Review of Atoms Elements Molecules and Bonding Atoms

Review of Atoms, Elements, Molecules, and Bonding

Atoms • Atoms are the smallest unit of matter • They are made up of neutron, protons (+), and electrons (-) • Electrons rotate round the atomic nucleus (protons and neutrons) in energy levels • The outside energy levels are where valence electrons are located • The valence electrons are involved in bonding

Elements • An element is a pure substance that cannot be broken down into other substances • Ex: Carbon, Hydrogen, Oxygen, Nitrogen

Molecules • Molecules form when two or more elements bond together • Ex: H 2 O

How Bonding Works • Most common type of chemical bond: Covalent • Bonds form between valence electrons • Each energy level has a specific number of electrons that it can hold at any time • The first can hold up to 2 electrons • The second can hold up to 8 electrons

How Bonding Works • A partially-filled energy level is not stable • Atoms become more stable by losing electrons or attracting electrons from other atoms • This results in the chemical formation of chemical bonds between atoms
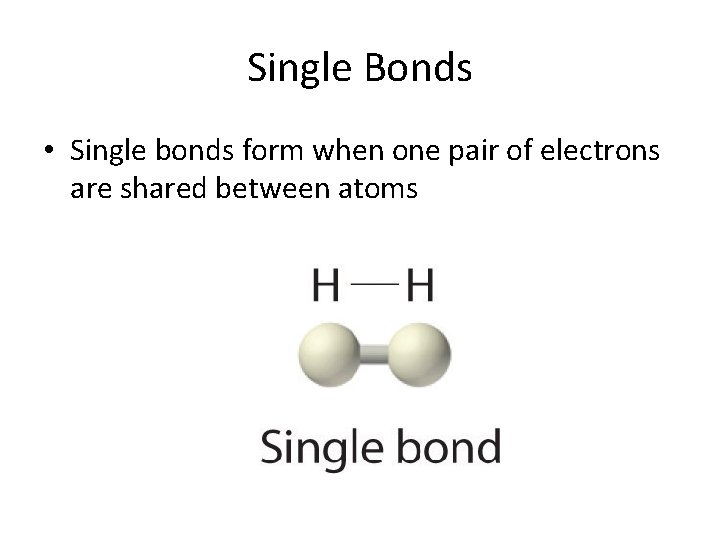
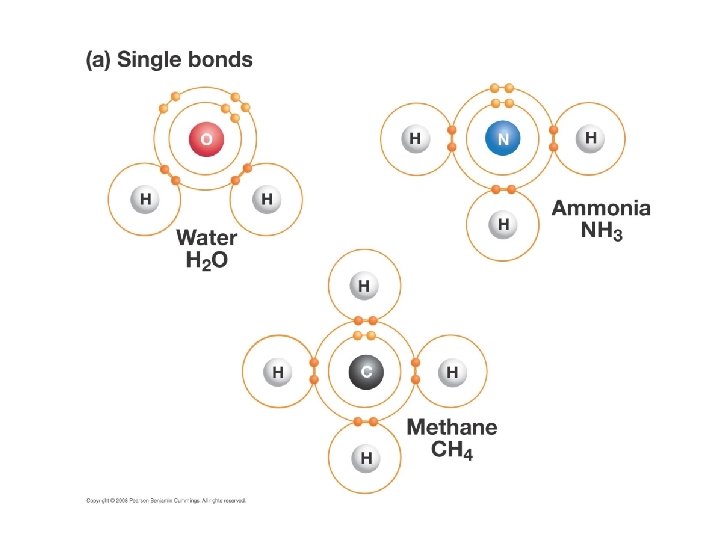
Single Bonds • Single bonds form when one pair of electrons are shared between atoms
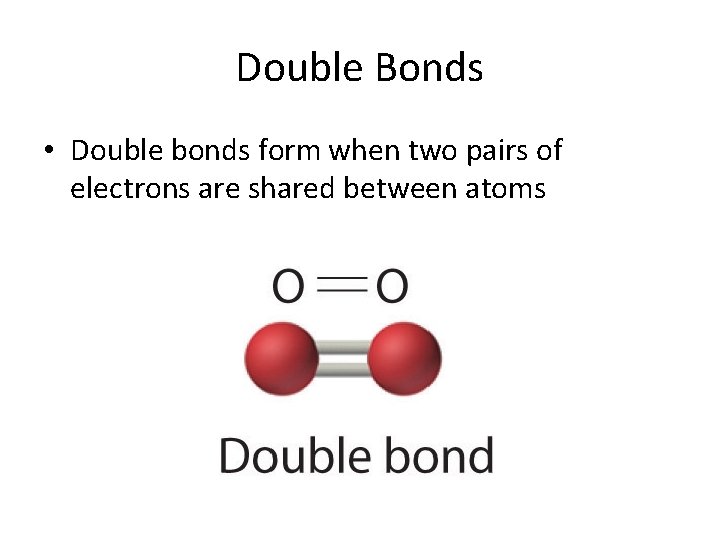
Double Bonds • Double bonds form when two pairs of electrons are shared between atoms

