Review Electron Configurations s orbital 1 spherical p
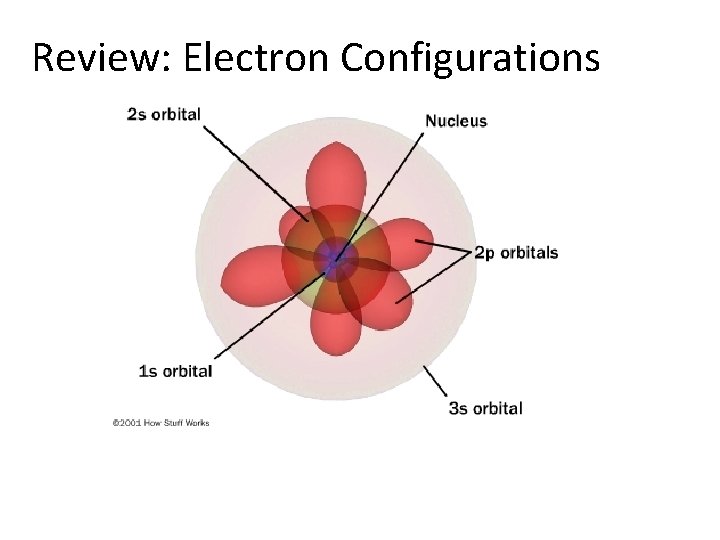
Review: Electron Configurations
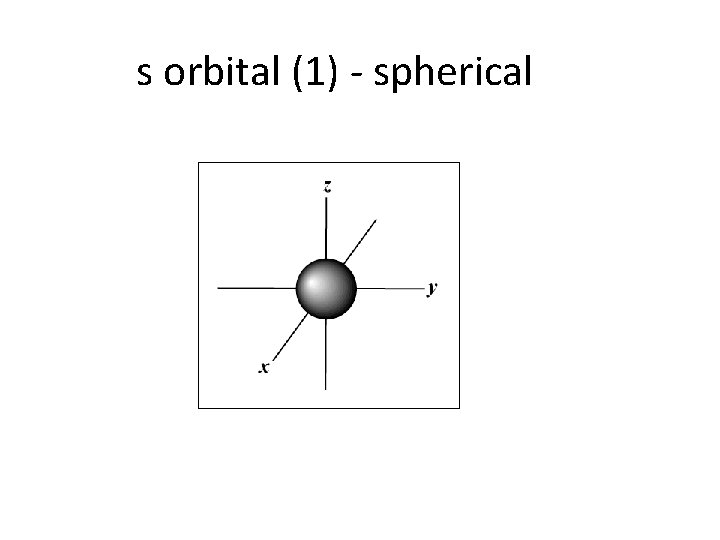
s orbital (1) - spherical
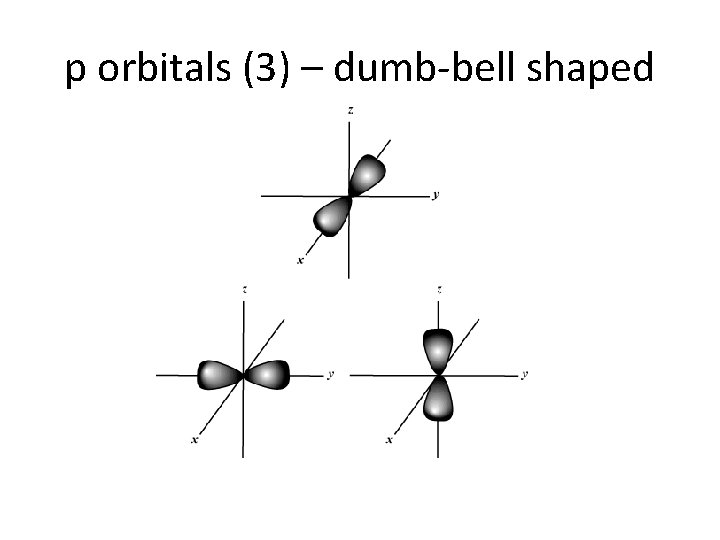
p orbitals (3) – dumb-bell shaped

