Review Periodic Table Mendeleev arranged in order of

Review Periodic Table • Mendeleev- arranged in order of increasing atomic mass. • Moseley – in order of increasing atomic number • Atomic mass = #protons + # neutrons, atomic number = #protons • Period- row of elements, properties change gradually • Group – column of elements, similar properties • Electron configuration – all the elements in a group have the same configuration of electrons in their outer shell

In the beginning • When God started building the atoms of elements he began with a design. We may have discovered that design. • He began adding electrons, protons and neutrons according to the design. • The first element had 1 electron and 1 proton and either 0, 1 or 2 neutrons. • The second element had 2 electrons and 2 protons and either 1, 2 or 3 neutrons. • Simulation

Uncertainty Principle W. Heisenberg 1901 -1976 The problem of defining the nature of electrons in atoms was solved by W. Heisenberg. We cannot simultaneously define the position and momentum (= m • v) of an electron. We can define e- energy exactly but we must accept the limitation that we do not know the exact position of the e-.

Electron Configuration • Electrons within the electron cloud have different energy levels. • Each electron is assigned 4 different “quantum numbers”. No two electrons within an atom can have the same quantum numbers. – Energy Level quantum number, there are 7 levels (17) – Shape quantum number -suborbits for electrons s, p, d, f – Orientation quantum number – Spin quantum number- right handed or left handed

QUANTUM NUMBERS Each orbital is a function of 3 quantum numbers: n, l, and ml n the principal quantum number or shell (or size and energy level) l the angular momentum quantum number or subshell (or shape) ml the magnetic quantum number designates an orbital within a subshell (or the orientation in space) In addition each electron has 1 of 2 Spins

QUANTUM NUMBERS Symbol Values Description n (major) 1, 2, 3, . . Orbital size and energy (shell) l (angular) 0, 1, 2, . . n-1 Orbital shape or type (subshell) ml (magnetic) - l. . 0. . + l Orbital orientation # of orbitals in subshell = 2 l + 1

Shells and Subshells When n = 1, then l = 0 and ml = 0 Therefore, in n = 1, there is 1 type of subshell and that subshell has a single orbital (ml has a single value 1 orbital) This subshell is labeled s Each n value (shell) has 1 orbital labeled s, and it is SPHERICAL in shape.

s Orbitals All s orbitals are spherical in shape.

p Orbitals When n = 2, then l = 0 and 1 Therefore, in the n = 2 shell there are 2 types of orbitals — 2 subshells For l = 0 ml = 0 this is a s subshell For l = 1 ml = -1, 0, +1 this is a p orbitals subshell with 3
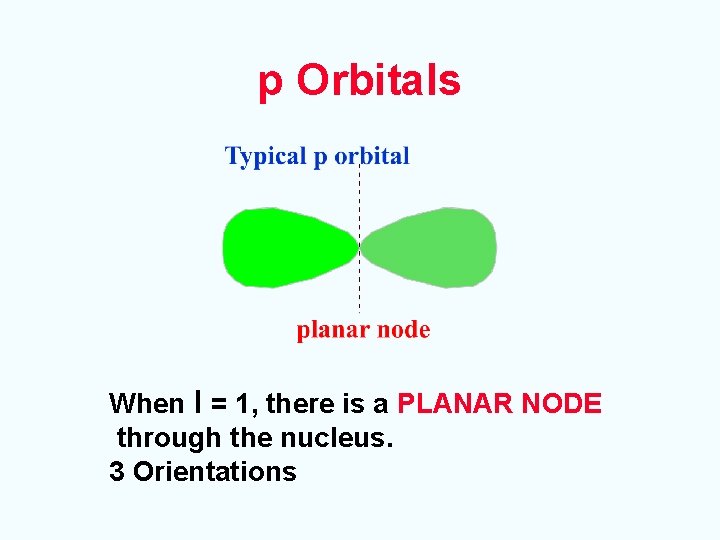
p Orbitals When l = 1, there is a PLANAR NODE through the nucleus. 3 Orientations

p Orbitals A p orbital The three p orbitals lie 90 o apart in space

d Orbitals When n = 3, l = 0, 1, 2 and so there are 3 subshells in the shell. For l = 0, ml = 0 s subshell with single orbital For l = 1, ml = -1, 0, +1 p subshell with 3 orbitals For l = 2, ml = -2, -1, 0, +1, +2 d subshell with 5 orbitals (orientations)

d Orbitals s orbitals have no planar node (l = 0) and so are spherical. p orbitals have l = 1, have 1 planar node, and are “dumbbell” shaped. This means d orbitals (with l = 2) have 2 planar nodes

d Orbitals

f Orbitals When n = 4, l = 0, 1, 2, 3 so there are 4 types of subshells in the shell. For l = 0, ml = 0 s subshell with single orbital For l = 1, ml = -1, 0, +1 p subshell with 3 orbitals For l = 2, ml = -2, -1, 0, +1, +2 d subshell with 5 orbitals For l = 3, ml = -3, -2, -1, 0, +1, +2, +3 f subshell with 7 orbitals

f Orbitals

Arrangement of Electrons in Atoms Electrons are arranged in Various Way of Describing Energy Levels SHELLS (n) { SUBSHELLS (l) ORBITALS (ml)

Arrangement of Electrons in Atoms Each orbital can be assigned no more than 2 electrons! This is tied to the existence of a 4 th quantum number, the electron spin, ms.

