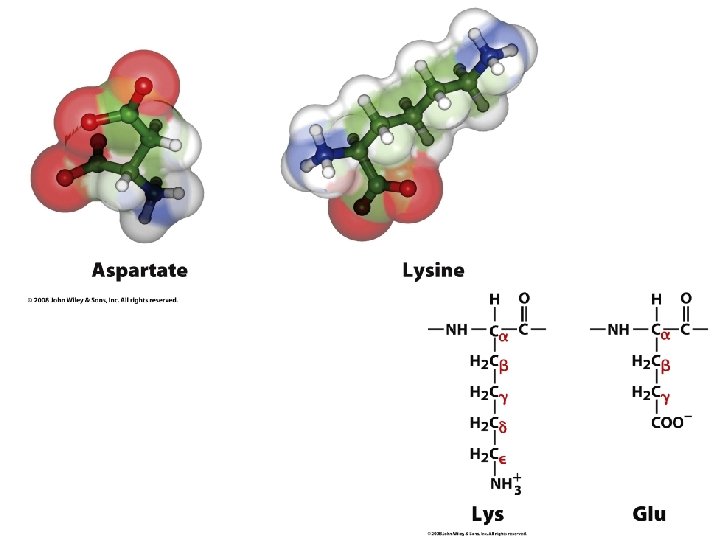
Proteinogenic Amino Acids An amino acid contains an amine group a carboxylic acid group, a side-chain (or R-group, all attached to the same chiral carbon atom (the Cα) There are twenty 'standard' amino acids, distinguished by their sidechains. The standard amino acids are encoded by the genetic code throughout the tree of life. The are three non-standard (non-canonical) amino acids pyrrolysine (found in methanogenic organisms and other eukaryotes), selenocysteine (present in many noneukaryotes as well as most eukaryotes), N-Formylmethionine.
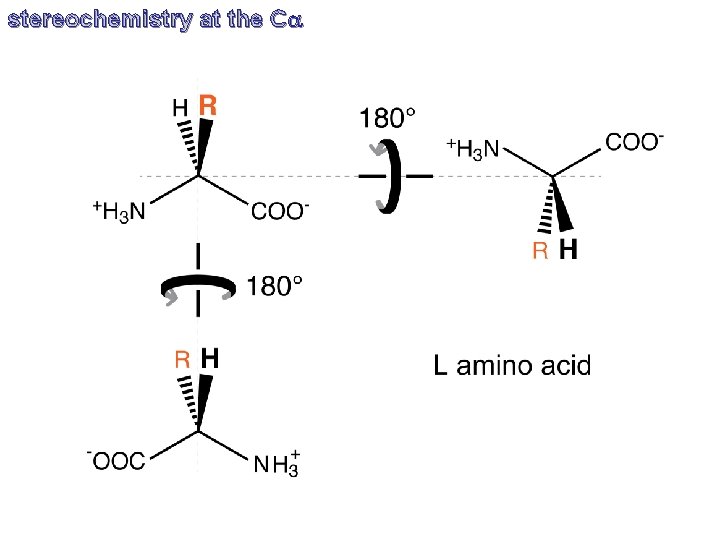
stereochemistry at the Ca
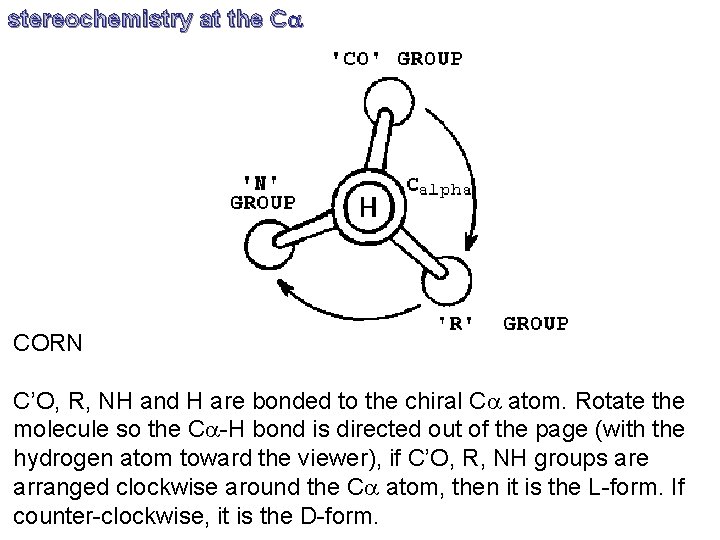
stereochemistry at the Ca CORN C’O, R, NH and H are bonded to the chiral Ca atom. Rotate the molecule so the Ca-H bond is directed out of the page (with the hydrogen atom toward the viewer), if C’O, R, NH groups are arranged clockwise around the Ca atom, then it is the L-form. If counter-clockwise, it is the D-form.