d orbitals (5)
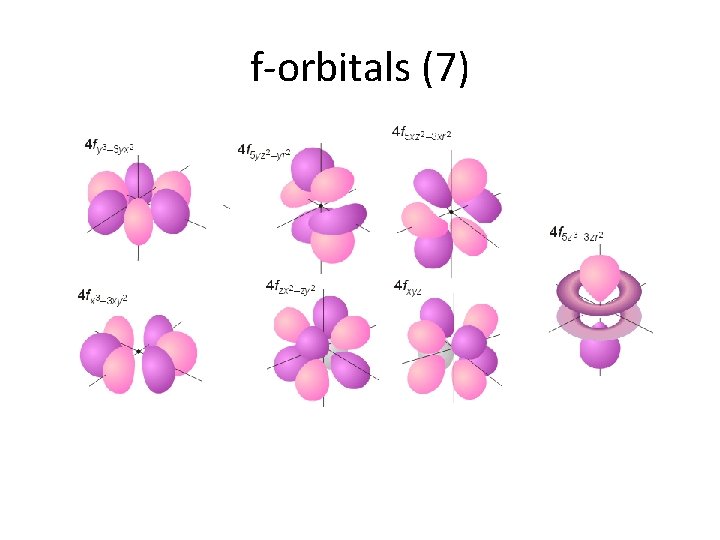
f-orbitals (7)

Review: Electron Configurations A. Principal energy levels: n = 1 to 7 (row #) B. Energy sublevels: s, p, d, f C. Orbitals: describes the electron’s location orbitals are not necessarily spherical maximum of 2 electrons per orbital s=2 p= 6 d = 10 f = 14 Example: Sodium (Na)-11 electrons 1 s 22 p 63 s 1
Noble Gas Configuration

What are Noble Gases? • Noble gases are found in group 8 A • The elements are called noble because they are non-reactive and very stable. • They do not tend to form compounds 1 A 2 A 3 4 5 6 7 8 A A A

Complete Electron Configuration What is the electron configuration for Ne? Ne: 1 s 22 p 6 What is the electron configuration for Mg? Mg: 1 s 22 p 63 s 2 What do both electron configurations have in common? • [Ne] =1 s 22 p 6 • • •

Noble Gas Configuration (Abbreviated Configuration) • Using neon’s configuration and then adding magnesium’s extra electrons we can get the noble gas configuration. • Ne: 1 s 22 p 6 • Mg: 1 s 22 p 63 s 2 • Noble gas configuration Mg: [Ne]3 s 2 • Only use noble gases in the brackets.

Which Noble Gas is Used? • To figure out which noble gas to use find the noble gas that is closest to the element without going over in atomic number • Which noble gas is closest without going over? • Rb [Kr] • Cl [Ne] • Ra [Rn]
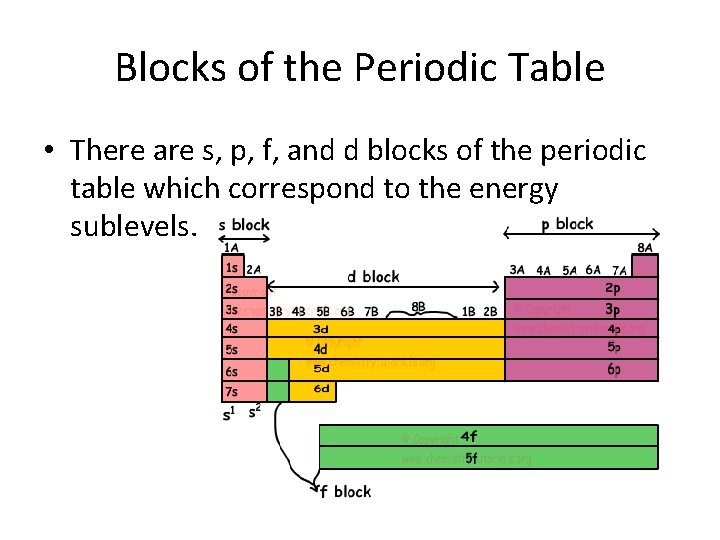
Blocks of the Periodic Table • There are s, p, f, and d blocks of the periodic table which correspond to the energy sublevels.
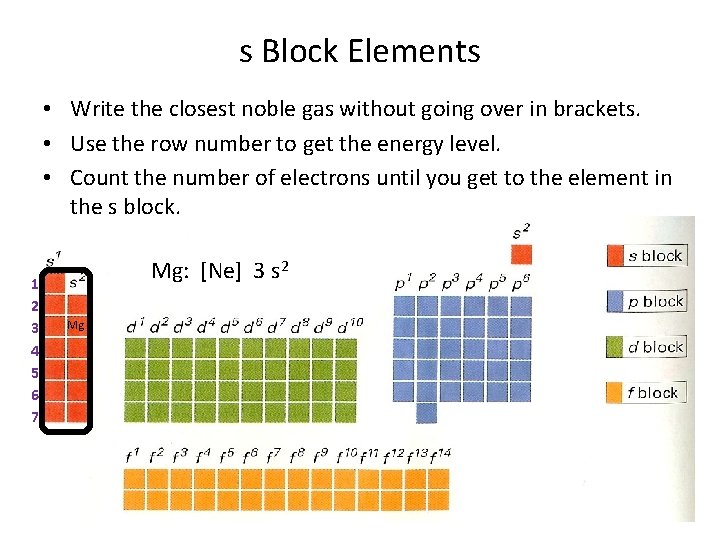
s Block Elements • Write the closest noble gas without going over in brackets. • Use the row number to get the energy level. • Count the number of electrons until you get to the element in the s block. 1 2 3 4 5 6 7 Mg: [Ne] 3 s 2 Mg