Electron Spin, ms Any spinning electric charge creates a magnetic field, comparable to N and S. There are two spin directions that are designated by ms = +1/2 and -1/2.

Pauli Exclusion Principle No two electrons in the same atom can have the same set of 4 quantum numbers. That is, each electron has a unique address.

Aufbau Principle Page 111 • An electron occupies the lowest energy orbital that can receive it. • Fill lower energy orbitals first • Energy levels overlap • 1 s, 2 p, 3 s, 3 p, 4 s, 3 d, 4 p, 5 s, 4 d, 5 p, 6 s, 4 f, 5 d, 6 p, 7 s, 5 f, 6 d
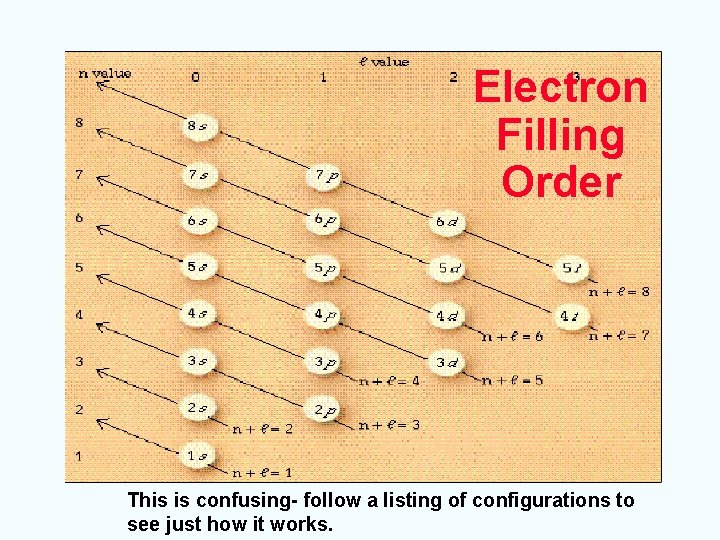
Electron Filling Order This is confusing- follow a listing of configurations to see just how it works.

Hund’s Rule • Orbitals of equal energy are each occupied by one electron before any orbital is occupied by a second electron, and all electrons in singly occupied orbitals must have the same spin state.

Writing Atomic Electron Configurations There are two ways of writing configurations. One is called spectroscopic notation.

Writing Atomic Electron Configurations The other method of writing econfigurations is called the orbital box notation. One electron has n = 1, l = 0, ms = + 1/2 Other electron has n = 1, l = 0, ms = - 1/2

See Electron Configuration in Chem. Land.

Lithium Group 1 A Atomic number = 3 1 s 22 s 1 3 total electrons
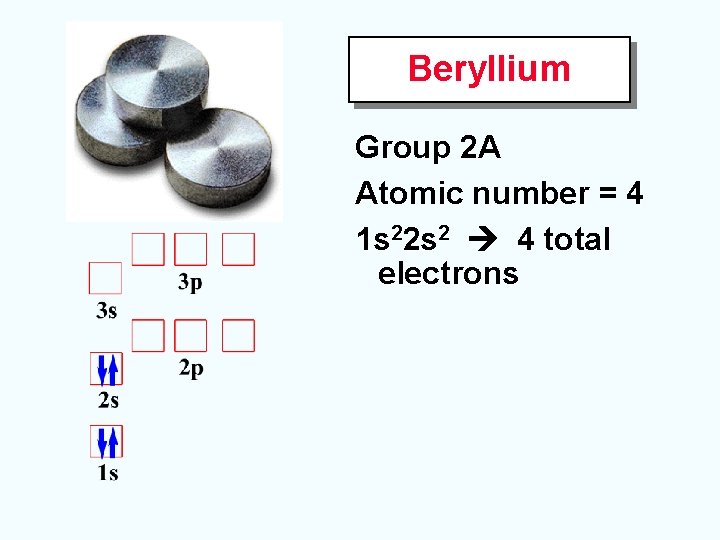
Beryllium Group 2 A Atomic number = 4 1 s 22 s 2 4 total electrons
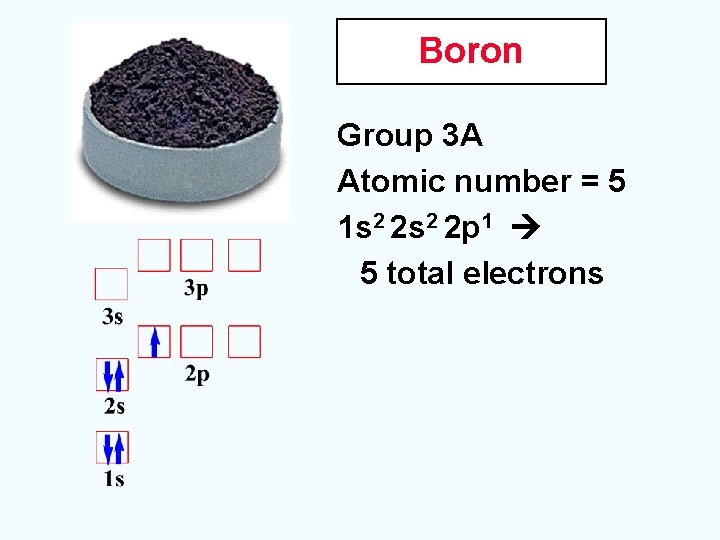
Boron Group 3 A Atomic number = 5 1 s 2 2 p 1 5 total electrons