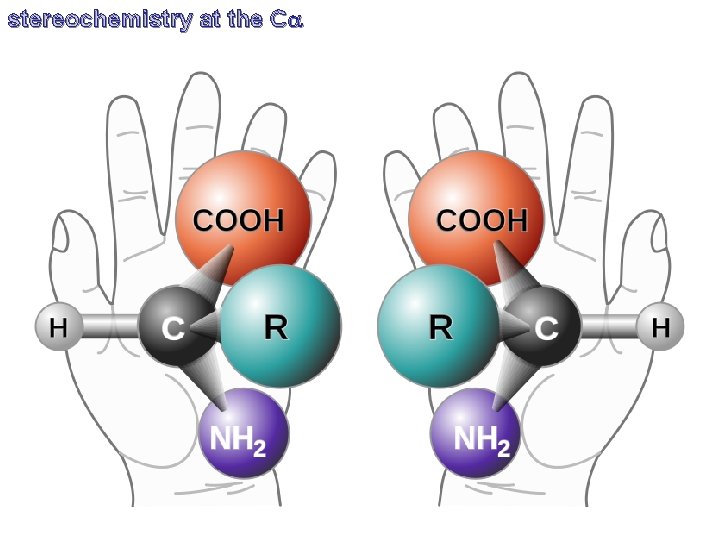
stereochemistry at the Ca
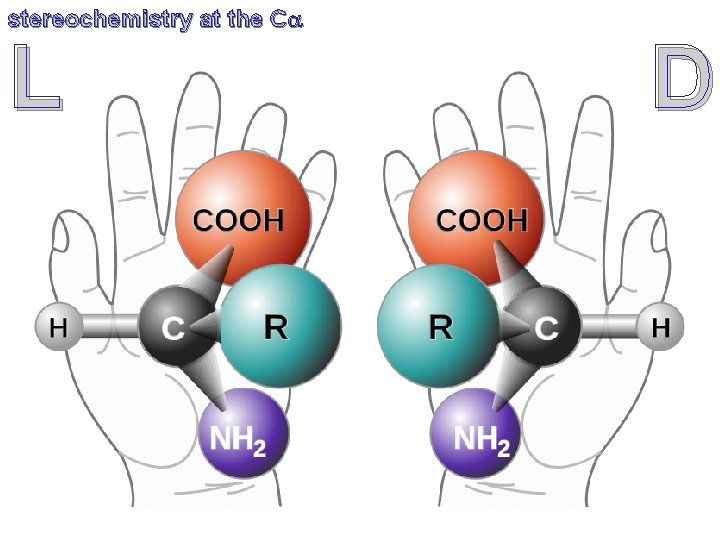
stereochemistry at the Ca L D
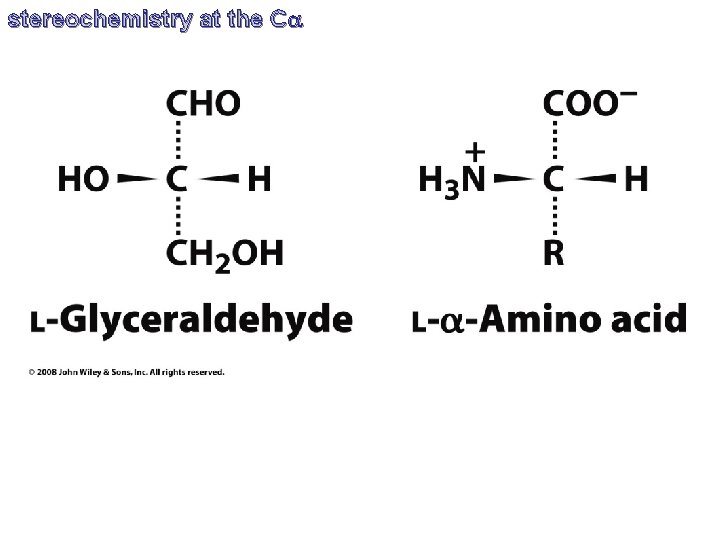
stereochemistry at the Ca
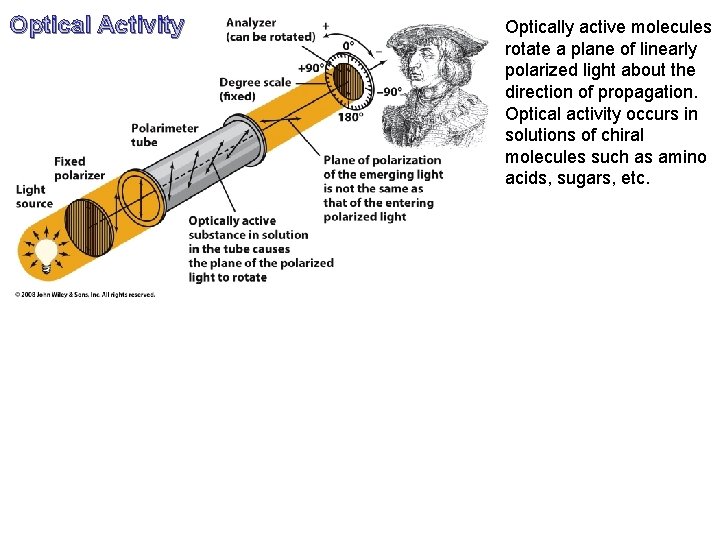
Optical Activity Optically active molecules rotate a plane of linearly polarized light about the direction of propagation. Optical activity occurs in solutions of chiral molecules such as amino acids, sugars, etc.
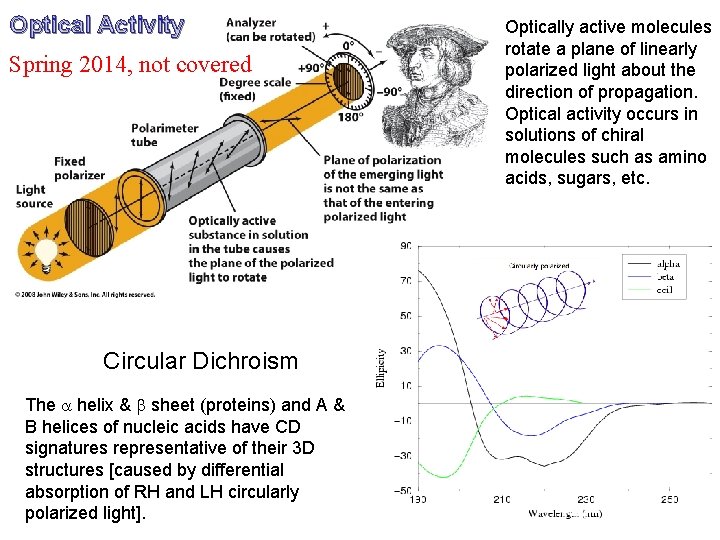
Optical Activity Spring 2014, not covered Circular Dichroism The a helix & b sheet (proteins) and A & B helices of nucleic acids have CD signatures representative of their 3 D structures [caused by differential absorption of RH and LH circularly polarized light]. Optically active molecules rotate a plane of linearly polarized light about the direction of propagation. Optical activity occurs in solutions of chiral molecules such as amino acids, sugars, etc.
