Review 1 Instrument types of UVvisible photometers or

Review 1) Instrument types of UV-visible photometers or spectrophotometers. 2) How to measure the absorbance (A) using a single beam instrument? 3) What’s the function of beam-slitter? 歐亞書局

Chapter 14 Applications of Ultraviolet-Visible Molecular Absorption Spectrometry Absorption of UV or visible radiation: excitation of bonding electrons. Most widely used quantitative analysis techniques. Beer’s Law: A = ebc e = 8. 7 x 1019 PA P: transition probability (0 ~1) A: capture cross section (~10 -15 cm 2/molecule)
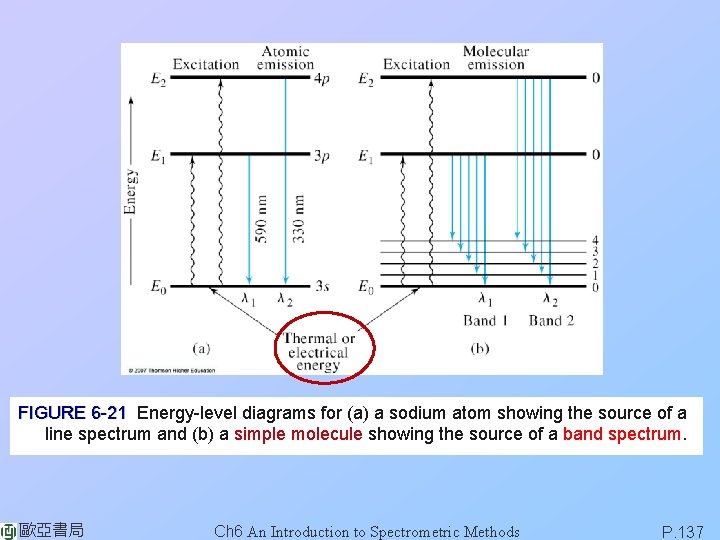
FIGURE 6 -21 Energy-level diagrams for (a) a sodium atom showing the source of a line spectrum and (b) a simple molecule showing the source of a band spectrum. 歐亞書局 Ch 6 An Introduction to Spectrometric Methods P. 137
