Review Reaction mechanism Br 2l C 5 H
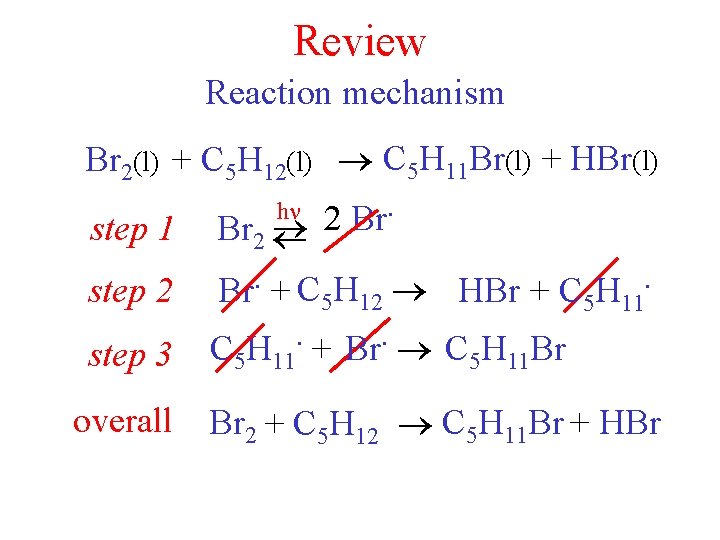
Review Reaction mechanism Br 2(l) + C 5 H 12(l) C 5 H 11 Br(l) + HBr(l) h . 2 Br step 1 Br 2 step 2 step 3 . . Br + C 5 H 12 HBr + C 5 H 11. + Br. C 5 H 11 Br overall Br 2 + C 5 H 12 C 5 H 11 Br + HBr

Br 2(l) + C 5 H 12(l) C 5 H 11 Br(l) + HBr(l) h 2 Br. step 1 Br 2 step 2 . . C H Br + 5 12 HBr + C 5 H 11. . C H + Br C H Br step 3 5 11 assume step 2 is rate determining (slow) rate = k 2[Br. ][C 5 H 12] . Br = intermediate. [Br ]2 rate = k 2 k’[Br 2]1/2[C 5 H 12] rate = k’[Br 2]1/2[C 5 H 12] 11/2 order reaction Keq = . [Br ] = [Br 2] Keq 1/2 [Br 2]1/2
![bimolecular elementary steps increase [react] increase rate of reaction increase T increase rate of bimolecular elementary