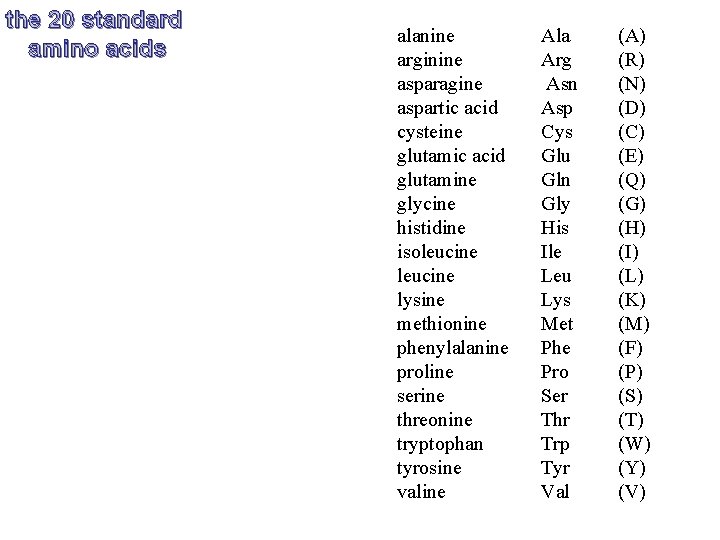
the 20 standard amino acids alanine arginine asparagine aspartic acid cysteine glutamic acid glutamine glycine histidine isoleucine lysine methionine phenylalanine proline serine threonine tryptophan tyrosine valine Ala Arg Asn Asp Cys Glu Gln Gly His Ile Leu Lys Met Phe Pro Ser Thr Trp Tyr Val (A) (R) (N) (D) (C) (E) (Q) (G) (H) (I) (L) (K) (M) (F) (P) (S) (T) (W) (Y) (V)
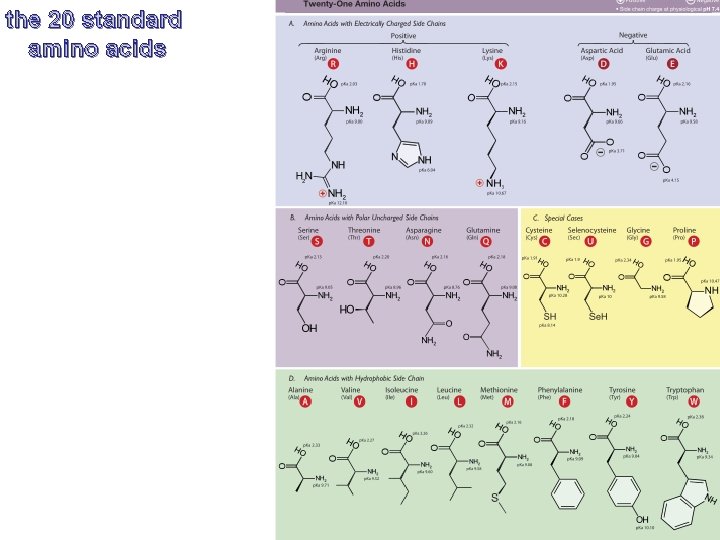
the 20 standard amino acids
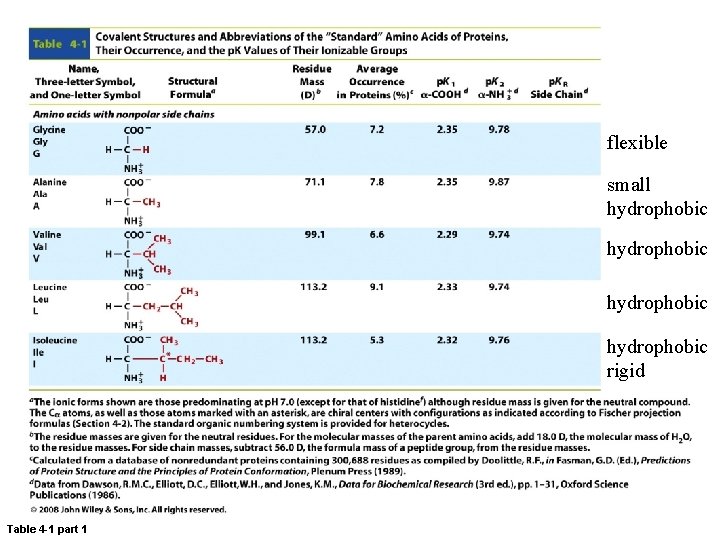
flexible small hydrophobic rigid Table 4 -1 part 1
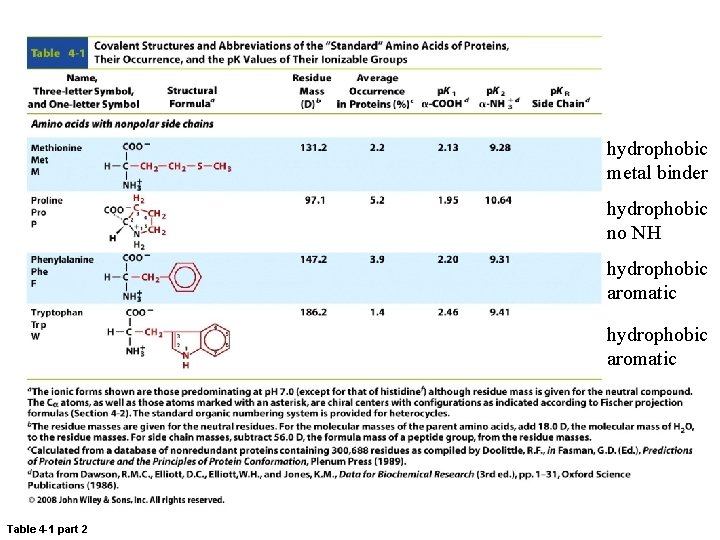
hydrophobic metal binder hydrophobic no NH hydrophobic aromatic Table 4 -1 part 2
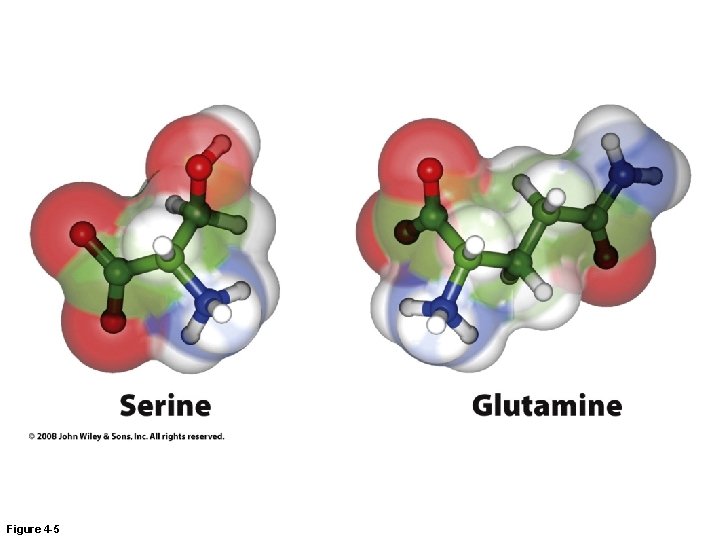
