REVIEW OF CLINICAL MICROBIOLOGY PART II MJ Weintraub

REVIEW OF CLINICAL MICROBIOLOGY PART II MJ Weintraub, DVM

Prokaryotes Bacteria • Cell wall/envelope structure – GN, GP, AF & wall less • Genome • Morphology, arrangements, capsule/slime layer, flagella, fimbriae, pilus • Horizontal gene transfer mechanisms • 70 s ribosome • 1 Circular, double stranded DNA chromosome/genome • Plasmids: extrachromosomal, circular, ds. DNA, self replicating

Eukaryotes Fungi, Protozoa & Helminths • • Organelles 80 s ribosome Genome in nucleus Protozoa – flagella, cilia, pseudopods or non motile – Some cysts, trophozoites • Fungi – – Dimorphism Chitin in cell wall Ergosterols plasma membranes Some capsules • Helminths – Developmental stages if any

Viruses & Prions • Viruses – – – Intracellular obligate parasites Acellular agent RNA or DNA genomes Capsid &/or envelope Bacteriophages • Prions – Infectious pieces of protein

Common Stains - Bacteria • Gram – Understand technical errors – GN & GP – Endospores – clear within cell • Acid Fast – AF – red & non AF – blue – Ziehl Neelsen (heat) – Kinyoun (cold detergent) • Acridine Orange fluochrome – Bacteria difficult id in a specimen – blood

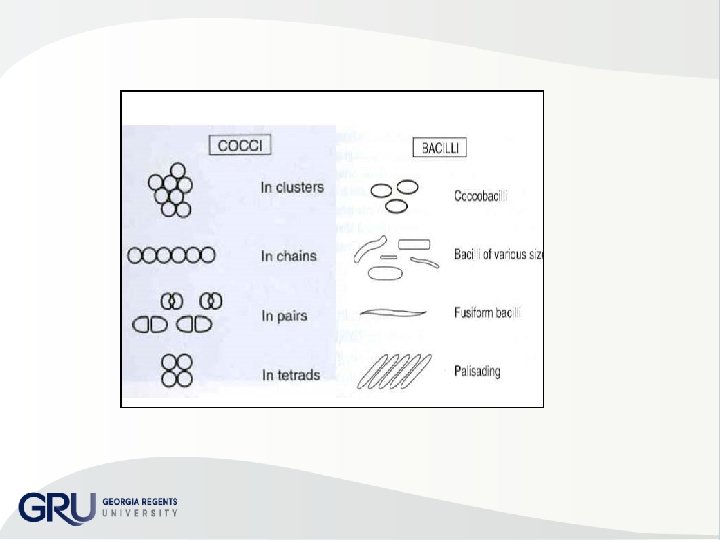
COCCI – – Single or pairs (diplococci) Chains (streptococci) Clusters (staphylococci) tetrads BACILLI • vary size & length • Ends may be square or rounded • Long & filamentous • Coccobacilli very short • Fusiform tapered or pointed ends • Curved • Pleomorphic species varies shape within pure culture • Single or chains or palisades SPIROCHETES helical turns – Vary in length & number

Common Stains - Bacteria • Methylene Blue – Metachromatic granules Corynebacterium diphtheriae – AF counterstain • Endospore – Red/pink endospore green living cell

Common Stains - Fungi • Lactophenol Cotton Blue (LCB) – Medically important fungi • Calcofluor White – fluorochrome – Fungi – bright apple green or blue white • India ink – Cryptococcus neoformans capsule

Types of Growth Media • Minimal media – Contents are simple – Not usually used in clinical microbiology lab • Nutrient media – Complex and made of extracts of meat or soy beans – Ex. Nutrient broth, Trypticase Soy Broth, TSA agar • Enriched media – Contains added growth factors, such as blood, vitamins, and yeast extract – Ex. Blood agar & Chocolate agar