Question Time • Try other s-block elements. Write the noble gas configuration of the following elements 1 [Xe]6 s • Cs 2 [Ar]4 s • Ca
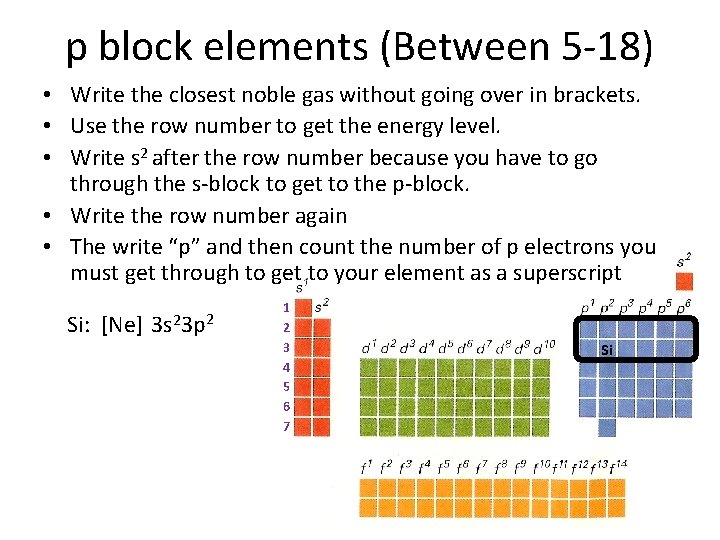
p block elements (Between 5 -18) • Write the closest noble gas without going over in brackets. • Use the row number to get the energy level. • Write s 2 after the row number because you have to go through the s-block to get to the p-block. • Write the row number again • The write “p” and then count the number of p electrons you must get through to get to your element as a superscript Si: [Ne] 3 s 23 p 2 1 2 3 4 5 6 7 Si
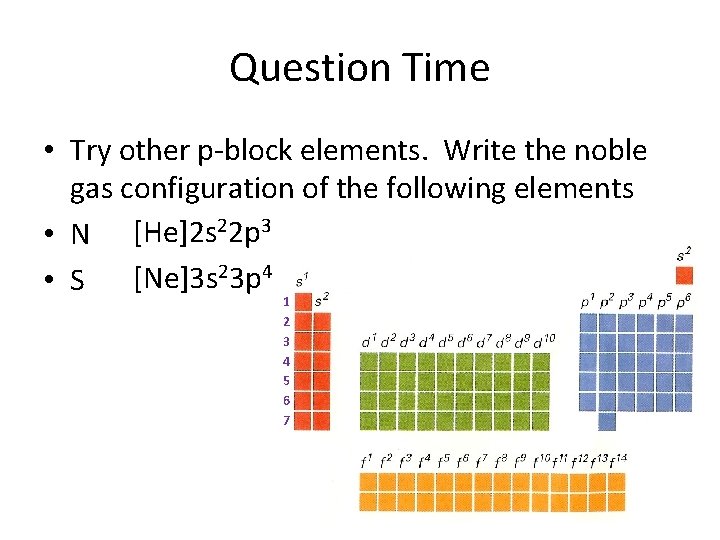
Question Time • Try other p-block elements. Write the noble gas configuration of the following elements • N [He]2 s 22 p 3 • S [Ne]3 s 23 p 4 1 2 3 4 5 6 7
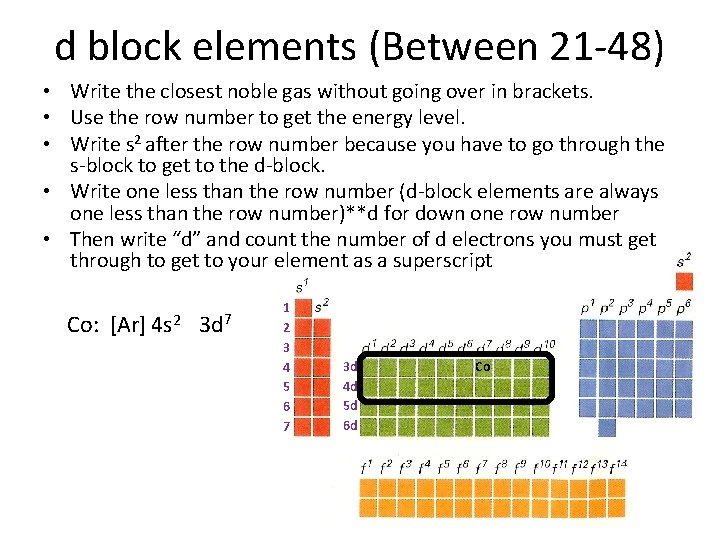
d block elements (Between 21 -48) • Write the closest noble gas without going over in brackets. • Use the row number to get the energy level. • Write s 2 after the row number because you have to go through the s-block to get to the d-block. • Write one less than the row number (d-block elements are always one less than the row number)**d for down one row number • Then write “d” and count the number of d electrons you must get through to get to your element as a superscript Co: [Ar] 4 s 2 3 d 7 1 2 3 4 5 6 7 3 d 4 d 5 d 6 d Co