Figure 4 -5
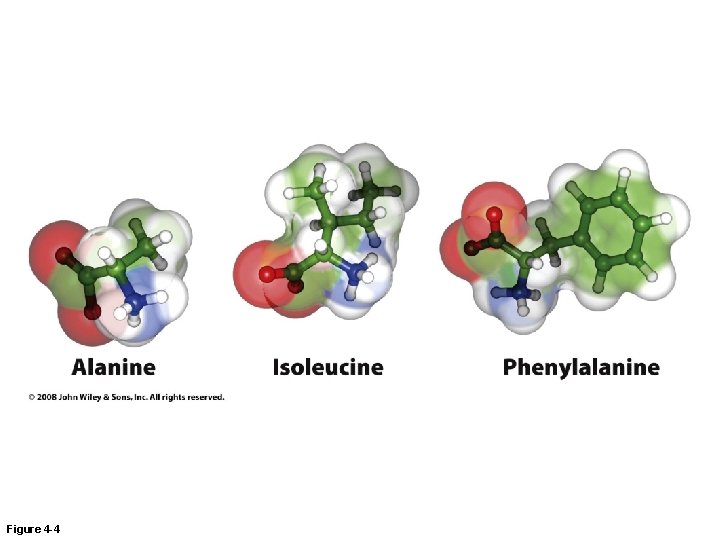
Figure 4 -4
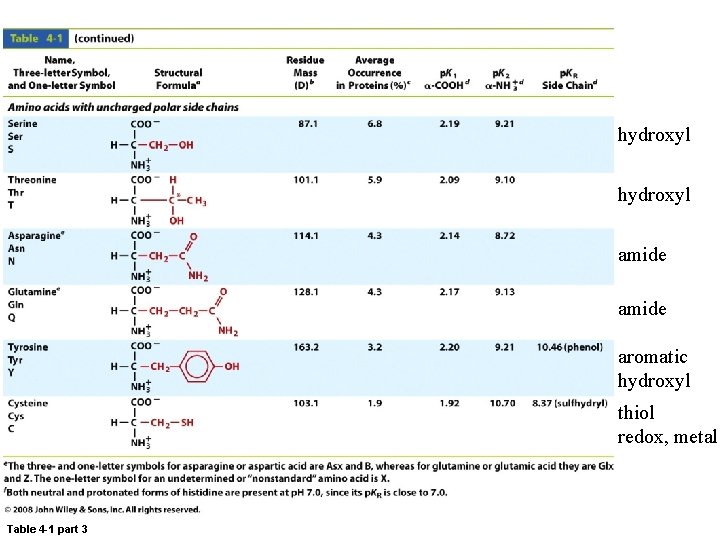
hydroxyl amide aromatic hydroxyl thiol redox, metal Table 4 -1 part 3
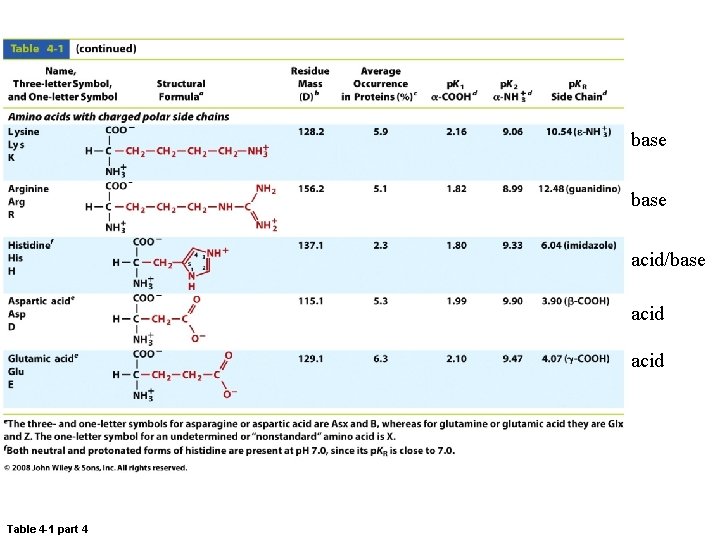
base acid/base acid Table 4 -1 part 4
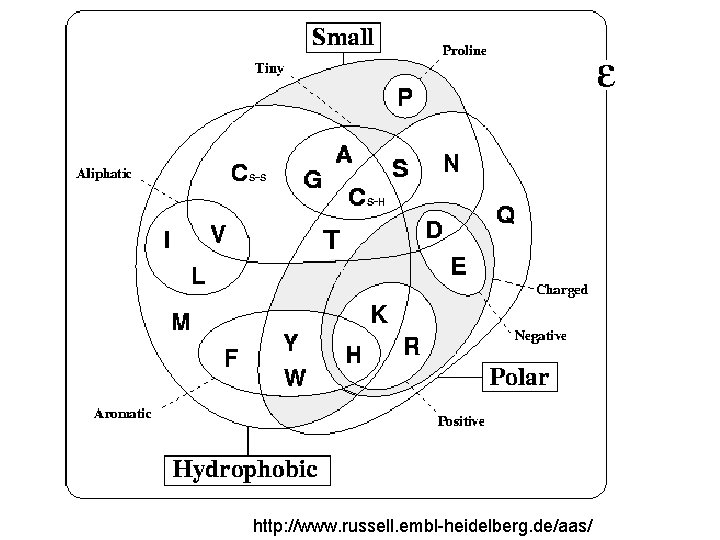
http: //www. russell. embl-heidelberg. de/aas/

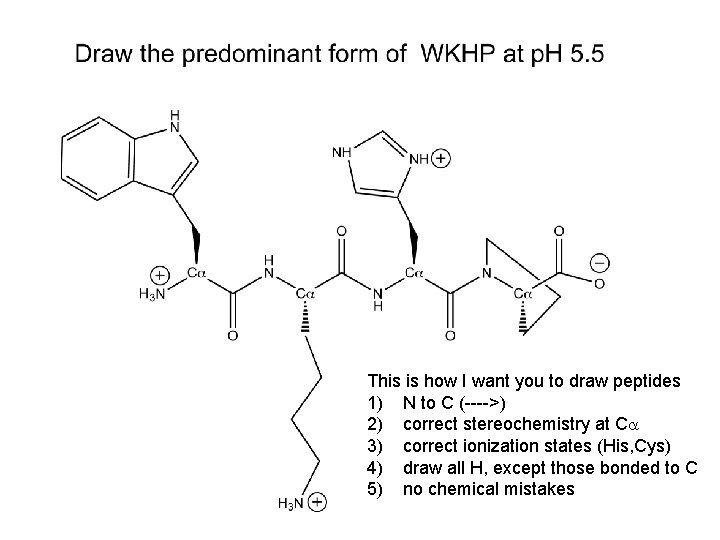
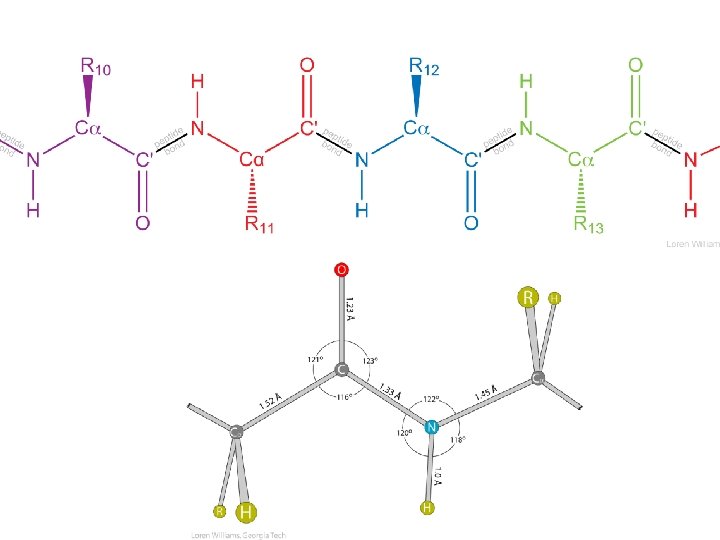
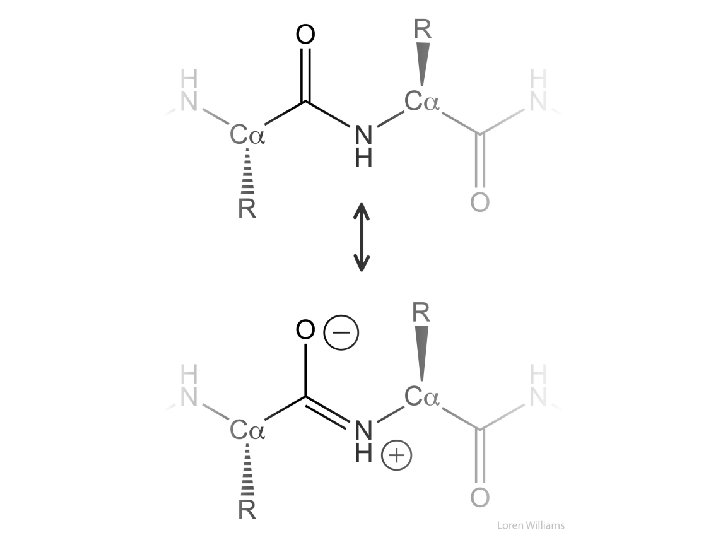
This is how I want you to draw peptides 1) N to C (---->) 2) correct stereochemistry at Ca 3) correct ionization states (His, Cys) 4) draw all H, except those bonded to C 5) no chemical mistakes
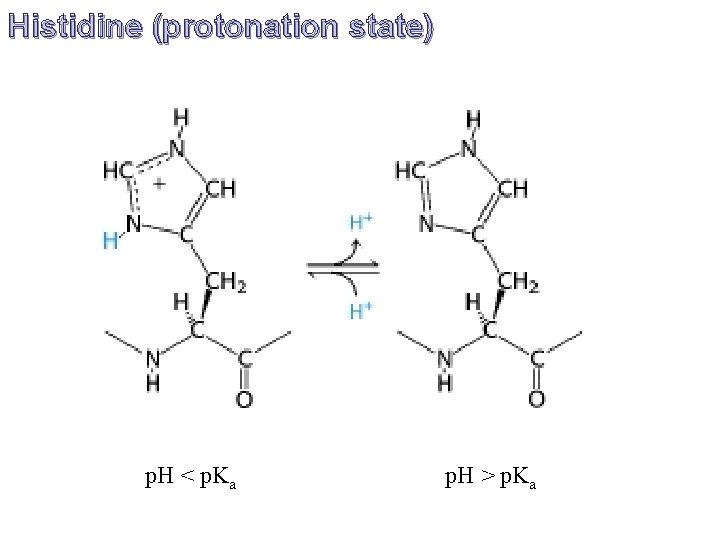
