Review major reaction types Thermal processes Photochemistry Free




![N 2 ----> 2 N N ------> N+ < 100 nm + e[O] >>[O N 2 ----> 2 N N ------> N+ < 100 nm + e[O] >>[O](https://slidetodoc.com/presentation_image/b2bb56821553aaa15d9d8a036a10c4ec/image-5.jpg)














![[http: //www. cpc. ncep. noaa. gov/products/stra tosphere/tovsto/latest_ll. gif] 34 [http: //www. cpc. ncep. noaa. gov/products/stra tosphere/tovsto/latest_ll. gif] 34](https://slidetodoc.com/presentation_image/b2bb56821553aaa15d9d8a036a10c4ec/image-34.jpg)


















- Slides: 60

Review major reaction types Thermal processes Photochemistry Free radicals 1

2 N ----> N 2 ----> 2 N H = -946 k. J/mol (exothermic reaction) H = + 946 k. J/mol (endothermic reaction) Where does the energy come from to break N-N bonds? Sunlight E(photon) = hv = hc/ E = 6. 6 x 10 -34 j/s * 3. 0 x 108 m/s *6. 0 x 1023 E = (1. 19 x 105)/ k. J/mol 2

N 2 ----> 2 N H = + 946 k. J/mol N ------> N+ + e- H = + 1400 k. J/mol O 2 ------> O 2+ H = + 1160 k. J/mol Reverse Processes Produce Heat Free Radical Reactions: 3

4
![N 2 2 N N N 100 nm eO O N 2 ----> 2 N N ------> N+ < 100 nm + e[O] >>[O](https://slidetodoc.com/presentation_image/b2bb56821553aaa15d9d8a036a10c4ec/image-5.jpg)
N 2 ----> 2 N N ------> N+ < 100 nm + e[O] >>[O 2] O 2+, O+, NO+ 200 -330 nm O 2+, NO+ < 330 nm O 3 5

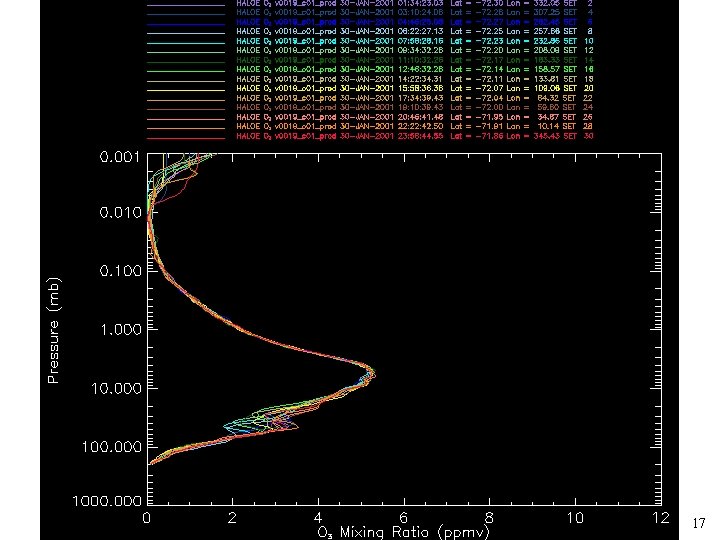
Lets talk about the ozone layer. • What is the ozone layer? – Ozone is a trace gas species in the atmosphere which reaches maximum concentrations in the stratosphere. – The maximum concentration is 10 ppmv (what is this unit) – 1 ppmv = 1 x 10 -6 p(total) – So to determine pressure we need to know ppmv O 3 and p(total) – p(total) at 30 km = 10 mbar, do the calculation. • The final result is that all the ozone in the atmosphere at STP would form a layer only 3 mm thick! 6

0 15 7

Glossary of Terms Dobson Unit The unit of measure for total ozone. If you were to take all the ozone in a column of air stretching from the surface of the earth to space, and bring all that ozone to standard temperature (0 Celsius) and pressure (1013. 25 millibars, or one atmosphere, or "atm"), the column would be about 0. 3 centimeters thick. Thus, the total ozone would be 0. 3 atmcm. To make the units easier to work with, the "Dobson Unit" is defined to be 0. 001 atmcm. Our 0. 3 atm-cm would be 300 Dobson Units (DU). 8