steps increase [react] increase rate of reaction increase T increase rate of](http://slidetodoc.com/presentation_image_h2/17ec8df62c0daa53ec2a30227aaf0f77/image-3.jpg)
bimolecular elementary steps increase [react] increase rate of reaction increase T increase rate of reaction increase number of collisions increase force of collisions
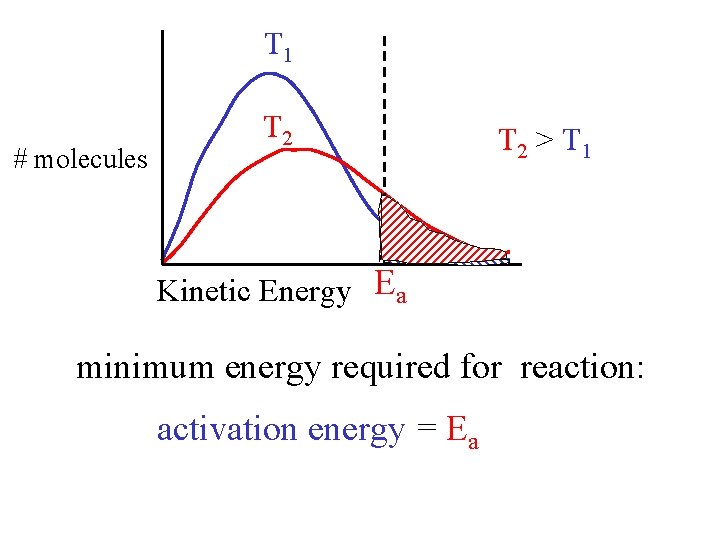
T 1 # molecules T 2 > T 1 Kinetic Energy Ea minimum energy required for reaction: activation energy = Ea

Arrhenius Equation T dependence of a rate constant, k k = z p e-Ea/RT k a) increases with T decreases with Ea k b) a) decreases b) increases Ea = activation energy (k. J/mol) R = gas constant (8. 314 x 10 -3 k. J/K mol) T = temperature (K) z = collision frequency p = steric factor (<1)