Histidine (protonation state) p. H < p. Ka p. H > p. Ka

HA There is a sign error here that microsoft won’t let me fix. H+ + A –
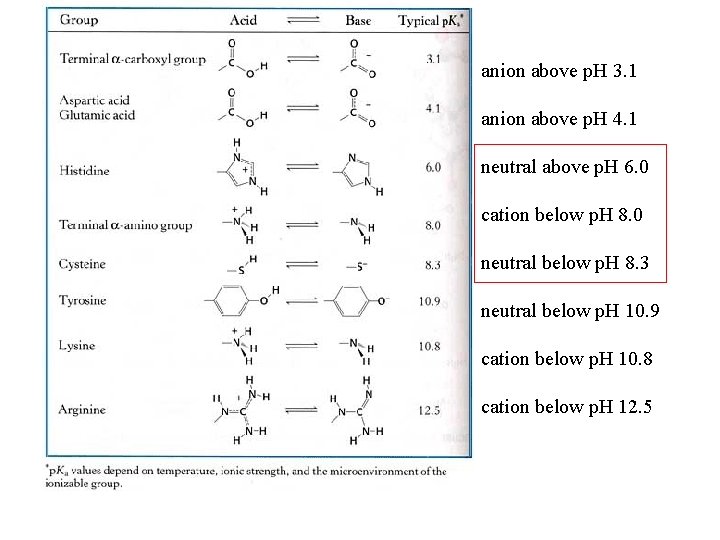
anion above p. H 3. 1 anion above p. H 4. 1 neutral above p. H 6. 0 cation below p. H 8. 0 neutral below p. H 8. 3 neutral below p. H 10. 9 cation below p. H 10. 8 cation below p. H 12. 5

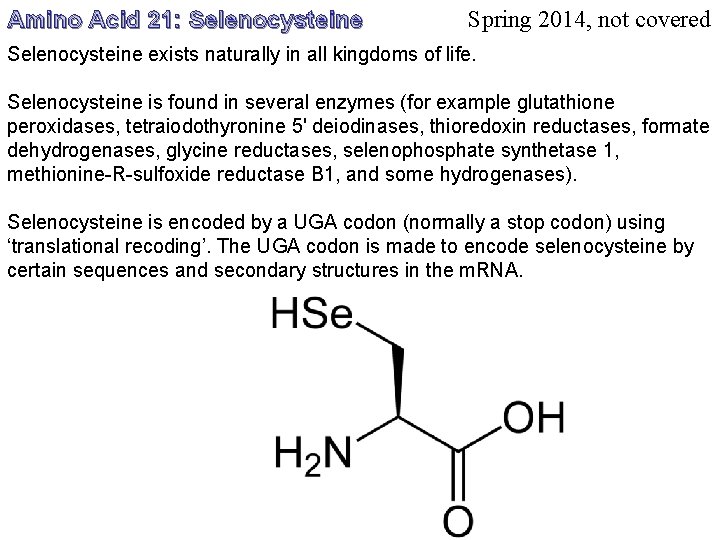
Amino Acid 21: Selenocysteine Spring 2014, not covered Selenocysteine exists naturally in all kingdoms of life. Selenocysteine is found in several enzymes (for example glutathione peroxidases, tetraiodothyronine 5' deiodinases, thioredoxin reductases, formate dehydrogenases, glycine reductases, selenophosphate synthetase 