Thermodynamics of O 3 9

10

11

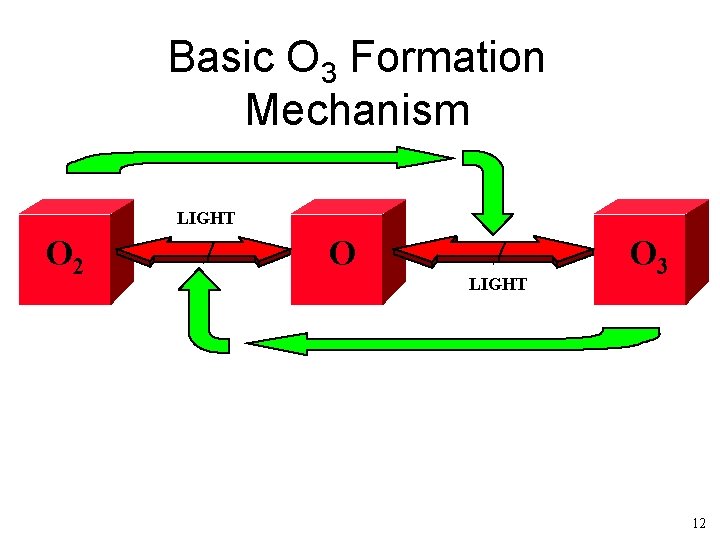
Basic O 3 Formation Mechanism LIGHT O 2 O LIGHT O 3 12