Carbon Group 4 A Atomic number = 6 1 s 2 2 p 2 6 total electrons Here we see for the first time HUND’S RULE. When placing electrons in a set of orbitals having the same energy, we place a single electron in each orbital as long as possible.

Nitrogen Group 5 A Atomic number = 7 1 s 2 2 p 3 7 total electrons
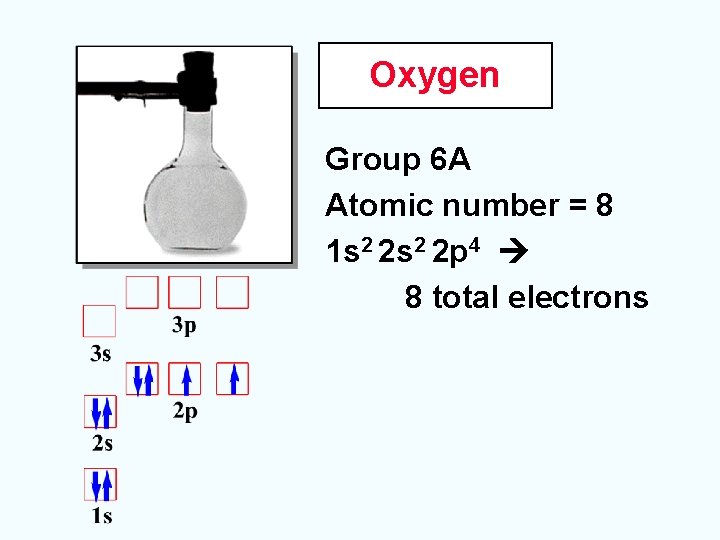
Oxygen Group 6 A Atomic number = 8 1 s 2 2 p 4 8 total electrons

Fluorine Group 7 A Atomic number = 9 1 s 2 2 p 5 9 total electrons
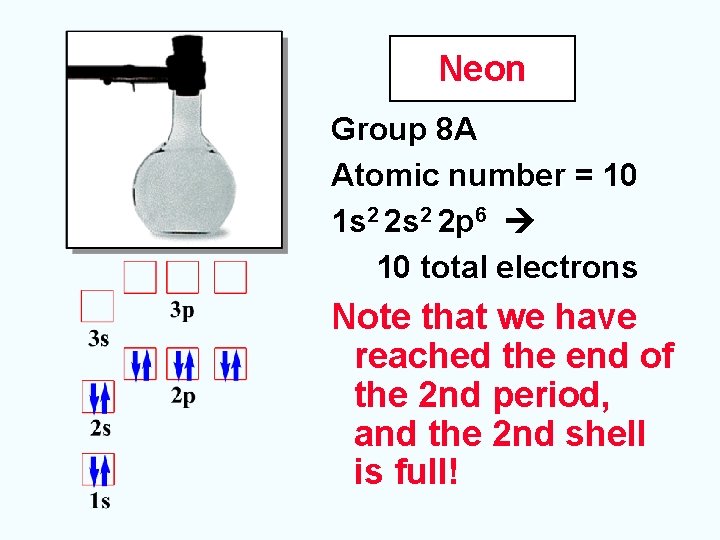
Neon Group 8 A Atomic number = 10 1 s 2 2 p 6 10 total electrons Note that we have reached the end of the 2 nd period, and the 2 nd shell is full!

Electrons in Atoms When n = 1, then l = 0 this shell has a single orbital (1 s) to which 2 e- can be assigned. When n = 2, then l = 0, 1 l = 0 is the 2 s orbital which has 2 el = 1 is the three 2 p orbitals which have 6 e. TOTAL = 8 e- in 4 orbitals

Electrons in Atoms When n = 3, then l = 0, 1, 2 l = 0 is the 3 s orbital l = 1 is the three 3 p orbitals l = 2 is the five 3 d orbitals TOTAL = 2 e 6 e 10 e- 18 e- in 9 orbitals

Electrons in Atoms When n = 4, then l = 0, 1, 2, 3 4 s orbital three 4 p orbitals five 4 d orbitals seven 4 f orbitals TOTAL = Chem. Land: 2 e 6 e 10 e 14 e 32 e- Atomic Structure Quantum Numbers
- Slides: 37