1, methionine-R-sulfoxide reductase B 1, and some hydrogenases). Selenocysteine is encoded by a UGA codon (normally a stop codon) using ‘translational recoding’. The UGA codon is made to encode selenocysteine by certain sequences and secondary structures in the m. RNA.
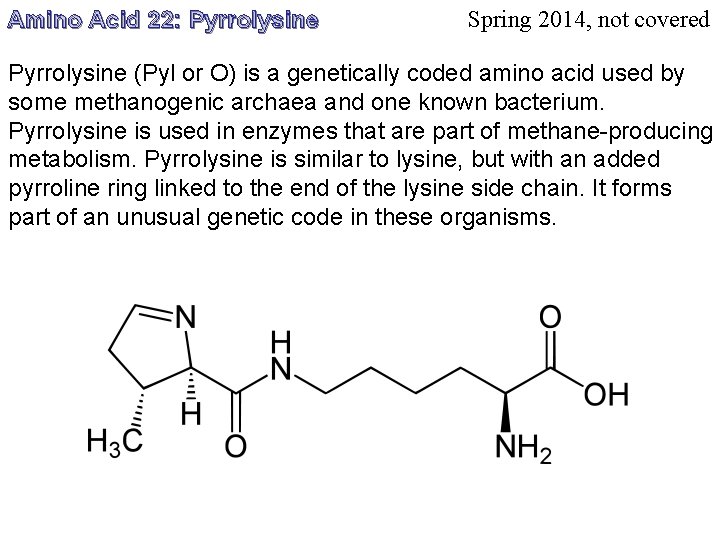
Amino Acid 22: Pyrrolysine Spring 2014, not covered Pyrrolysine (Pyl or O) is a genetically coded amino acid used by some methanogenic archaea and one known bacterium. Pyrrolysine is used in enzymes that are part of methane-producing metabolism. Pyrrolysine is similar to lysine, but with an added pyrroline ring linked to the end of the lysine side chain. It forms part of an unusual genetic code in these organisms.