S&S 1996, pg 151. 13

14

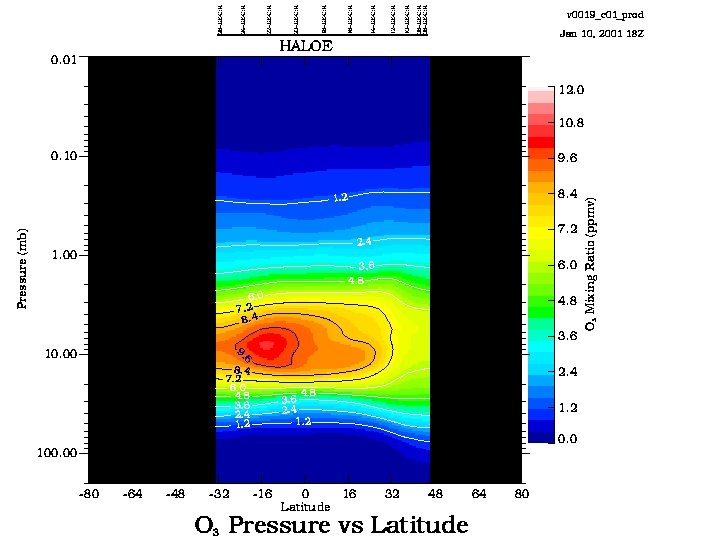
15

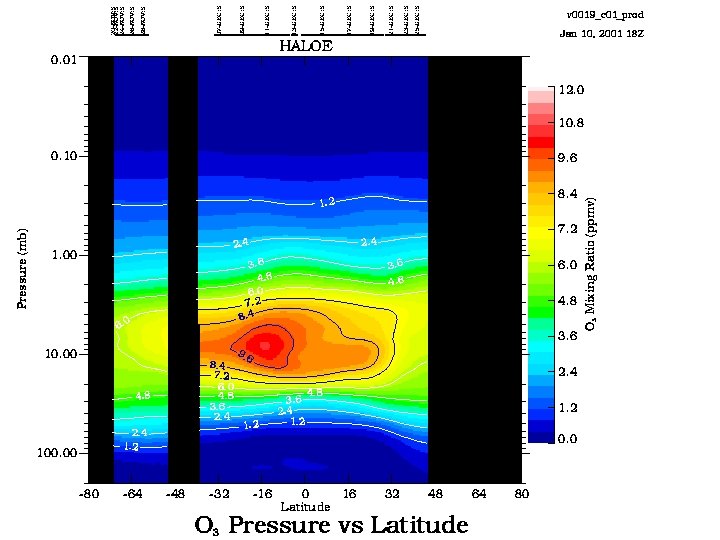
16

17

Should we worry about NOx NO WAY N 2 + O 2 ---> 2 NO H = 180 k. J mol-1 18

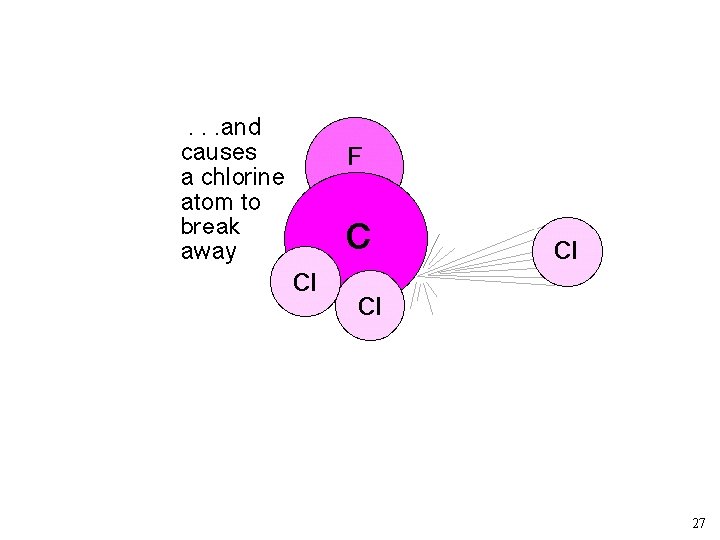
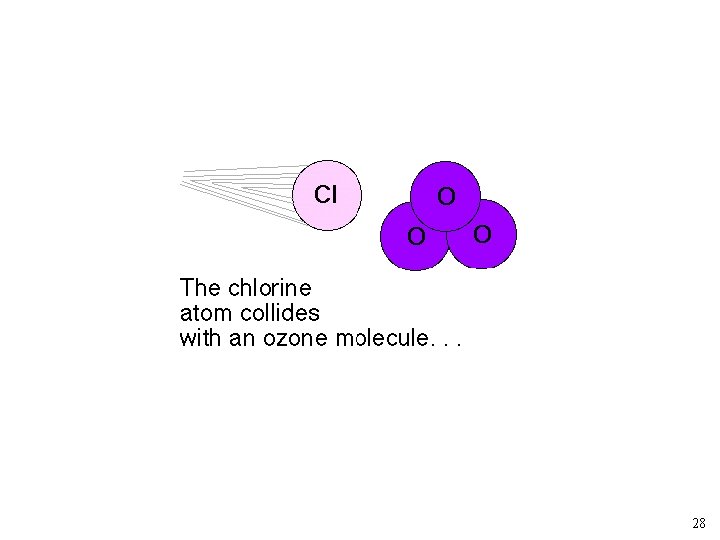
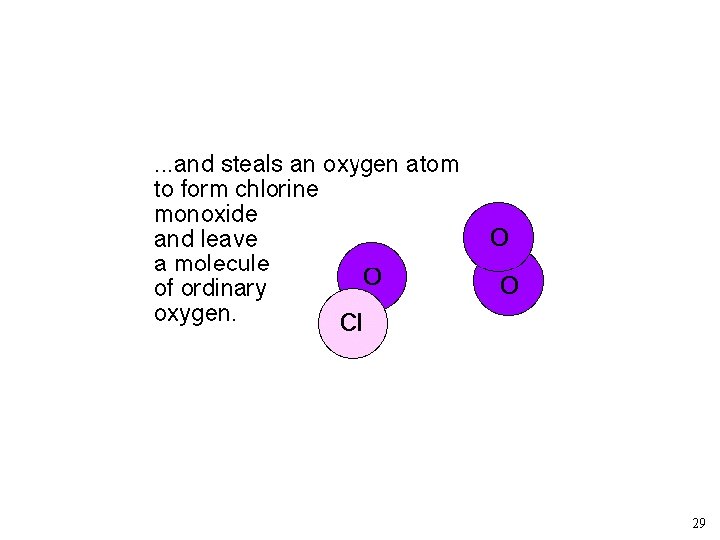
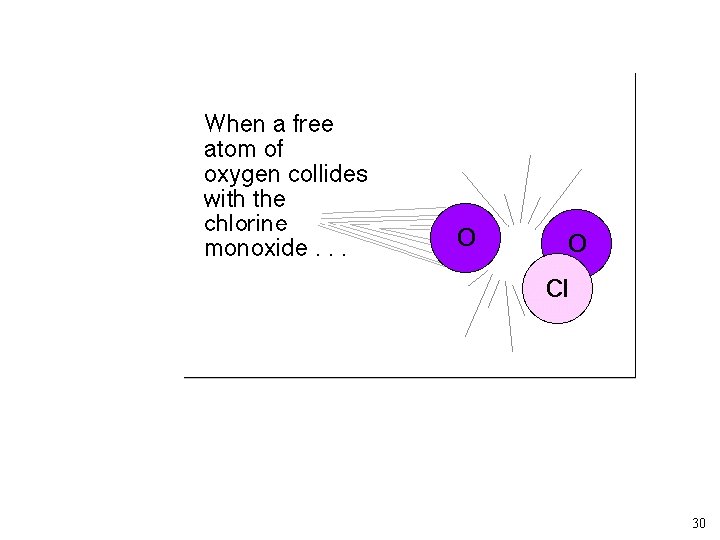
As rate constant measurements improved, the simple “Chapman” model for O 3 predicted too much O 3. Other loss mechanisms? ? NO + O 3 ---> NO 2 + O 2 NO 2 + O ---> NO + O 2 Source of NO: N 2 O + O ---> 2 NO (at 30 km) Other Reactions: X + O 3 --> XO + O 2 XO + O --> X + O 2 X: Cl, and OH 19