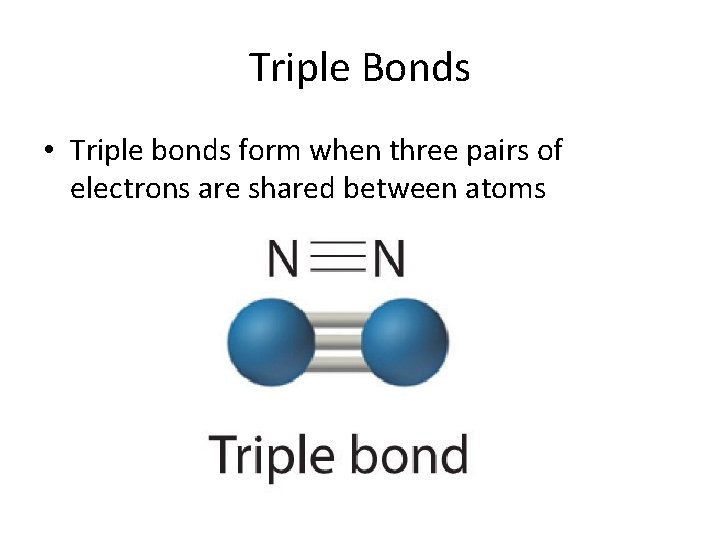
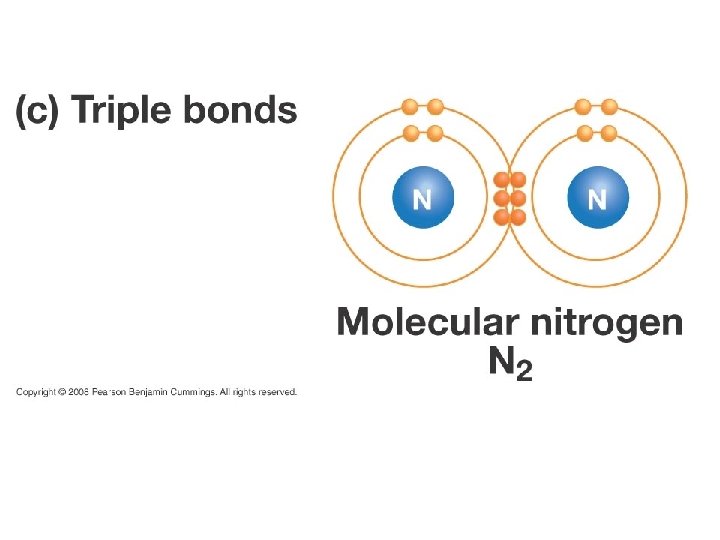
Triple Bonds • Triple bonds form when three pairs of electrons are shared between atoms
- Slides: 12