Types of Media • Selective media – Contains additives that inhibit growth of some bacteria but supports others to grow – Mac. Conkey, CNA, Mannitol Salt • Differential media – See metabolic differences between groups or species of bacteria – Mac. Conkey, Mannitol Salt • Transport medium – Holding medium – Preserves viability of microorganisms in specimen but does not allow multiplication – Ex. Stuart broth, Cary Blair
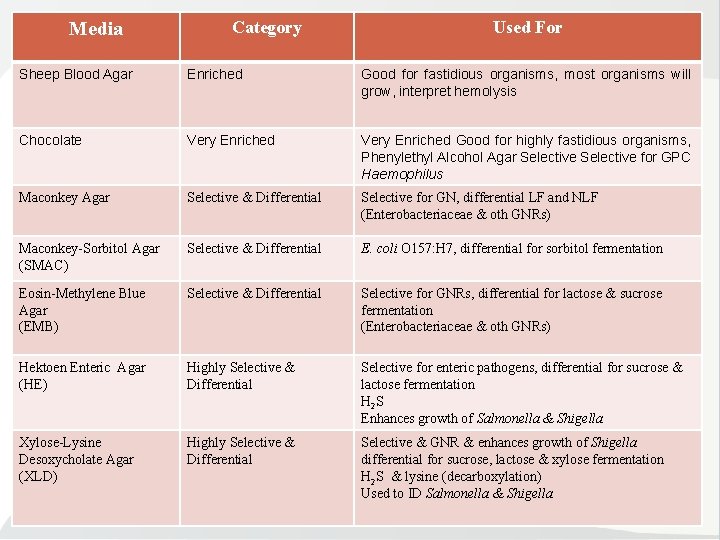
Media Category Used For Sheep Blood Agar Enriched Good for fastidious organisms, most organisms will grow, interpret hemolysis Chocolate Very Enriched Good for highly fastidious organisms, Phenylethyl Alcohol Agar Selective for GPC Haemophilus Maconkey Agar Selective & Differential Selective for GN, differential LF and NLF (Enterobacteriaceae & oth GNRs) Maconkey Sorbitol Agar (SMAC) Selective & Differential E. coli O 157: H 7, differential for sorbitol fermentation Eosin Methylene Blue Agar (EMB) Selective & Differential Selective for GNRs, differential for lactose & sucrose fermentation (Enterobacteriaceae & oth GNRs) Hektoen Enteric Agar (HE) Highly Selective & Differential Selective for enteric pathogens, differential for sucrose & lactose fermentation H 2 S Enhances growth of Salmonella & Shigella Xylose Lysine Desoxycholate Agar (XLD) Highly Selective & Differential Selective & GNR & enhances growth of Shigella differential for sucrose, lactose & xylose fermentation H 2 S & lysine (decarboxylation) Used to ID Salmonella & Shigella
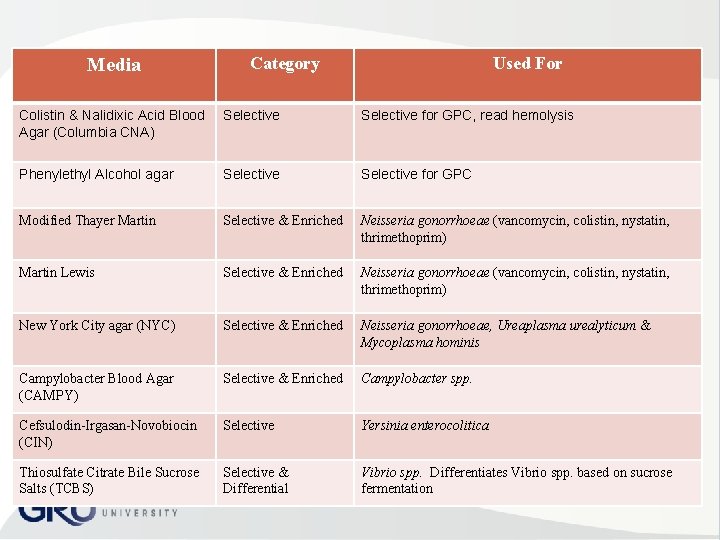
Media Category Used For Colistin & Nalidixic Acid Blood Agar (Columbia CNA) Selective for GPC, read hemolysis Phenylethyl Alcohol agar Selective for GPC Modified Thayer Martin Selective & Enriched Neisseria gonorrhoeae (vancomycin, colistin, nystatin, thrimethoprim) Martin Lewis Selective & Enriched Neisseria gonorrhoeae (vancomycin, colistin, nystatin, thrimethoprim) New York City agar (NYC) Selective & Enriched Neisseria gonorrhoeae, Ureaplasma urealyticum & Mycoplasma hominis Campylobacter Blood Agar (CAMPY) Selective & Enriched Campylobacter spp. Cefsulodin Irgasan Novobiocin (CIN) Selective Yersinia enterocolitica Thiosulfate Citrate Bile Sucrose Salts (TCBS) Selective & Differential Vibrio spp. Differentiates Vibrio spp. based on sucrose fermentation
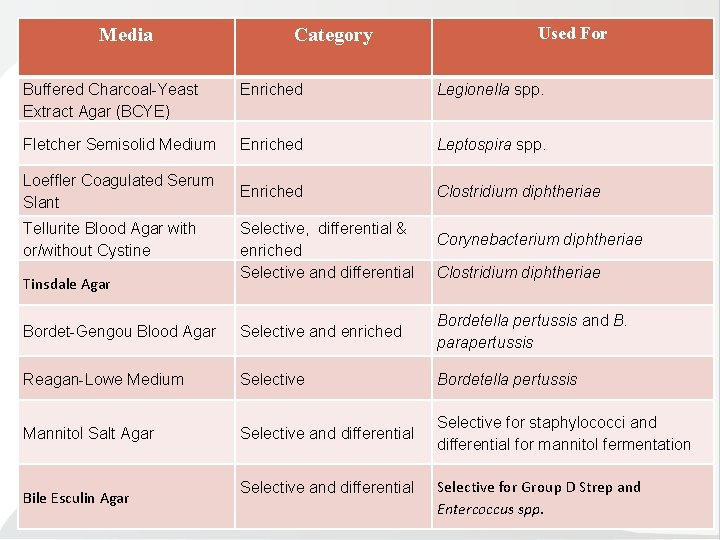
Media Category Used For Buffered Charcoal Yeast Extract Agar (BCYE) Enriched Legionella spp. Fletcher Semisolid Medium Enriched Leptospira spp. Loeffler Coagulated Serum Slant Enriched Clostridium diphtheriae Tellurite Blood Agar with or/without Cystine Tinsdale Agar Selective, differential & enriched Selective and differential Corynebacterium diphtheriae Clostridium diphtheriae Bordet Gengou Blood Agar Selective and enriched Bordetella pertussis and B. parapertussis Reagan Lowe Medium Selective Bordetella pertussis Mannitol Salt Agar Selective and differential Selective for staphylococci and differential for mannitol fermentation Bile Esculin Agar Selective and differential Selective for Group D Strep and Entercoccus spp.
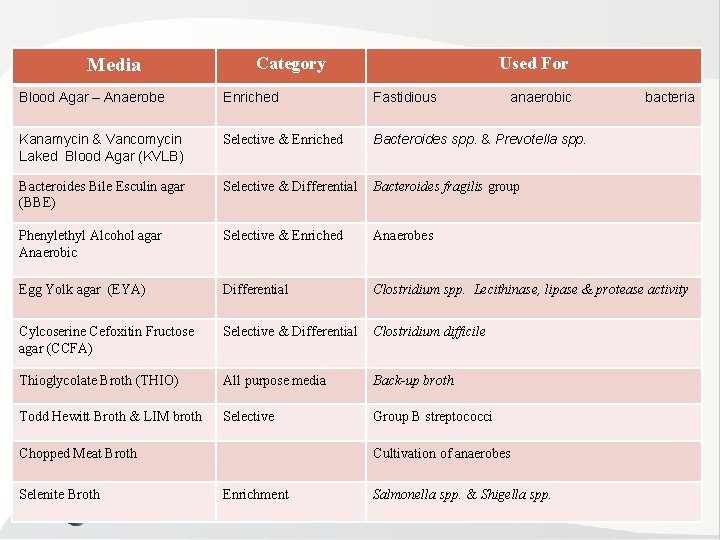
Media Category Used For Blood Agar – Anaerobe Enriched Fastidious Kanamycin & Vancomycin Laked Blood Agar (KVLB) Selective & Enriched Bacteroides spp. & Prevotella spp. Bacteroides Bile Esculin agar (BBE) Selective & Differential Bacteroides fragilis group Phenylethyl Alcohol agar Anaerobic Selective & Enriched Anaerobes Egg Yolk agar (EYA) Differential Clostridium spp. Lecithinase, lipase & protease activity Cylcoserine Cefoxitin Fructose agar (CCFA) Selective & Differential Clostridium difficile Thioglycolate Broth (THIO) All purpose media Back-up broth Todd Hewitt Broth & LIM broth Selective Group B streptococci Chopped Meat Broth Selenite Broth anaerobic Cultivation of anaerobes Enrichment Salmonella spp. & Shigella spp. bacteria

• JEMBEC transport system – Neisseria gonorrhoeae – Sodium bicarbonate & sodium citrate – Inhibition of contaminating bacteria and fungi

Environmental Factors • p. H – Most pathogenic bacteria grow best at a neutral p. H – media 7. 0 – 7. 5 • Temperature – Categories • Psychrophiles – optimal growth 10 to 20 O C • Mesophiles – optimal growth 20 to 40 O C • Thermophiles – optimal growth 50 to 60 O C