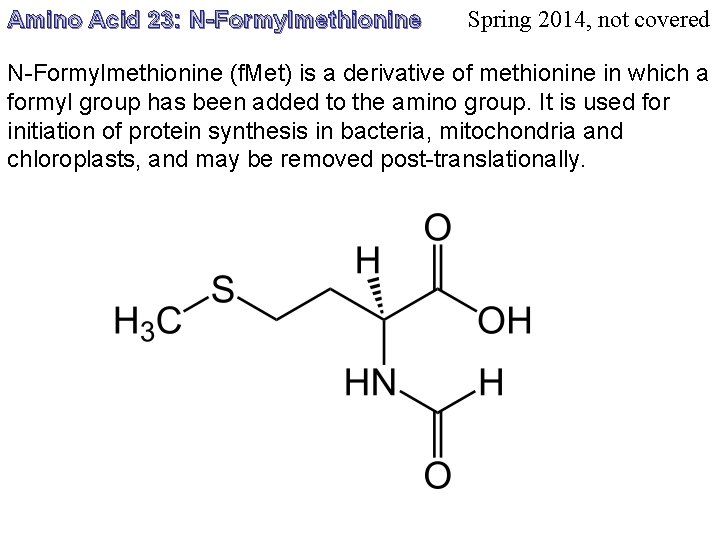
Amino Acid 23: N-Formylmethionine Spring 2014, not covered N-Formylmethionine (f. Met) is a derivative of methionine in which a formyl group has been added to the amino group. It is used for initiation of protein synthesis in bacteria, mitochondria and chloroplasts, and may be removed post-translationally.
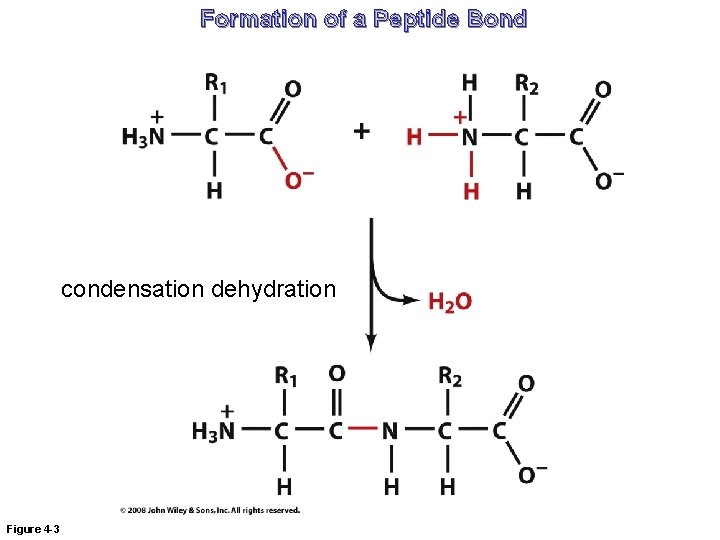
Formation of a Peptide Bond condensation dehydration Figure 4 -3
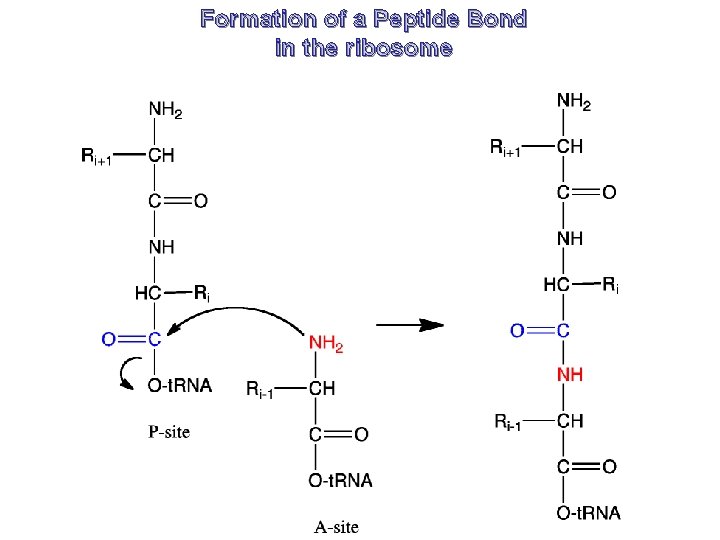
Formation of a Peptide Bond in the ribosome