20

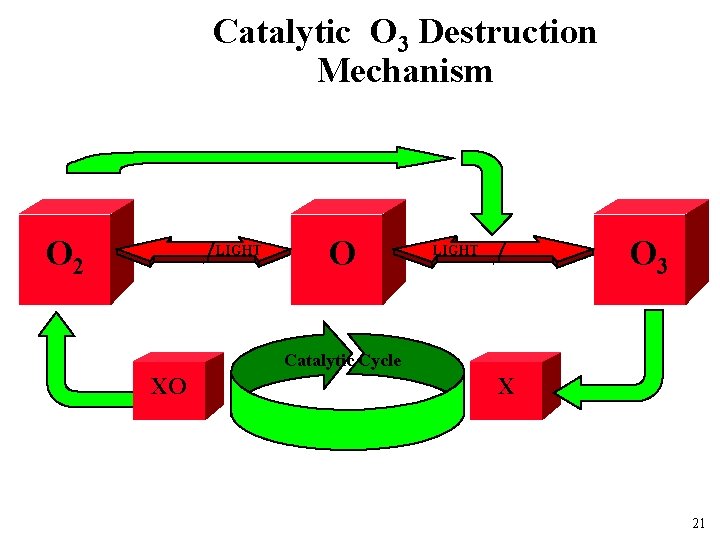
Catalytic O 3 Destruction Mechanism O 2 LIGHT O O 3 LIGHT Catalytic Cycle XO X 21

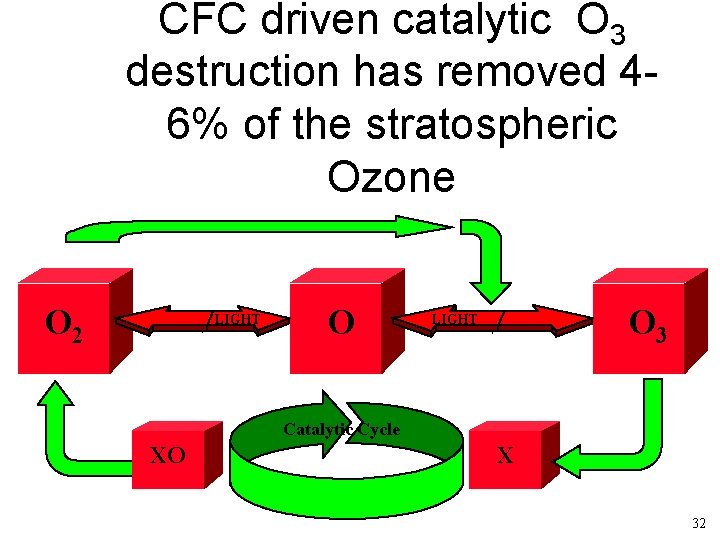
Relative rates of O 3 removal NO/NO 2 uncatalized OH/HO 2 Cl/Cl. O H/OH 70% 20% 10% 6 % (variable and increasing) insignificant 22

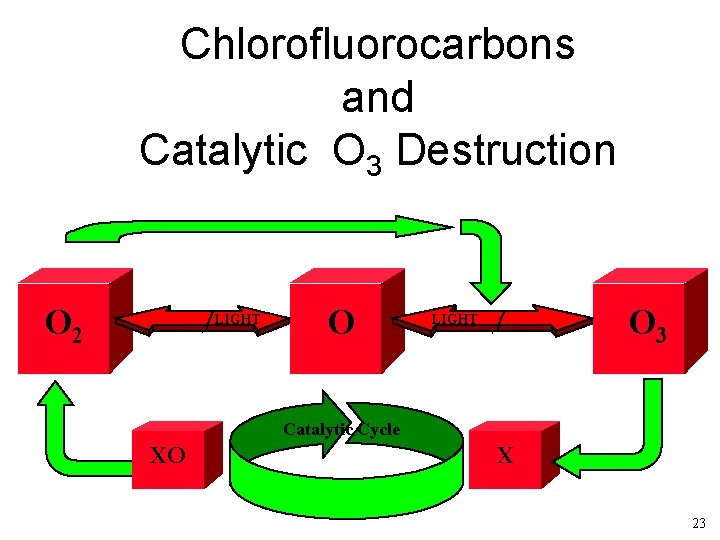
Chlorofluorocarbons and Catalytic O 3 Destruction O 2 LIGHT O O 3 LIGHT Catalytic Cycle XO X 23

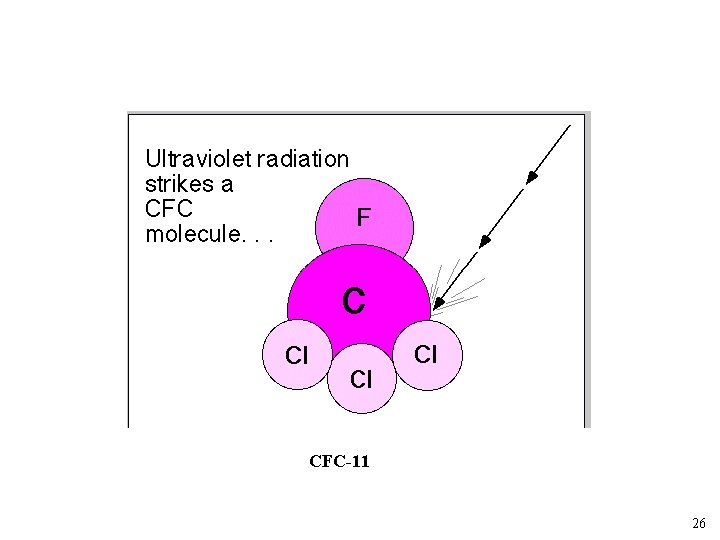
What are Chlorofluorocarbons Low molecular weight carbon based molecules. CFC-11 CFC-12 CFC-113 24