Atmospheric requirements • Obligate aerobe – require oxygen for growth • Aerotolerant anaerobe – can survive in presence of O 2 but do not use O 2 in metabolism • Obligate anaerobe – cannot grow in presence of oxygen • Facultative anaerobe – grow either with or without oxygen – Most opportunistic pathogens • Capnophilic – Best at increased CO 2 – Capnophilic bacteria need 5% 10% CO 2 • Microaerophilic – reduced level of oxygen to grow • Ex. Campylobacter spp. – requires 5% to 6% O 2

Fermentation & Respiration Bacteria use biochemical pathways to catabolize CH 2 O & produce energy • Respiration (Oxidation) – efficient energy generating process – Obligate aerobes & facultative anaerobes • Fermentation – Anaerobic process – Obligate & facultative anaerobes – Less efficient in energy generation

Fermentation vs. Oxidation • Used in biochemical testing based on: – presence mixture of end products accumulates in medium – Changes in p. H • Change in p. H – KIA, TSI, CTA CH 2 O, HE • Products of process: Methyl Red – mixed acids & VP acetoin • NOTE: if evaluating a CH 2 O & not glucose, no glucose should be in the media – Bacteria preferentially ferment glucose 1 st

Lactose Fermentation LF, NLF & s. LF • Key in ID of Enterobacteriaceae • 1 st Step – ß galactoside permease transports lactose across cell wall • 2 nd Step – ß galactosidase breaks galactoside bond releases glucose • all organisms that ferment lactose ferment glucose • s. LF fermenters lack or have a weak β galactoside permease – ONPG test to id s. LF as does not require β galactoside permease

GPC

Staphylococcus spp. • • (+) Catalase Non motile Non spore forming Most are facultative anaerobes – � except S. saccharolyticus • • S. aureus S. epidermidis S. saprophyticus S. lugdunensis – More virulent, may have mec. A gene

Staphylococcus spp. Isolation • SBA • Thioglycollate broth • Selective media □ Mannitol Salt agar □ Columbia Colistin Nalidixic Acid agar (CNA) □ Phenylethyl alcohol agar (PEA) General characteristics • GPC, in clusters • Colonies creamy, white, golden & may be “buttery looking” • Some β hemolytic

COAGULASE TESTS SLIDE TEST • Cell bound coagulase (Clumping Factor) • enzyme that clots human, rabbit, or pig plasma • If (+) reported as Staphylococcus aureus • If ( ) the tube test must be done TUBE TEST • Free coagulase (staphylocoagulase) • extracellular enzyme that causes clot to form when bacterial are incubated in plasma • If (+) reported as Staphylococcus aureus – 5% of S. aureus do not produce cell bound coagulase (CF) • If ( ) reported as Co. NS

• Coagulase – (+) Staphylococcus aureus – ( ) other staphylococci (Co. NS) – S. lugdunensis • (+) Cellbound (CF) coagulase & ( ) free (staphylocoagulase) • Novobiocin on Co. NS urine isolates – (R) Staphylococcus saprophyticus – (S) other Co. NS • Hemolysis – β: Staphylococcus aureus – No hemolysis: S. epidermidis & S. saprophyticus –
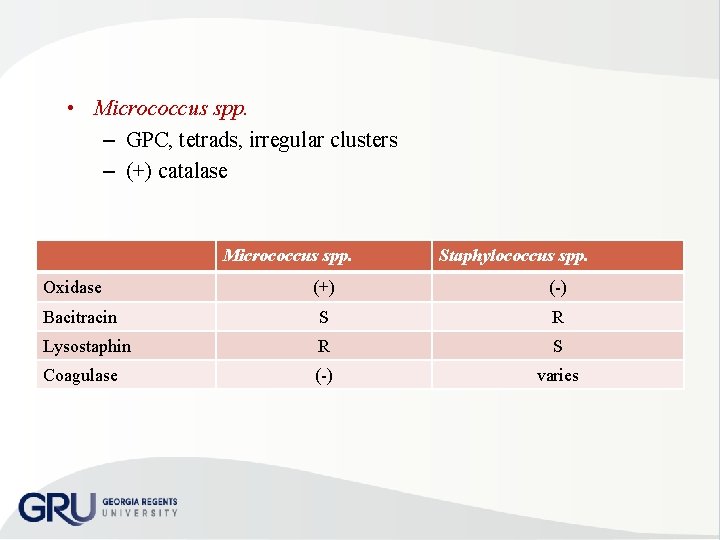
• Micrococcus spp. – GPC, tetrads, irregular clusters – (+) catalase Micrococcus spp. Oxidase Staphylococcus spp. (+) ( ) Bacitracin S R Lysostaphin R S Coagulase ( ) varies

• Staphylococcus aureus – must perform antimicrobial susceptibility on all isolates – Most penicillin(R) – beta lactamases • MRSA, HA MRSA, CA MRSA – Vancomycin VISA & VRSA MRSA KEY TEST • Oxacillin susceptibility but now Cefoxtin (better inducer of mec. A mediated resistance) is preferred • GOLD STANDARD – Detection mec. A gene – same day tests: Nucleic Acid probes or PCR D TEST • Macrolide resistance • Inducible clindamycin resistance presence of erythromycin

Streptococcus spp. • GPC, chains, pairs • ( ) catalase • Most facultative anaerobes • Streptococcus spp. • Enterococci spp. • LANCEFIELD CLASSIFICATION – C carbohydrate on cell wall – A, B, C, D, F, G & N – most common
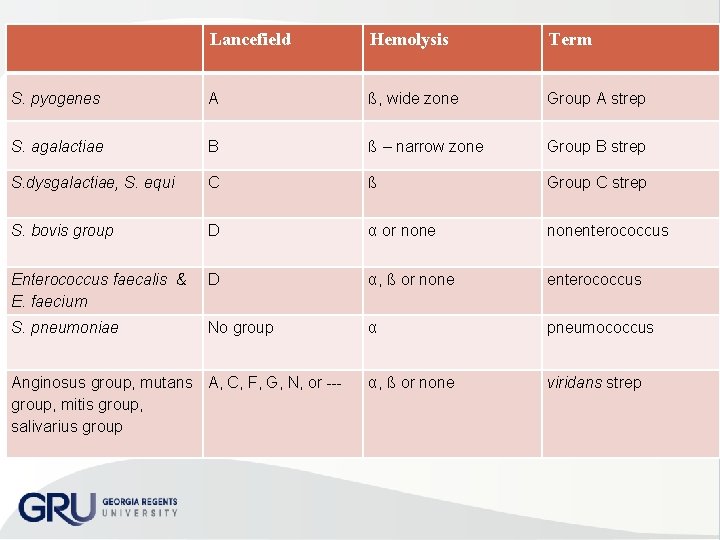
Lancefield Hemolysis Term S. pyogenes A ß, wide zone Group A strep S. agalactiae B ß – narrow zone Group B strep S. dysgalactiae, S. equi C ß Group C strep S. bovis group D α or nonenterococcus Enterococcus faecalis & E. faecium D α, ß or none enterococcus S. pneumoniae No group α pneumococcus α, ß or none viridans strep Anginosus group, mutans A, C, F, G, N, or group, mitis group, salivarius group