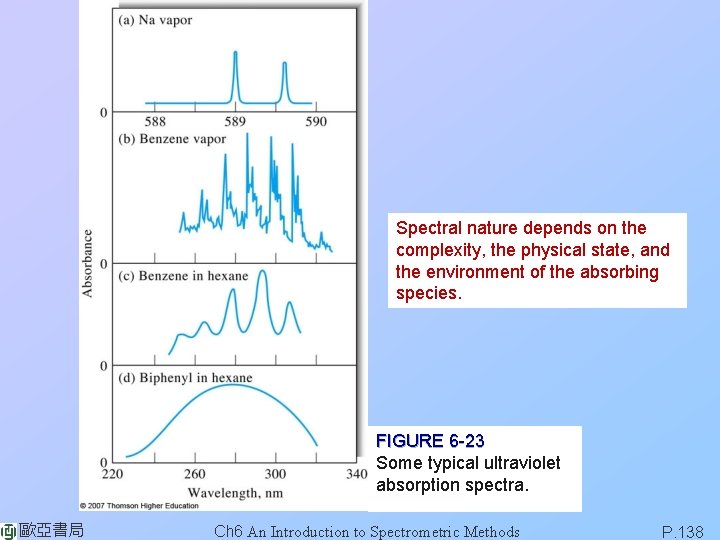
Spectral nature depends on the complexity, the physical state, and the environment of the absorbing species. FIGURE 6 -23 Some typical ultraviolet absorption spectra. 歐亞書局 Ch 6 An Introduction to Spectrometric Methods P. 138
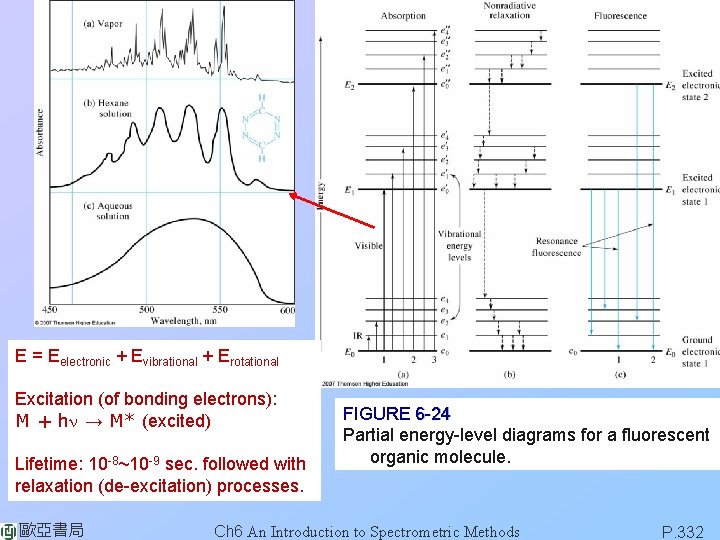
E = Eelectronic + Evibrational + Erotational Excitation (of bonding electrons): M + hn → M* (excited) Lifetime: 10 -8~10 -9 sec. followed with relaxation (de-excitation) processes. 歐亞書局 FIGURE 6 -24 Partial energy-level diagrams for a fluorescent organic molecule. Ch 6 An Introduction to Spectrometric Methods P. 332

TABLE 14 -1 Absorption Characteristics of Some common Chromophores. 歐亞書局 Ch 14 Applications of Ultraviolet-Visible Molecular Absorption Spectrometry P. 333
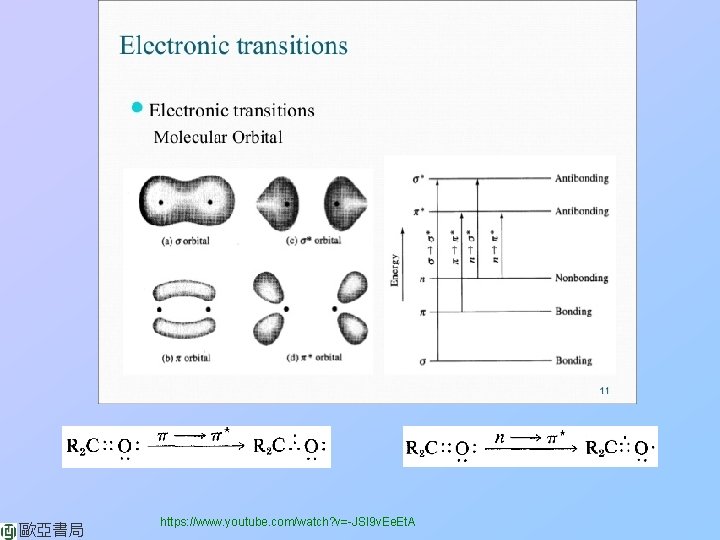