Common Halogenated Compounds Compound CFCl 3 CF 2 Cl 2 CHF 2 Cl 2 CF 2 Cl. CFCl 2 CH 3 CCl 3 CCl 4 CH 3 Cl (CFC No. ) 11 12 22 113 natural Lifetime (yr) 70 110 25 90 <10 10 Conc. (ppbv) 0. 8 1. 0 0. 2 0. 5 0. 6 25

CFC-11 26

27

28

29

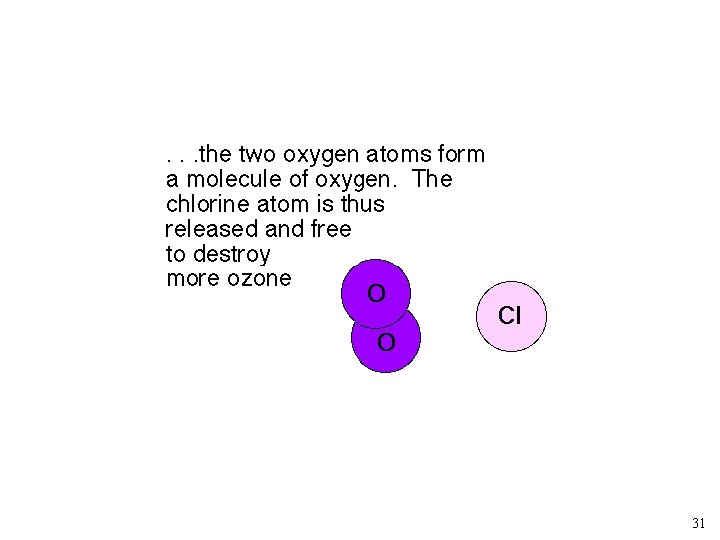
30

31

CFC driven catalytic O 3 destruction has removed 46% of the stratospheric Ozone O 2 LIGHT O O 3 LIGHT Catalytic Cycle XO X 32

HALOE RESULTS HALOE data indicates chlorine in the middle atmosphere is decreasing, demonstratingthe effectiveness of the Montreal Protocol (Anderson et al. , J. Geophys. Res. , p. 4483, 2000) HALOE observations of a slowdown in the rate of increase of HF in the lower mesosphere (Considine et al. , Geophys. Res. Letts. , p. 3217, 1997). The solar cycle effects on mesospheric water vapor (Chandra et al. , Geophys. Res. Letts. , p. 639, 1997) Reevaluation of the upper stratospheric ozone budget (Crutzen et al. , Science, p. 705, 1995; Crutzen, P. , Science, p. 1951, 1997). Presence of polar summer ozone deficit which is correlated with elevated level of NOx and. HCl (Park and Russell, J. Atmos. Sci. , p. 2903, 1994; Luo et al. , J. Geophys. Res. , p. 16145, 1997). 33
![http www cpc ncep noaa govproductsstra tospheretovstolatestll gif 34 [http: //www. cpc. ncep. noaa. gov/products/stra tosphere/tovsto/latest_ll. gif] 34](https://slidetodoc.com/presentation_image/b2bb56821553aaa15d9d8a036a10c4ec/image-34.jpg)
[http: //www. cpc. ncep. noaa. gov/products/stra tosphere/tovsto/latest_ll. gif] 34

http: //www. osdpd. noaa. gov/PSB/OZONE/TOAST/ For up-to -date data. 35

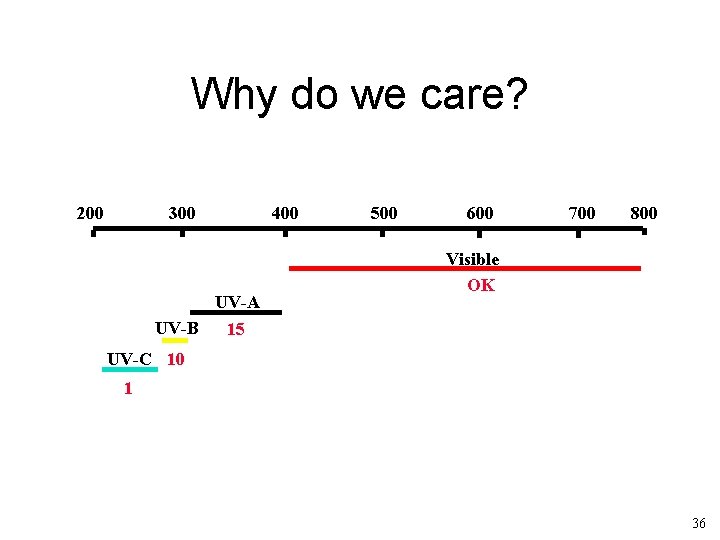
Why do we care? 200 300 UV-A UV-B 15 400 500 600 700 800 Visible OK UV-C 10 1 36