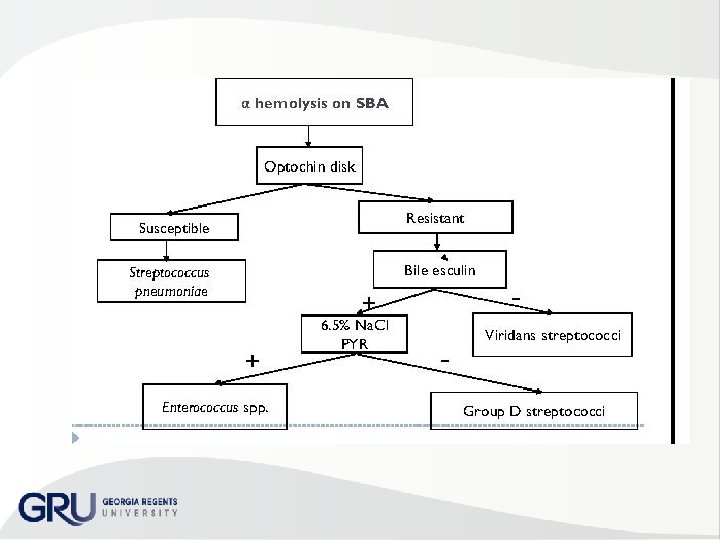
Susceptibility Tests • Bacitracin – identify Group A strep – Low concentration 0. 04 U selectively inhibits growth of Group A strep • Sulfamethoxazole & Trimethoprim (SXT) – used with bacitracin to improve accuracy Group A strep – Groups A & B are resistant to SXT • Optochin susceptibility – used on α hemolytic strep – ethylhydrocupreine hydrochloride – Streptococcus pneumoniae is sensitive to optochin

• Bile Solubility (sodium deoxycholate & sodium taurocholate) – Correlates well with optochin susceptibility – (+) Streptococcus pneumoniae in solution lyses & solution clears – ( ) viridans streptococci do not lyse • Hippurate hydrolysis – Differentiate Group B strep from other ß hemolytic strep – (+) Streptococcus agalactiae (GBS) • PYR hydrolysis test – Substrate is L pyrrolidonyl ß naphthylamide & L pyroglutamic acid ß naphthylamide – (+) Streptococcus pyogenes & Enterococcus spp. – ( ) other streptococci – more specific for Group A strep than bacitracin – (+) Aerococcus spp. & Gemella spp

• CAMP test – CAMP factor – PRES ID Group B strep – Enhanced hemolysis • ß hemolytic Staphylococcus aureus & CAMP factor producing β- hemolytic or some non hemolytic Group B strep – (+) arrow shaped hemolysis pattern

• Bile Esculin – Grow in presence of 40% bile – Hydrolyze esculin – black complex – (+) Group D streptococcus & Enterococcus spp. – ( ) viridans Strep • 6. 5% sodium chloride broth – (+) Enterococcus spp. – ( ) Group D Enterococcus faecalis • Leucine Amino. Peptidase test (LAP) (+) Streptococcus & Enterococcus spp. & Pediococcus spp. – ( ) Aerococcus spp. & Leuconostoc spp.

Susceptibility Testing • Penicillin – drug of choice for most streptococcal infections – Penicillin resistant organisms • Some Streptococcus pneumoniae & viridans streptococci • Vancomycin – Effective against GPC organisms – Vancomycin resistance now being encountered – Enterococci & some streptococci are showing resistance

Streptococcus pyogenes (Group A) • Pharyngitis & tonsillitis • Scarlet fever, skin infections, and other septic infections • Rheumatic fever, acute glomerulonephritis Laboratory • • Pinpoint colonies, ß hemolysis (+) PYR (S) Bacitracin ( ) Hippurate hydrolysis ( ) CAMP ( ) Bile esculin ( ) 6. 5% Na. Cl

Streptococcus agalactiae • invasive disease in newborns • Elderly, immunodeficient & women who become ill after childbirth or abortion • All pregnant women should be screened for GBS (group B Stept) at 35 37 weeks gestation • Laboratory – Grayish white colonies, small zone of ß hemolysis – ( ) catalase – (+) Hippurate hydrolysis – (+) CAMP test
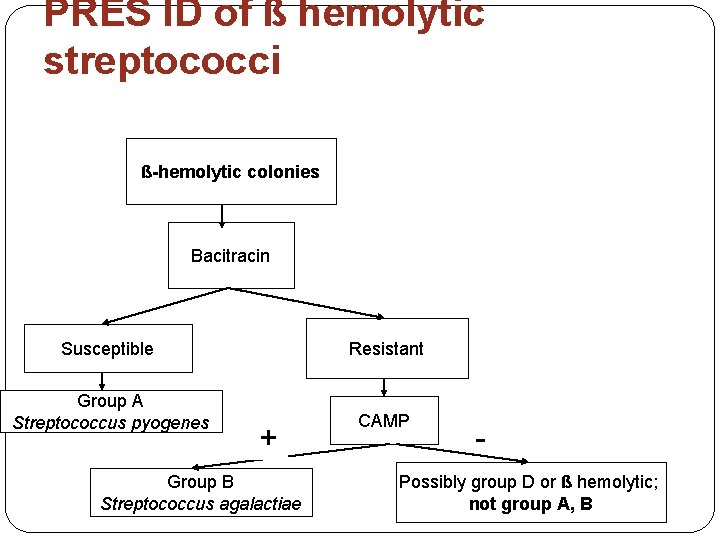
PRES ID of ß hemolytic streptococci ß-hemolytic colonies Bacitracin Susceptible Resistant Group A Streptococcus pyogenes CAMP + Group B Streptococcus agalactiae Possibly group D or ß hemolytic; not group A, B

Group D streptococci • Streptococcus bovis group • group D antigen • endocarditis, UTI, abscesses, wound infections & associated bacteremia resulting from presence of gi tumors • Penicillin – (S) Group D – (R) Enterococcus spp. , usually • Laboratory – small, translucent colonies α or no hemolysis – (+) Bile Esculin – ( ) 6. 5% Na. Cl – ( ) PYR