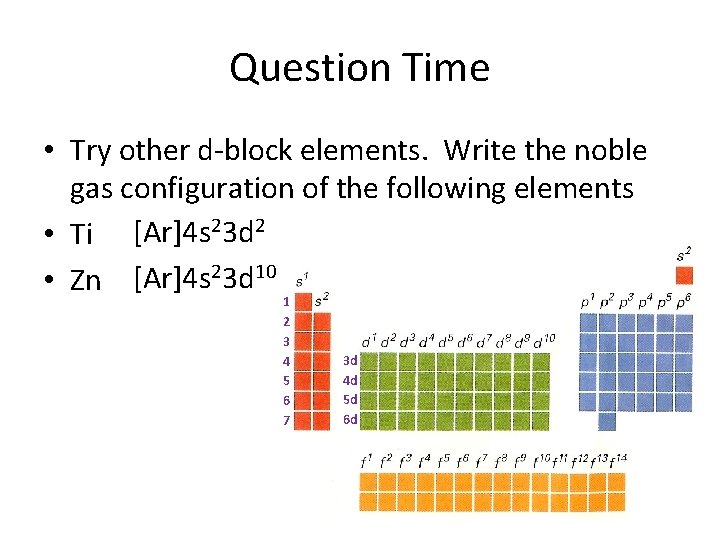
Question Time • Try other d-block elements. Write the noble gas configuration of the following elements • Ti [Ar]4 s 23 d 2 • Zn [Ar]4 s 23 d 10 1 2 3 4 5 6 7 3 d 4 d 5 d 6 d
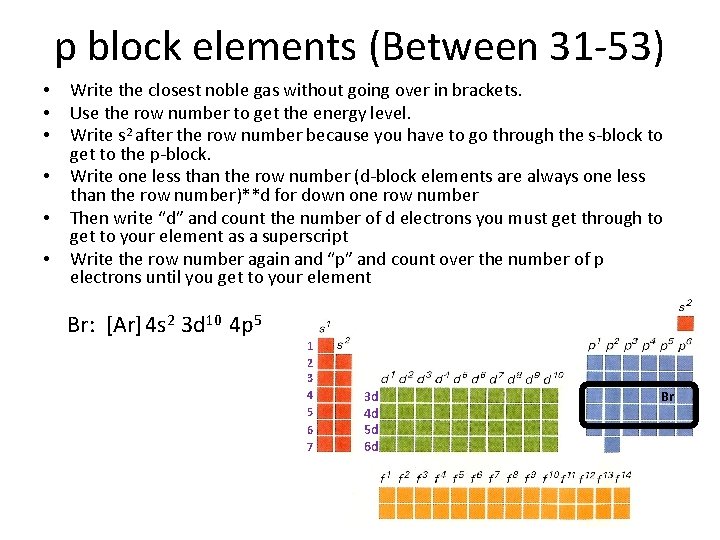
p block elements (Between 31 -53) • • • Write the closest noble gas without going over in brackets. Use the row number to get the energy level. Write s 2 after the row number because you have to go through the s-block to get to the p-block. Write one less than the row number (d-block elements are always one less than the row number)**d for down one row number Then write “d” and count the number of d electrons you must get through to get to your element as a superscript Write the row number again and “p” and count over the number of p electrons until you get to your element Br: [Ar] 4 s 2 3 d 10 4 p 5 1 2 3 4 5 6 7 3 d 4 d 5 d 6 d Br
Question Time • Try other p-block elements. Write the noble gas configuration of the following elements • Sn [Kr]5 s 24 d 105 p 2 • Se [Ar]4 s 23 d 104 p 4 1 2 3 4 5 6 7

- Slides: 21