http: //www. epa. gov/sunwise/uvandhealth. html 37

S&S 1996, pg 146. T = I/Io = e- l 38

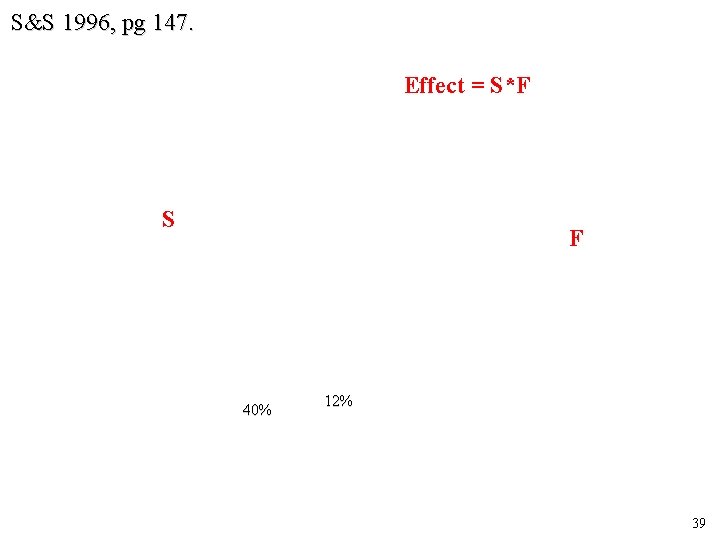
S&S 1996, pg 147. Effect = S*F S F 40% 12% 39

S&S 1996, pg 148. 40

How do we deal with the sun? Move to Maine Use Sun Screens We really care what the UV is doing to the ecosystem. 41

Common Halogenated Compounds Compound CFCl 3 CF 2 Cl 2 CHF 2 Cl 2 CF 2 Cl. CFCl 2 CH 3 CCl 3 CCl 4 CH 3 Cl (CFC No. ) 11 12 22 113 natural Lifetime (yr) Conc. (ppbv) 70 110 25 90 <10 10 0. 8 1. 0 0. 2 0. 5 0. 6 http: //www. nasa. gov/About/Education/Ozone/depletion. html 42