Enterococcus spp. • • Normal flora in intestinal tract Enterococcus faecalis – most common isolate Enterococcus faecium Group D antigen UTIs followed by bacteremia & endocarditis Show resistance to many antibiotics VRE – Vancomycin Resistant Enterococcus Laboratory – Β or α or no hemolysis – May have pseudocatalase reaction – (+) Bile Esculin – (+) 6. 5% Na. Cl – (+) PYR
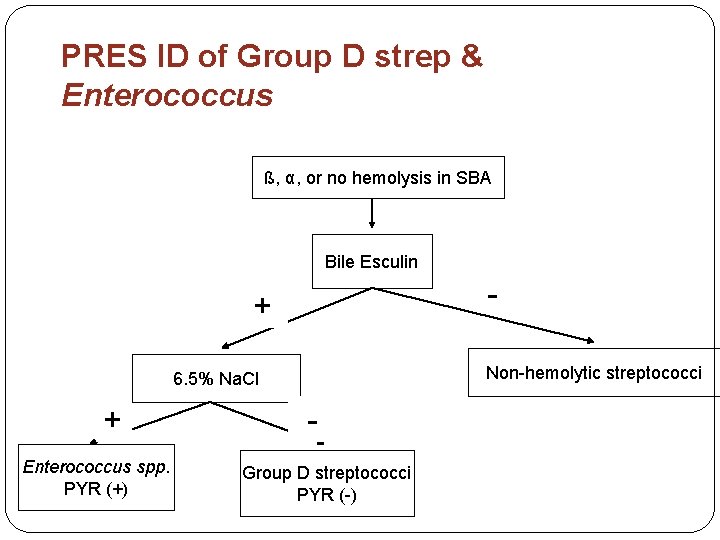
PRES ID of Group D strep & Enterococcus ß, α, or no hemolysis in SBA Bile Esculin + Non hemolytic streptococci 6. 5% Na. Cl + Enterococcus spp. PYR (+) Group D streptococci PYR ( )

Streptococcus pneumoniae pneumococcus • Normal RT flora • Pneumonia, sinusitis, otitis media, bacteremia, meningitis & 30% acute septicemia • Quellung reaction – capsule swelling • allows ID of Streptococcus pneumoniae & specific serotypes • treat with penicillin but some strains show resistance & are treated with erythromycin or chloramphenicol • Vaccine – childhood & > 50 yrs.

Streptococcus pneumoniae Laboratory diagnosis – GPC lancet shaped & usually have capsule – SBA or Choc – Some require increased CO 2 – Large zone of α hemolysis – round, glistening, wet, mucoid, dome shaped & with time collapse of colony's center – Optochin • (S) S. pneumoniae • (R) other α hemolytic streptococci – Bile solubility • (+) S. pneumoniae • ( ) other α hemolytic strep

viridans streptococci • flora URT, female genital tract & gi tract • S. anginosus group, S. mutans group, S. mitis group, S. salivarius group & S. bovis group • • Low Virulence, opportunist Most common cause of subacute bacterial endocarditis gingivitis, dental caries, meningitis, abscesses, osteomyelitis & empyema Penicillin but some resistant strains reported Laboratory – SBA may show zone of α hemolysis – ( ) Bile solubility – (R) optochin

Abiotrophia spp. & Granulicatella spp. • Nutritionally Variant Streptococci (NVS) • Satellite colonies @ other bacteria • Bacteremia, endocarditis, otitis media • Should be suspected in blood cultures Laboratory – GPC resembling strep – Do not grow on subculture – Subculture • Staphyloccous aureus streak – Satellitism

GNC

Neisseria spp. • Aerobic, non motile, non spore forming • GNDC • (+) Oxidase • (+) Catalase – Except for: • N. elongata GNR ( ) catalase • N. weaveri GNR (+)catalase • Capnophilic • fastidious organisms, need enriched media & moisture for optimal recovery • Neisseria gonorrhoeae (true pathogen); N. meningitidis (commensal URT of carriers) & other Neisseria spp. (opportunistic pathogens)

Neisseria gonorrhoeae • urogenital tract infections & conjunctiva • Disseminated infections – joints, meningitis • Specimen: males–urethra; females–endocervix & rectum, pharynx & joint fluid • Best chance for recovery is direct plating of specimen to gonococcal selective media • incubate at 35 O C in 3 5% CO 2 • PCR assay N. gonorrhoeae vs. Chlamydia spp.

Neisseria gonorrhoeae Direct Microscopic exam • urogenital specimens • Not recommended for pharyngeal specimens • Evidence of gonococcal infection – GNDC intracellular from symptomatic male with discharge correlates at a rate of 95% with culture – Females have vaginal commensal flora that resemble gonococci – culture is necessary • Gram stain >5 PMNs/field but no bacteria may suggest Non. Gonococcal Urethritis (NGU) • � NGU rule out: Chlamydia trachomatis & Ureaplasma urealyticum

Neisseria gonorrhoeae • Requires chocolate agar (+) selective agents • Thayer-Martin (TM) • Vancomycin (GP) , Colistin (GN), Nystatin(fungi) • � Modified Thayer-Martin (MTM) – Vancomycin, Colistin, Nystatin, Trimethoprim (Proteus spp. ) • Martin Lewis (ML) – Vancomycin, Colistin, Anisomycin (fungi), Trimethoprim • New York City (NYC) – Vancomycin, Colistin, Amphotericin B (fungi), Trimethoprim

Neisseria gonorrhoeae PRES ID 1. Small, gray/tan, transluscent raised colonies 24 48 hrs. 2. Gram stain to verify GNDC kidney bean shaped – GNRs Kingella spp. & Acinetobacter spp. may grow on gonococcal media , may look similar, to differentiate, inoculated onto plate with 10 unit penicillin disc, growth on edge of zone of incubation to evaluate morphology 3. (+) Oxidase – Must be done on all isolates Antimicrobial resistance – All strains must be tested for ß-lactamase production – Must be performed on primary culture plate as plasmid can be lost on subculture

Neisseria gonorrhoeae Definitive Identification • Other oxidase (+) GNDC, can be recovered from gonococcal selective media Neisseria cinerea, N. meningitidis, Moraxella catarrhalis CH 2 O Utilization • Cystine Trypticase Agar (CTA) – (+) Glucose fermentation & ( ) Maltose, sucrose & lactose fermentation Chromogenic enzymes • Multitest • Matrix assisted Laser Desorption/Ionization Time of Flight (MALDI Tof) Non-culture • Nucleic Acid Amplification Test (NAATs)

Neisseria meningitidis meningococcus • meningitis, meningococcemia, rare pneumonia, purulent arthritis & endophthalmitis • nasopharynx & oropharynx of 3 30% of asymptomatic individuals Laboratory • CSF, blood, nasopharyngeal swabs & aspirates, sputum & urogenital sites • Direct exam – intracellular & extracellular GNDC & may have capsule – Helps to concentrate to specimen • grows on gonococcal selective agars, SBA & Choc • small, gray, sometimes mucoid, convex colonies • (+) Oxidase • (+) Catalase • CTA sugar: s (+) glucose & maltose fermentation & ( ) sucrose & lactose

Moraxella catarrhalis • GNDC • Normal commensal RT, opportunistic pathogen • Pneumonia, sinusitis, otitis media & systemic disease Laboratory • Grows on SBA & Chocolate • smooth, opaque, gray to white colonies “hockey puck” • Usually inhibited by colistin in gonococcal selective media • (+) Oxidase • (+) Catalase • Asaccharolytic in CH 2 O degradation tests � • (+) DNase • (+) Butyrate esterase

Fastidious GNR • HACEK • Haemophilus spp. • Aggregatibacter spp. (formerly Actinobacillus) • Cardiobacterium hominis • Eikenella corrodens • Kingella spp. • • • Capnocytophaga spp. Pasteurella spp. Brucella spp. Francisella spp. Legionella spp. Bordetella spp.