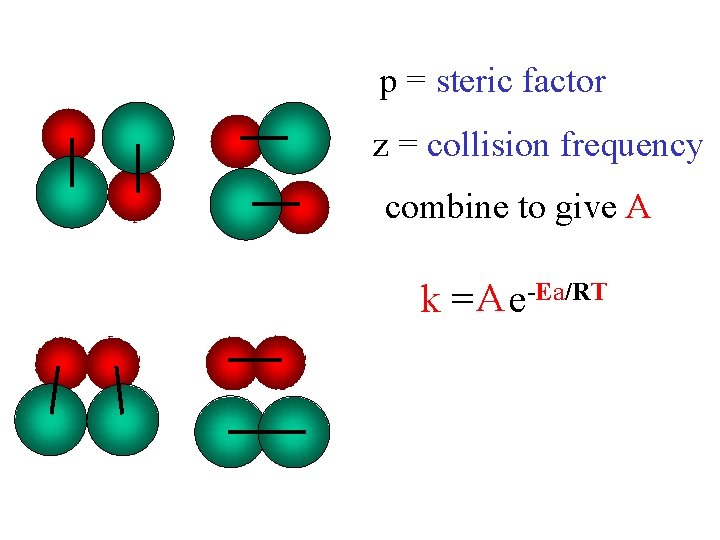
p = steric factor z = collision frequency combine to give A k = A e-Ea/RT

Arrhenius Equation k = A e-Ea/RT - (E(1/T) ln k = -ln(EAa/R) + ln A a/R) (1/T) y = m x + b plot ln k v. s. 1/T slope = -Ea/R intercept = ln A ln (k 2/ k 1) = (Ea /R) (1/T 1 - 1/T 2)
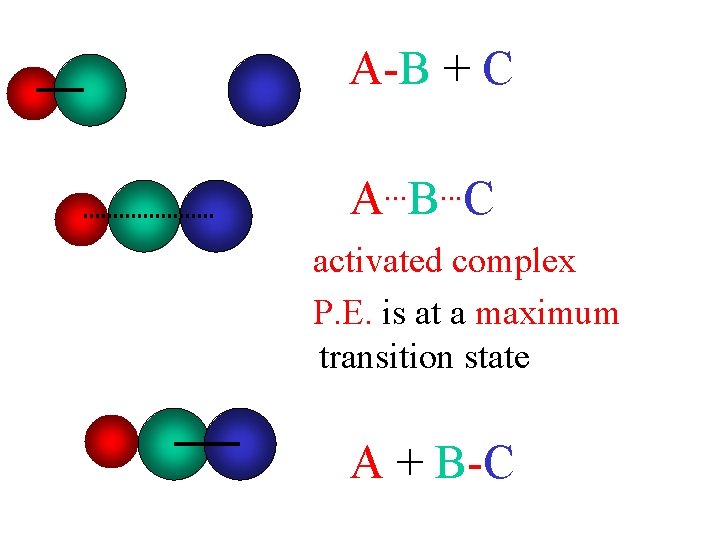
A-B + C. . . A B C activated complex P. E. is at a maximum transition state A + B-C
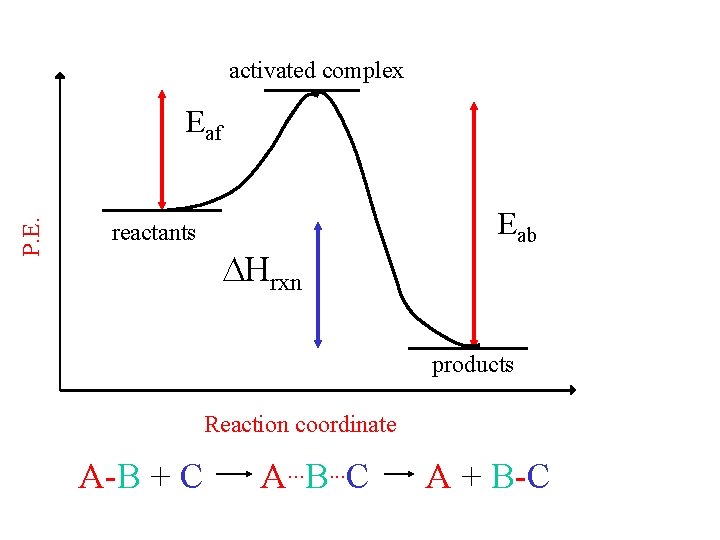
activated complex P. E. Eaf reactants Hrxn Eab products Reaction coordinate A-B + C A. . . B. . . C A + B-C
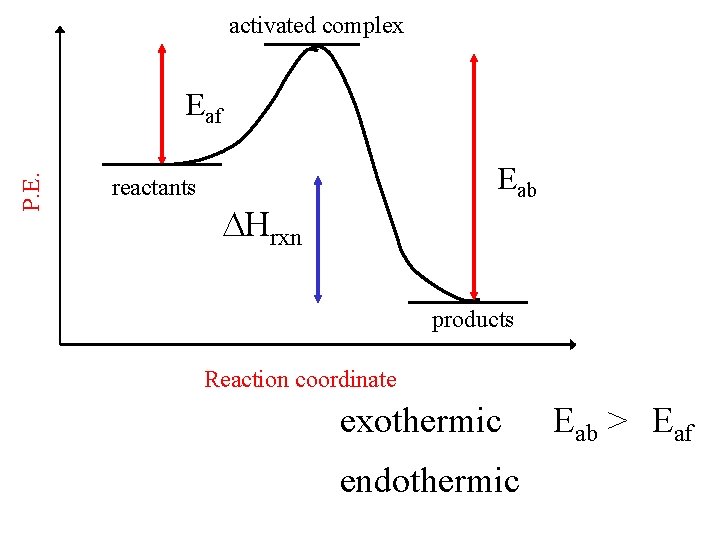
activated complex P. E. Eaf reactants Eab Hrxn products Reaction coordinate exothermic endothermic Eab > Eaf
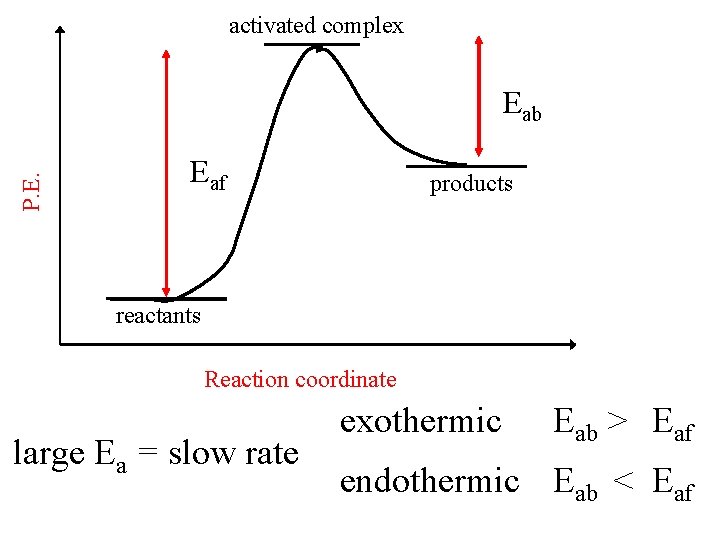