What are the Polar Ozone Holes 43

44

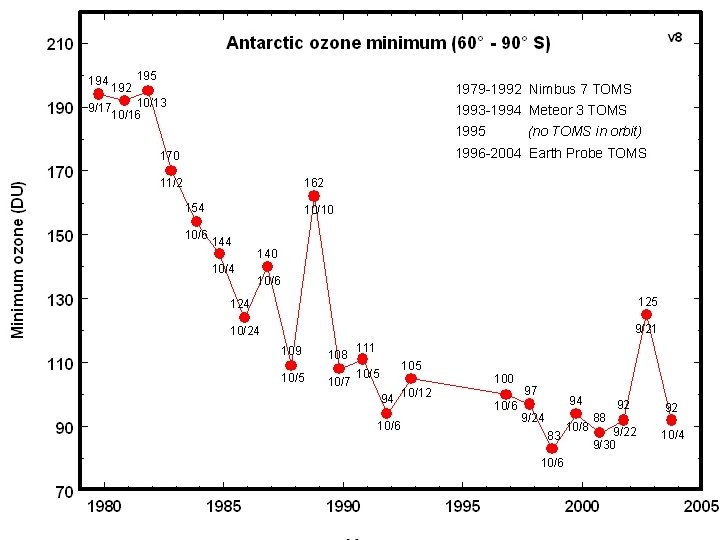
Ozone in 1998 45

http: //toms. gsfc. nasa. gov/multim. html#hole 46

47

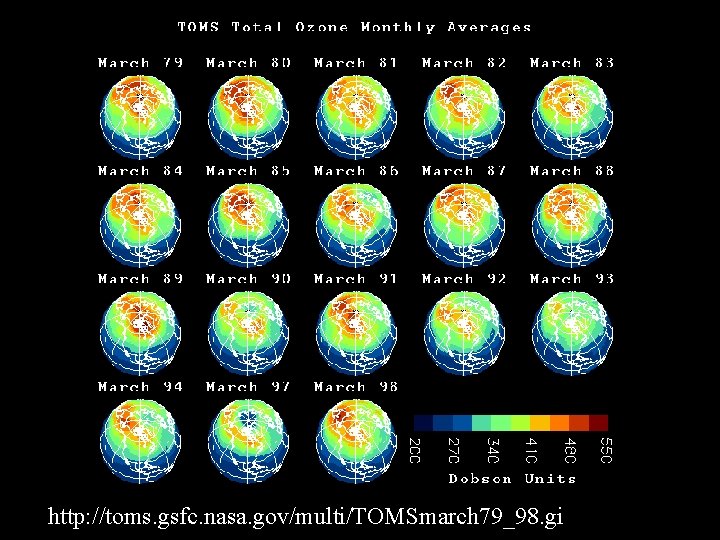
http: //toms. gsfc. nasa. gov/multi/TOMSmarch 79_98. gi 48

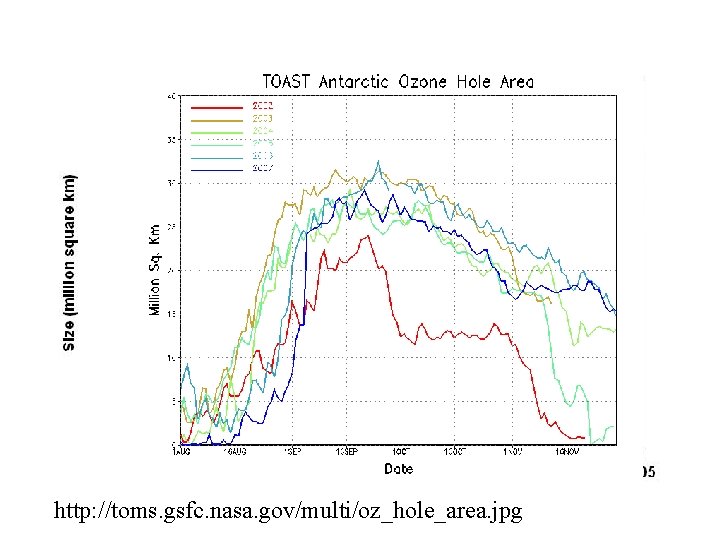
http: //toms. gsfc. nasa. gov/multi/oz_hole_area. jpg 49

50

Brian Dunbar Headquarters, Washington, DC (Phone: 202/358 -1547) December 19, 1994 Allen Kenitzer Goddard Space Flight Center, Greenbelt, MD (Phone: 301/286 -8955) RELEASE: 94 -215 NASA'S UARS CONFIRMS CFCs CAUSED ANTARCTIC OZONE HOLE Three years of data from NASA's Upper Atmosphere Research Satellite (UARS) have provided conclusive evidence that human-made chlorine in the stratosphere is the cause of the Antarctic ozone hole. UARS instruments have found chlorofluorocarbons (CFCs)--human-made products used in electronics and refrigeration systems--in the stratosphere. The satellite's global data set also has traced worldwide buildup of stratospheric fluorine gases corresponding to the breakdown of CFCs, according to NASA scientists. 51

For many years, scientists have warned that the widespread use of chlorofluorocarbons in refrigeration, spray cans and foam packaging was responsible for stratospheric ozone loss. The stratospheric ozone layer protects people, animals and plants from too much ultraviolet sunlight. The Antarctic ozone hole is a dramatic example of stratospheric ozone loss, which most scientists believe is a new phenomenon caused by the release of chlorine from human-made chlorofluorocarbons. In the past few years, some debate has occurred over the origin of ozone-destroying chlorine. Sea spray and volcanic gases have been put forth as possible sources for chlorine reaching the stratosphere. The UARS data have ended that debate. "These new results confirm our theories about CFCs, " said Dr. Mark Schoeberl, UARS Project Scientist. "The detection of stratospheric fluorine gases, which are not natural, eliminates the possibility that chlorine from volcanic eruptions or some other natural source is responsible for the ozone hole. " In addition to CFCs, UARS has detected hydrogen fluoride, a product of the chemical breakdown of CFCs, in the stratosphere. 52

"Hydrogen fluoride has no natural source, it is not produced by volcanic eruptions or salt spray, " said Dr. Anne Douglass, UARS Deputy Project Scientist. "Furthermore, scientists can calculate how much chlorine in the stratosphere is man-made using the hydrogen fluoride data. " This calculation shows that almost all of the chlorine in the stratosphere comes from human-made chlorofluorocarbons. The UARS measurements of chlorofluorocarbons were made with the Cryogenic Limb Array Etalon Spectrometer, operated by Dr. Aiden Roche of Lockheed Palo Alto Research Laboratory. The hydrogen fluoride measurements were made with the Halogen Occultation Experiment, operated by Dr. James Russell of NASA's Langley Research Center, Hampton, VA. 53