HACEK Group Characteristics – GNR/CB – Capnophilic – Important cause of endocarditis • flora of oral cavity – allows for introduction into blood stream Haemophilus spp. • GNCB, pleomorphic • Most nonpathogenic or opportunistic pathogens • X factor hemin & V factor (NAD) “para” only V factor • H. influenzae H. parainfluenzae H. haemolyticus • H. parahaemolyticus H. aphrophilus H. paraphrophilus • H. aegyptius H. ducreyi •

Haemophilus spp. • Hemolysis on 5% horse’s or rabbit’s blood to differentiate • SBA only X factor available – V factor dependent do not grow on SBA • Chocolate agar – Lysed rbcs by heat results in containing both X & V factors • Satellitism – Staphylococcus aureus, Streptococcus pneumoniae, Neisseria spp. produce V factor as a byproduct of metabolism

• Haemophilus influenzae – Meningitis – decrease frequency due to vaccine (Hib) – Epiglottitis • � Haemophilus aegyptius - acute contagious conjunctivitis “pinkeye” • � H. influenzae biogroup aegyptius conjunctivitis • � H. ducreyi Chancroid – painful, soft – highly communicable STI Genital Ulcer Disease (GUD) • Laboratory – Almost any specimen but commonly blood, CSF, middle ear exudates, joint fluids, URT & LRT specimens, swabs conjunctival swabs, vaginal swabs, abscess drainage – Prompt transportation & processing is critical

Haemophilus spp. • Culture – Does not grow on SBA, grows on Chocolate • H. ducreyi – Special media, hold for at least 7 days at 33 O C • H. aegyptius – Special media, hold for at least 4 days • Colony morphology – On chocolate translucent, tannish, moist, smooth, convex colonies with “mousy” or “bleach like” odor • Microscopic morphology on direct smear – GNCB to long filaments – H. ducreyi “school of fish” formation – H. influenzae – intra & extracellular

Haemophilus spp. • 1 st clue to a Haemophilus spp. – No Growth (NG) on SBA, MAC or other enteric agars – GNCB, pleomorphic on Chocolate agar • X & V factor test • Porphyrin test – (+) Porphyrin heme producing will fluoresce – ( ) Porphyrin non heme producing will not fluoresce • Many strains produce ß-lactamase – rapid tests – Chromogenic cephalosporin test – Acidometric test – If (+) strain is resistant to ampicillin & amoxicillin

• Aggregatibacter aphrophilus – X dependent & X independent strains – ( ) catalase – granular, yellow with center opaque zone colonies on Choc • Aggregatibacter actinomycetemcomitans o (+) catalase o BA “star” 4 6 points in center 100 x • Cardiobacterium hominis – GNR or GV – forms rosettes – Slow grow SBA & CHOC – may see pitting

Eikenella corrodens • • flora mouth & gi Usually polymicrobial infection, often trauma human fights or bites GNCB ( ) motility (+) oxidase ( ) Catalase asaccharolytic • often produce yellow pigmented colonies – @45% pit the agar (if see 1 st think Eikenella spp. ) – Bleach-like odor • NG on Mac or EMB

o Kingella spp. o Often follow viral infection or poor oral care or oral surgery o GN CB/short rods with squared ends, in pairs or short chains, tend resists decolorization o Nutritionally fastidious o “twitching” motility (no flagella) o Capnocytophaga Spp. – – – – o Resemble HACEK Normal flora mouth & oral cavities GNR Thin & fusiform Fastidious, pitting but fermentative May see gliding motility on agar surfaces Often yellow orange pigment Isolated from blood from granulocytopenic patients who have oral ulcers, juvenile periodontal disease, and endocarditis
Pasteurella multocida • • Most common isolated Pasteurella spp. Zoonosis GNCB, pleomorphic Bipolar staining Laboratory – SBA nonhemolytic – (+) oxidase – (+) catalase – weak TSI (glucose fermentation weak acid production) – (+) ODC – (+) Indole – (+) Urease – Does not grow on Mac

Brucella spp. • • Zoonosis Brucellosis, Undulant fever, Mediterranean, Crimean & Malta Fevers aerosol, percutaneous & oral routes of exposure remittent fevers, fluctuate regularly at consistent intervals & persist for days, months, or years • B. melitensis most common cause of brucellosis • Category B Select Biological Agent – Easily disseminated & moderate morbidity but low mortality – BSL 3 lab – lab exposure results in 30% 100% acquiring infection • Blood & bone marrow cultures performed with acute & convalescent sera for serologic testing

Brucella spp. Laboratory • Tiny, GNR/CB • facultative intracellular pathogen in phagocytes • Grow aerobically on Choc & BA in 18 hrs hold 4 days (also TM or Martin Lewis) • Smooth, raised & translucent colonies • 50% grow on MAC • ( ) motility • (+)oxidase • (+) catalase • (+) Nitrate reduction • (+) urease

Francisella spp. • Zoonosis –tularemia or “rabbit fever” • ingestion, inhalation, arthropod bite or contact infected tissues • Category A agent - easily disseminated (ID 50 = 50), high mortality rates • BSL 3 Laboratory diagnosis • tiny, poor staining GNCB • NG on Mac & may initially grow on SBA • CHOC, MTM, BCYE with cysteine for 36 48 hours • Smooth, gray white, raised colonies • (+) catalase weak • ( ) Oxidase • ( ) urease • (+) beta lactamase test • ( ) X & V factors

Legionella pneumonophila • Weakly staining tiny GNR, pleomorphic • Intracellular (phagocytes) & extracellular • Legionnaires’ disease & Pontiac fever Laboratory • Suspect if NG on SBA • Acid wash sputum & plate on selective media for best isolation • Subculture on BYCE & BYCE with L cysteine – 3 5 days “ground glass” center of gray & granular with pink/light blue periphery of colony – As age gray/white or blue/green, convex, glistening colonies • Urine for antigen detection – Detects L. pneumophilia serogroup 1 soluble antigen