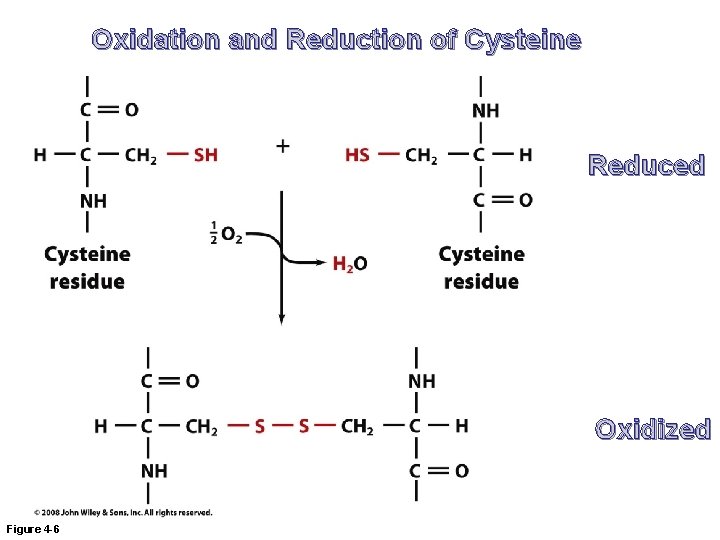
Oxidation and Reduction of Cysteine Reduced Oxidized Figure 4 -6
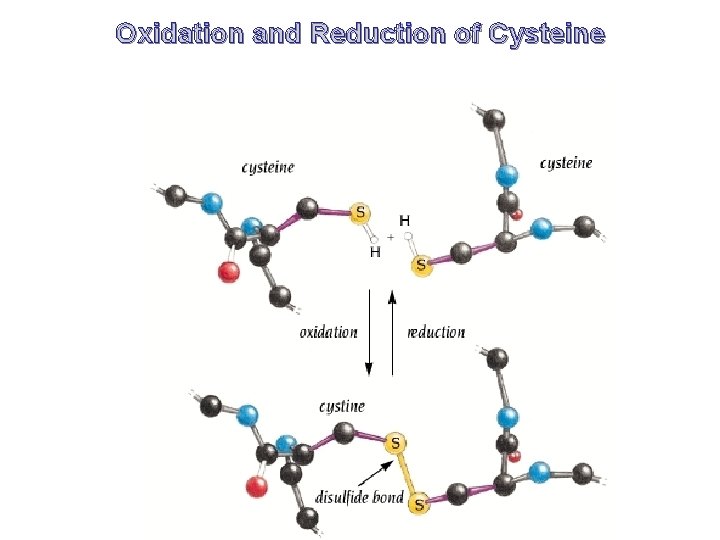
Oxidation and Reduction of Cysteine
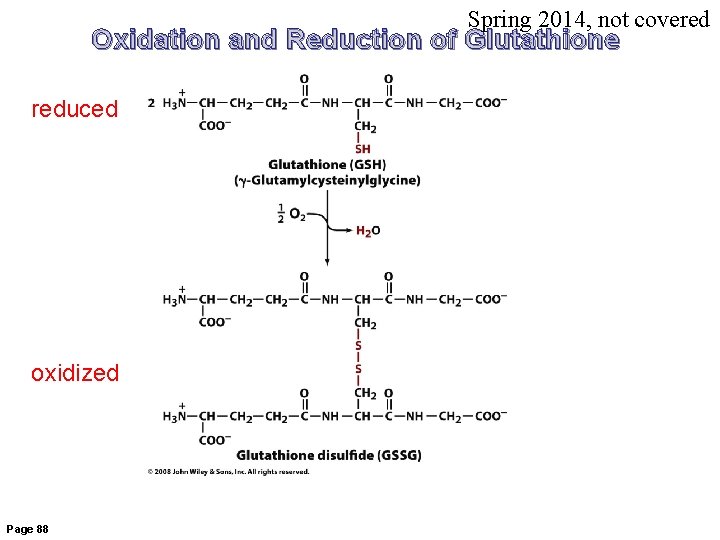
Spring 2014, not covered Oxidation and Reduction of Glutathione reduced oxidized Page 88
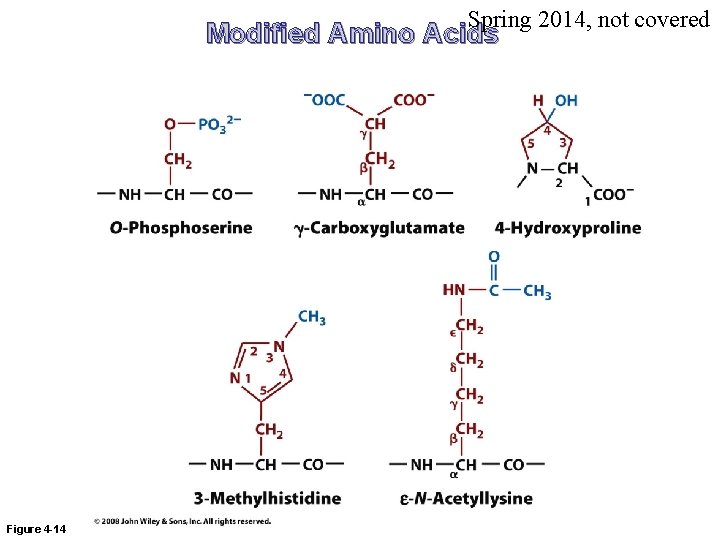
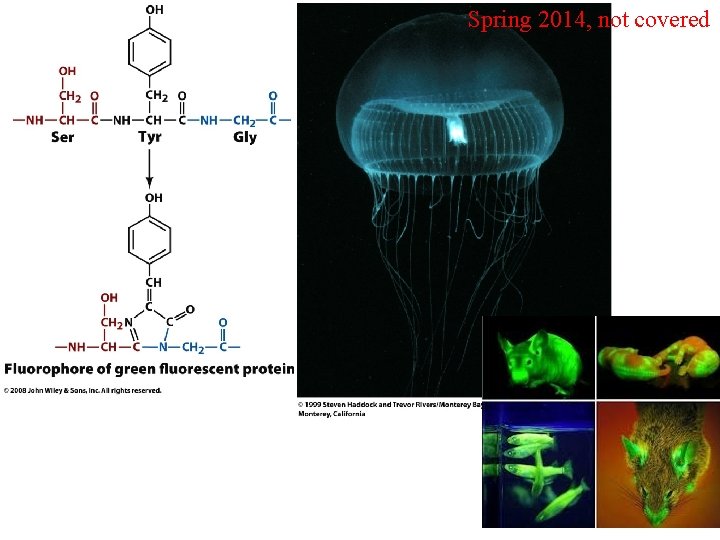
Spring 2014, not covered Modified Amino Acids Figure 4 -14
Spring 2014, not covered
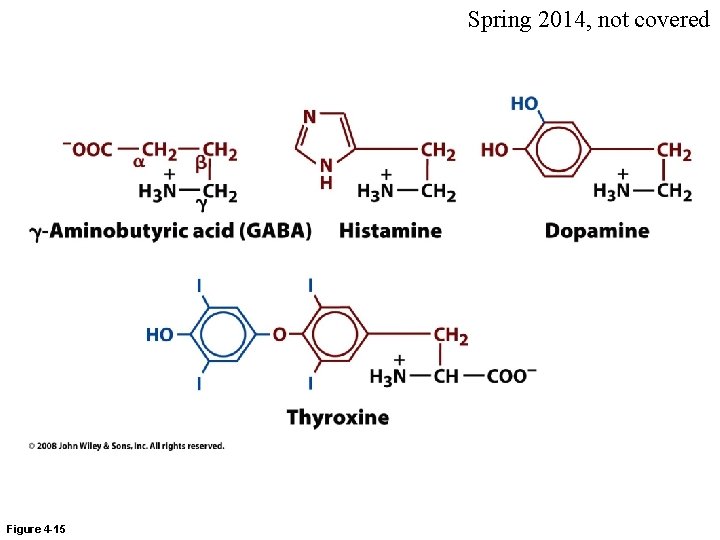
Spring 2014, not covered Figure 4 -15
- Slides: 38