UARS measurements have confirmed that the chlorine monoxide can build up to extreme levels in the polar regions after polar stratospheric clouds appear. UARS data also have shown that the meteorology of the polar stratosphere prevents the chlorine monoxide from dispersing, thus increasing the ozone loss. 54

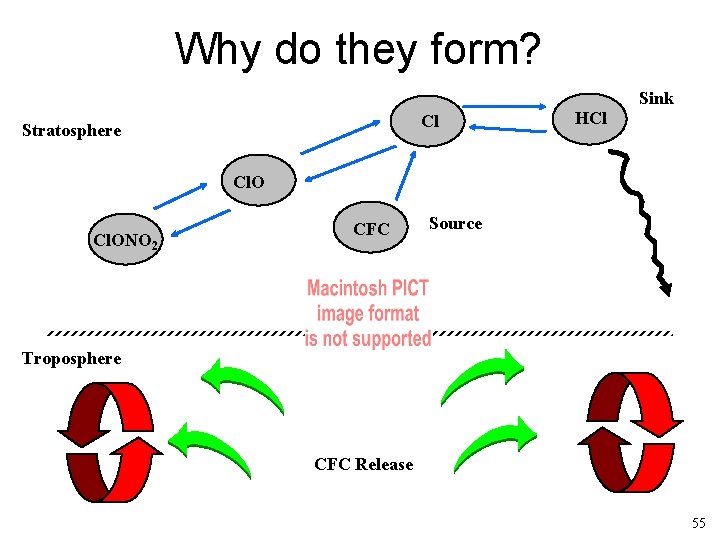
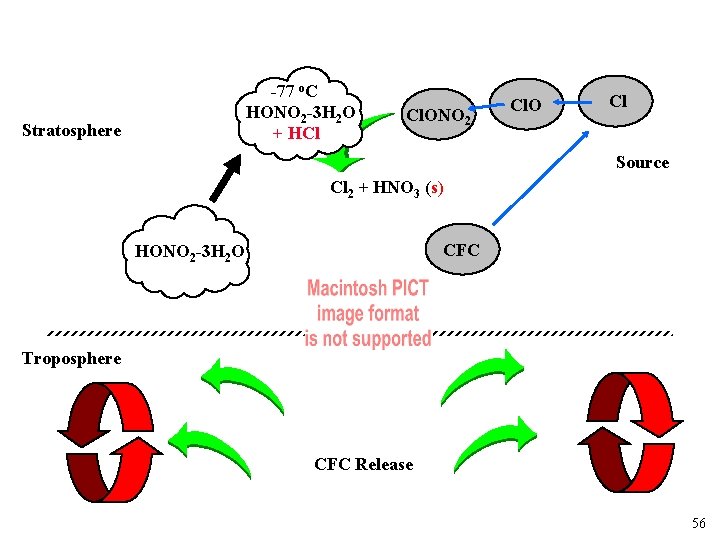
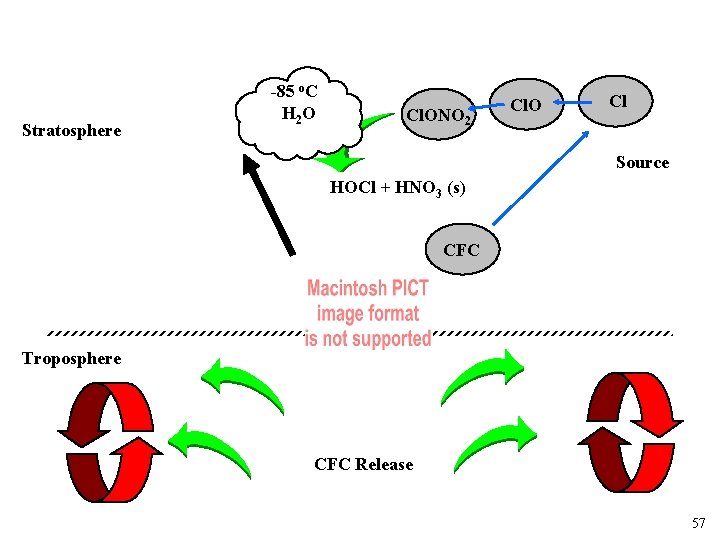
Why do they form? Sink Cl Stratosphere HCl Cl. ONO 2 CFC Source Troposphere CFC Release 55

-77 o. C HONO 2 -3 H 2 O + HCl Stratosphere Cl. ONO 2 Cl. O Cl Source Cl 2 + HNO 3 (s) CFC HONO 2 -3 H 2 O Troposphere CFC Release 56

Stratosphere -85 o. C H 2 O Cl. ONO 2 Cl. O Cl Source HOCl + HNO 3 (s) CFC Troposphere CFC Release 57

In the Spring sunlight starts the destruction on Ozone. 58

59

http: //www. atm. ch. cam. ac. uk/tour/anim_clono 2. html 60