Bordetella pertussis • Whooping cough • One most highly communicable diseases of childhood • DPT vaccine Laboratory • Tiny GNR or GNCB • Slow growing 3 5 days • Regan-Lowe Charcoal media – Young colonies mercury droplets, as age turn whitish gray • Bordet-Gengou Potato Infusion agar – Small, white, domed “pearls” – B. pertussis & B. parapertussis – hemolytic • Nucleic acid detection by PCR – Primary rapid diagnostic tool – Nasopharyngeal swabs
ENTEROBACTERIACEAE

ENTEROBACTERIACEAE culture • SBA or Choc - Large, grayish, smooth colonies, some hemolytic • Mac. Conkey (MAC) lactose in media – LF pink colonies & NLF –tan/colorless colonies • Eosin-Methylene Blue (EMB) lactose & sucrose in media – LF Pink colonies – E. coli blue black/green sheen colonies – NLF & NSF translucent colonies

STOOL CULTURES Enhance Salmonella spp. & Shigella spp. growth HE Agar lactose & sucrose in media • LF &/or SF–yellow colonies • NLF & NSF –green colonies • H 2 S – black centers XLD Xylose Lysine Deoxycholate (lactose, sucrose & xylose) • LF &/or SF &/or XF–yellow colonies • NLF & NSF & NXF red colonies • H 2 S – black centers • Salmonella spp. (+) lysine decarboxylase

Enterobacteriaceae Identification • ( ) oxidase except Plesiomonas shigelloides • (+) glucose fermentation • (+) NO 3 reduction • (+) motile except Shigella spp. , Klebsiella spp. & *Yersinia spp. (+) motile at 250 C

KIA or TSI PRES ID of Enterics • KIA = lactose in slant & glucose & H 2 S in butt & +/ gas • TSIA= lactose & sucrose in slant; glucose & H 2 S in butt & +/ gas • Phenol red indicator – alkaline p. H= red & acid p. H=yellow • Slant=aerobic & butt=anaerobic NOTE: • Enterobacteriaceae if A/A or K/A • Not an Enterobacteriaceae if K/K or A/K

TSIA & KIA Interpretation

• ONPG o-Nitrophenyl p D galactopyranoside – Determines if slow LF • CH 2 O fermentation – ability to ferment a specific 1% concentration of carbohydrate • MRVP – Methyl red (MR) • Mixed acid fermentation pathway (strong acids) – Vogues Proskauer (VP) • Butylene glycol pathway byproduct acetoin (acetylmethylcarbinol) • (+) Klebsiella spp. , Enterobacter spp. Serratia spp. & Hafnia sp. group • Decarboxylase. Tests – Decarboxylase remove carboxyl group of an amino acid – Lysine, ornithine & arginine

• PAD Phenylalanine Deaminase – deaminates phenylalanine to phenylpyruvic acid – (+) PPM group (Proteus, Providencia & Morganella) • Citrate Utilization Test – Utilize sodium citrate as a sole carbon source • DNase – Extracellular DNase produced by some bacteria – (+) most Serratia spp. • Gelatin Liquefaction – Proteolytic enzymes break down gelatin into amino acids

• Indole – Kovacs or Ehrlich – Degradation products of amino acid tryptophan – Add paradimethylaminobenzaldehyde red color • SIM – H 2 S production – Indole production – Motility • Malonate – use sodium malonate as a sole carbon source • Motility

• Nitrate reduction - NO 3 to NO 2 or to N (gas) – 1 st Step add sulfanilic acid & N, N dimethy α naphthylamine • (+) Red – If no color change go to 2 nd Step add zinc » ( ) Red and (+) No color change • C �ytochrome oxidase – Oxidizes reduced cytochrome with molecular oxygen

• Lysine Iron agar (Slant/Butt) – Purple/purple = (+) Lysine decarboxylase – Purple/yellow butt = ( ) Lysine decarboxylase – Cherry red/ yellow butt = (+) Lysine deaminase – H 2 S production masks (+) lysine decarboxylase reaction • Urease – Breaks down urea to NH 3 which decreases p. H

Enteric Pathogens • Salmonella, Shigella, Aeromonas, Campylobacter, Yersinia, Vibrio and E. coli 0157: H 7, Plesiomonas shigelloides • � All fecal specimens should be routinely screened for – Salmonella, Shigella, and Campylobacter – Many labs routinely screen for E. coli 0157: H 7 • Serologic groupings for Salmonella spp. & Shigella spp. – Based on O antigens – Slide Test • 1 drop antisera onto slide & add saline emulsion containing isolate. If ( ) may be due to capsule & requires emulsion heated to 1000 C to remove capsule & then re test

Escherichia coli • MAC – LF, bile precipitation • SBA – may appear ß hemolytic • EMB – green metallic sheen • IMVi. C + + - • TSI= A/A (+) gas • ( ) for DNAse; urease & PAD LF

Escherichia coli • • • Most common cause UTIs Meningeal (especially newborn) Gastrointestinal infections Wound Bacteremia • Five major categories of diarrheogenic E. coli – Enteropathogenic (EPEC) – Enterotoxigenic (ETEC) – Enteroinvasive (EIEC) – Enteroadherent – Enterohemorrhagic (EHEC) serotype 0157: H 7 • produces Verotoxins I and II LF

Enterohemorrhagic E. coli (EHEC) 0157: H 7 strain • SMAC – MAC with sorbitol instead of lactose (SMAC) – E. coli 0157: H 7 does not ferment sorbitol • MUG – Screen for E. coli 0157: H 7 • Rarely produces Beta glucuronidse • 92% of other strains do produce it • SMAC (-) &/or ( ) MUG – Serotype to confirm E. coli 0157: H 7 with anti serum

KLEBSIELLA TRIBE LF • Klebsiella spp. , Enterobacter spp. , Serratia spp. , Pantoea spp. , Cronobacter spp. & Hafnia spp. • • • ( ) H 2 S (+) urease few ( ) MR (+) VP (+) Citrate most ( ) indole most KES – most IMVi. C - - + + except K. oxytoca +-++

Klebsiella spp. LF • K. pneumoniae – Nosocomial – Frequent cause of LRT infections – wound infections, UTI, bacteremia • K. oxytoca – infections similar to K. pneumoniae • IMVi. C - - + + except K. oxytoca indole (+) • • TSIA = A/A, no H 2 S, no gas ( ) motility (+) urea ( ) ornithine decarboxylase

Enterobacter spp. LF • • Enterobacter aerogenes E. cloacae Isolated from wounds, urine, blood & CSF Mucoid colonies (+) motility IMVi. C - - + + TSI A/A, no H 2 S, no gas • E. aerogenes = ( ) urease & ( ) arginine decarboxylase • E. cloacae = (+) urease (65%) & (+) arginine decarboxylase