activated complex P. E. Eab Eaf products reactants Reaction coordinate large Ea = slow rate exothermic Eab > Eaf endothermic Eab < Eaf
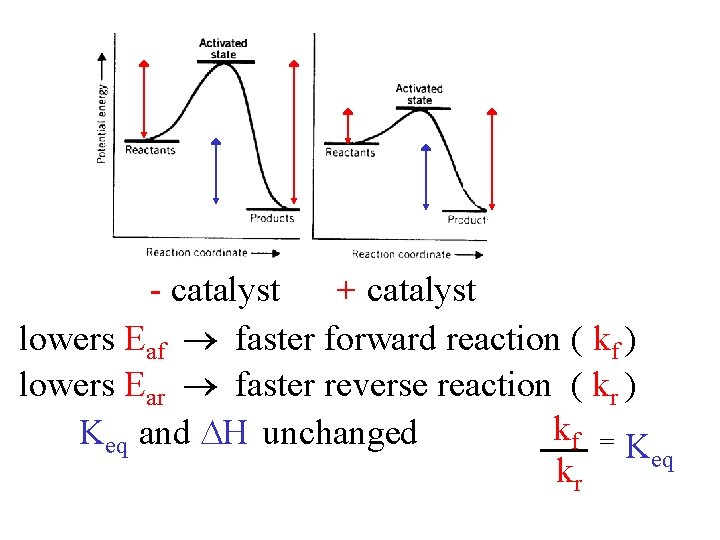
- catalyst + catalyst lowers Eaf faster forward reaction ( kf ) lowers Ear faster reverse reaction ( kr ) kf = K Keq and H unchanged eq kr
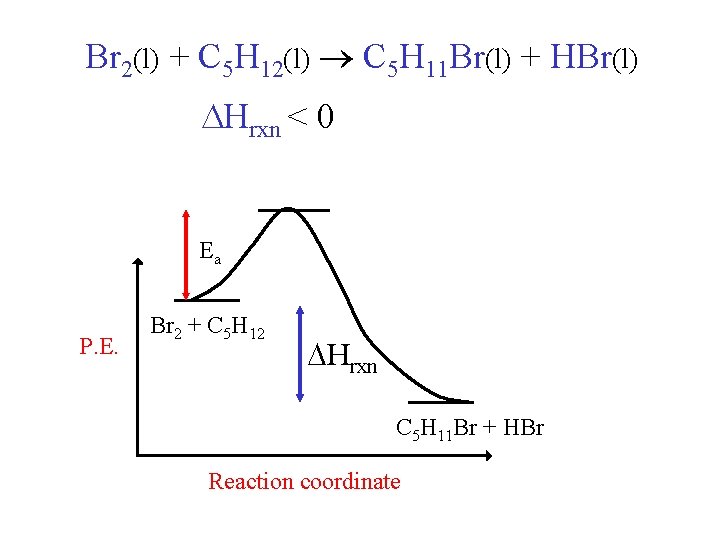
Br 2(l) + C 5 H 12(l) C 5 H 11 Br(l) + HBr(l) Hrxn < 0 Ea P. E. Br 2 + C 5 H 12 Hrxn C 5 H 11 Br + HBr Reaction coordinate
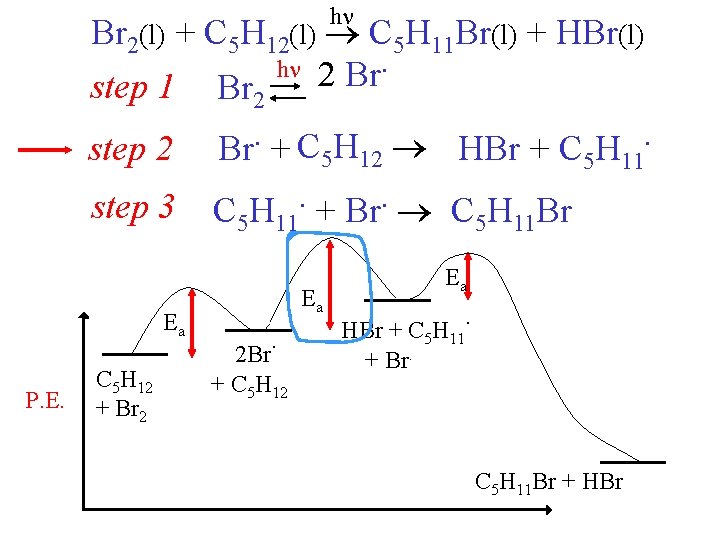
h Br 2(l) + C 5 H 12(l) C 5 H 11 Br(l) + HBr(l) h 2 Br. step 1 Br 2 step 2 Br. + C 5 H 12 HBr + C 5 H 11. step 3 . . C 5 H 11 + Br C 5 H 11 Br Ea P. E. C 5 H 12 + Br 2 Ea . 2 Br + C 5 H 12 Ea . HBr + C 5 H 11 + Br. C 5 H 11 Br + HBr
- Slides: 16