Serratia spp. SLF • Serratia marcescens • S. liquefaciens • S. rubidaea • S. marcescens – Opportunistic pathogen – nosocomial outbreaks – UT ; RT, bacteremia nurseries, cardiac surgery & burn units • Resistant to a wide range of antimicrobials – Susceptibility test MUST be performed on every isolate – some pink pigment at 300 C & look like a LF MAC at 18 hr even though SLF
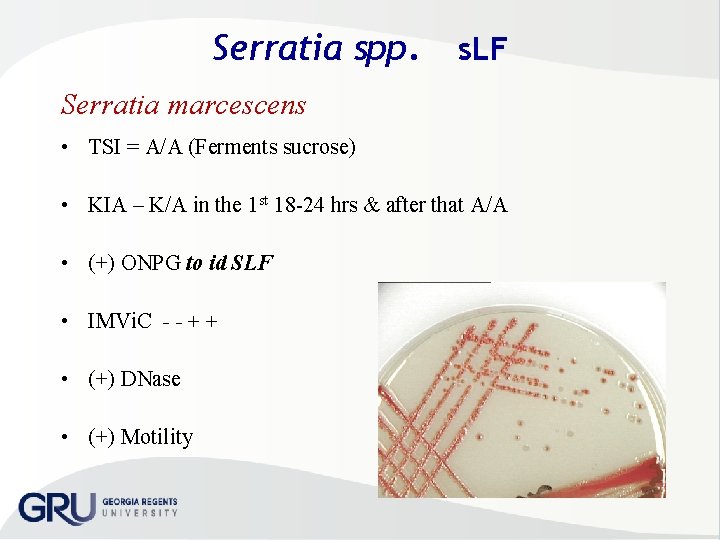
Serratia spp. s. LF Serratia marcescens • TSI = A/A (Ferments sucrose) • KIA – K/A in the 1 st 18 24 hrs & after that A/A • (+) ONPG to id SLF • IMVi. C - - + + • (+) DNase • (+) Motility

Proteus spp. , Morganella spp. & Providencia spp. NLF • Normal intestinal flora Opportunistic pathogens • Differentiate from other Enterobacteriaceae – (+) PAD – (+) Lysine deaminase • Proteus spp. – – – Proteus mirabilis Proteus vulgaris urine, wounds, ear s & bacteremia swarming colonies on non selective media SBA “burned chocolate” odor (+) H 2 S production

PPM Group • Morganella morganii – May UTI & diarrhea • Providencia spp. – P. rettgeri • UTIs & caused occasional nosocomial outbreaks – P. stuartii • nosocomial outbreaks burn units & been isolated from urine • cannot differentiate between Morganella & Providencia spp. with standard tests
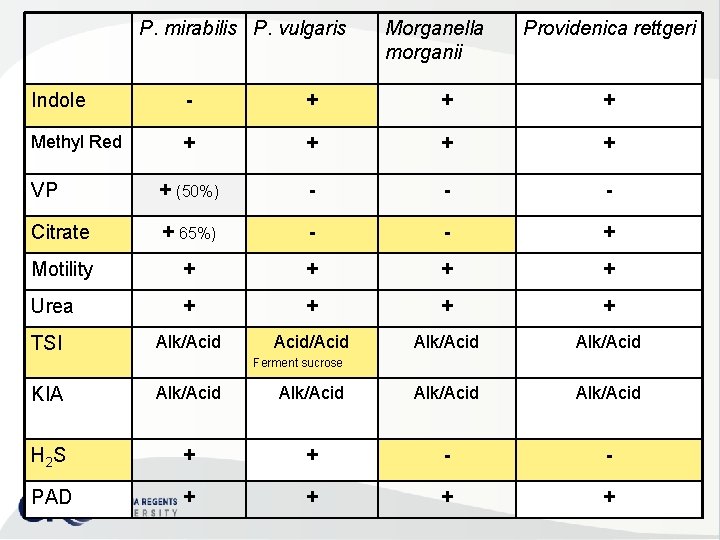
P. mirabilis P. vulgaris Morganella morganii Providenica rettgeri Indole - + + + Methyl Red + + VP + (50%) - - - Citrate + 65%) - - + Motility + + Urea + + Alk/Acid/Acid Alk/Acid TSI Ferment sucrose KIA Alk/Acid H 2 S + + - - PAD + +

Edwardsiella tarda NLF • • • Bacteremia & wound infections ( ) Urea (+) Lysine decarboxylase (+) H 2 S (+) Indole ( ) Citrate Citrobacter spp. LF • • (+) urea slowly (+) citrate (+) methyl red C. freundii – UTI, pneumonias, intra abdominal abscesses – (+) H 2 S & some do not ferment lactose & can resemble Salmonella when isolated from stools

Salmonella spp. • Typhoid fever – Most severe form of enteric fever is caused by S. serotype Typhi – Other enteric fevers S. serotype Paratyphi, S. serotype Choleraesuis • Non typhoidal bacteremia • Carrier state • MAC, HE & XLD • • • IMVi. C - + TSI = K/A, no gas, (+) H 2 S ( ) PAD ( ) Urease (+) lysine decarboxylase

Shigella spp. NLF • True Pathogen that causes bacillary dysentery – S. dysenteriae (group A) – S. flexneri (group B) – S. boydii (group C) – S. sonnei (group D) – most common isolate US • S. dysenteriae – blood, mucous & pus in stool • Media – MAC, HE & XLD – SS agar inhibits GP, most coliforms & swarming Proteus spp. • LF (pink) from NLF & H 2 S production

Shigella spp. NLF • • • ( ) motile (+) glucose fermentation without gas except some S. flexneri (+) gas ( ) Urea ( ) H 2 S ( ) lysine decarboxylase • � S. sonnei – (+) ornithine decarboxylase – SLF after 48 hrs & see pink colonies on Mac – (+) ONPG

Yersinia spp. NLF • Y. pestis – plague – – – GNR short, plump Wayson Stain - bipolar staining “safety-pin” Grows at 37 OC but likes 25 -30 O C best Class A Bioterrorism Agent (-) urease (-) motility at 250 C & 350 C • Y. pseudotuberculosis - Similar to plague – Pseudotubercles (caseous swellings) – (+) motility at 25 OC & (-) at 35 O C – (+) urease

Yersinia enterocolitica NLF � acute enteritis; appendicitis like syndrome, arthritis & erythema nodosum • contaminated meat, esp. pork (chitterlings), vacuum packed deli meet, • Optimal growth 25 – 30 O C • (+) motility at 25 OC & ( ) at 35 O C • TSI A/A ( ) H 2 S ferments sucrose • � CIN Selective Media • cefsulodin, irgasan, novobiocin, bile salts & CV inhibitory • Inhibits normal colon organisms better than Mac • Yersinia selective agar (YSA) base add mannitol to make differential – (+) Mannitol fermentation colony has red center
- Slides: 99