歐亞書局 https: //www. youtube. com/watch? v=-JSl 9 v. Ee. Et. A
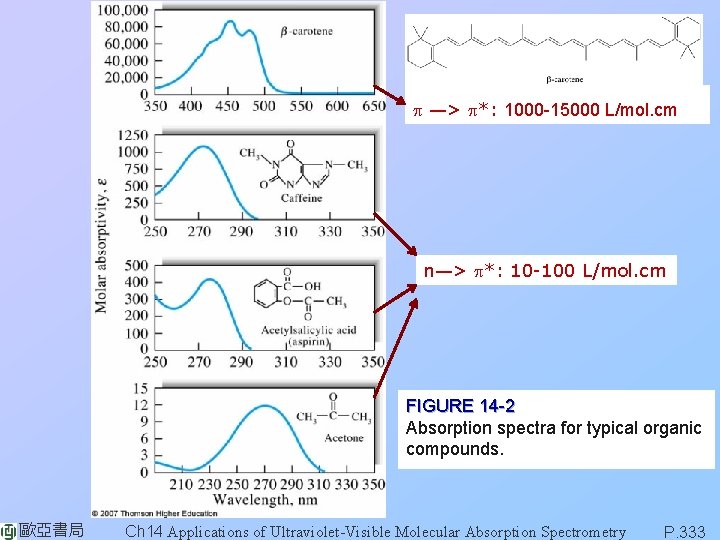
p —> p*: 1000 -15000 L/mol. cm n—> p*: 10 -100 L/mol. cm FIGURE 14 -2 Absorption spectra for typical organic compounds. 歐亞書局 Ch 14 Applications of Ultraviolet-Visible Molecular Absorption Spectrometry P. 333
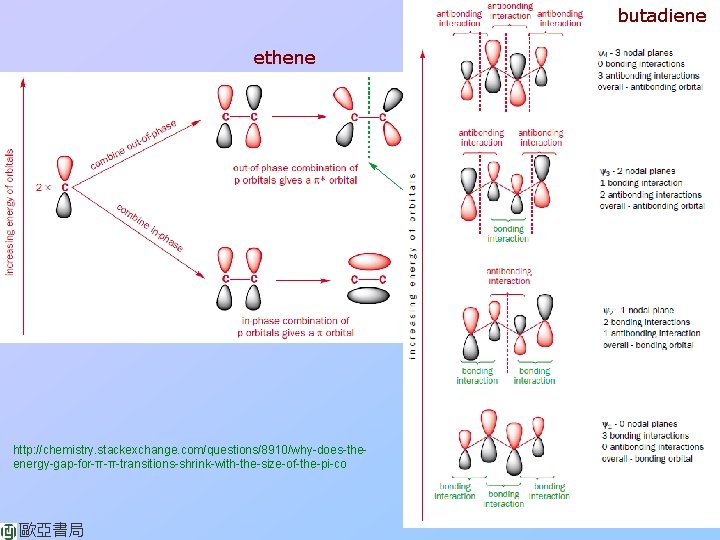
butadiene ethene http: //chemistry. stackexchange. com/questions/8910/why-does-theenergy-gap-for-π-π-transitions-shrink-with-the-size-of-the-pi-co 歐亞書局
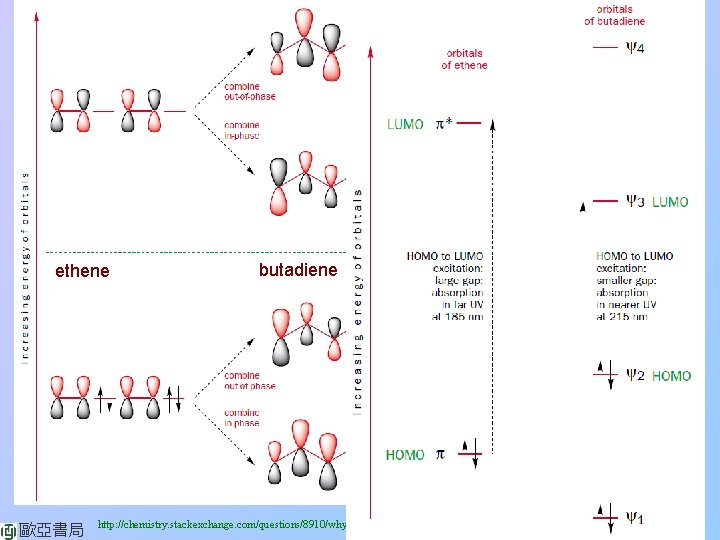
ethene 歐亞書局 butadiene http: //chemistry. stackexchange. com/questions/8910/why-does-the-energy-gap-for-π-π-transitions-shrink-with-the-size-of-the-pi-co
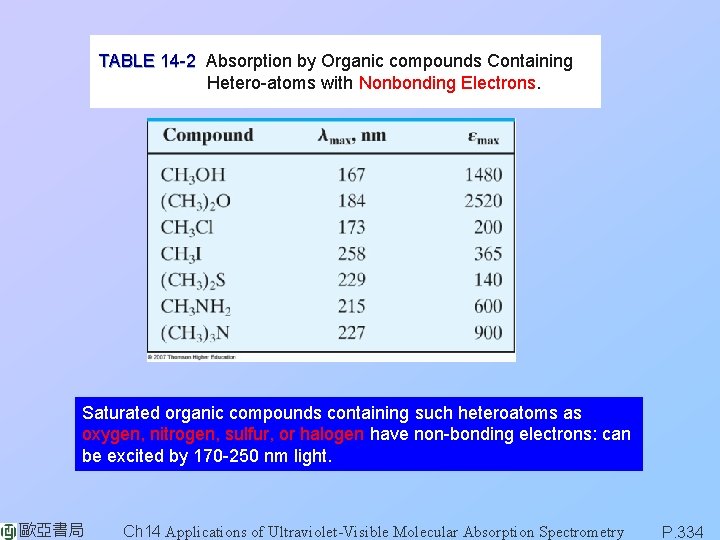
TABLE 14 -2 Absorption by Organic compounds Containing Hetero-atoms with Nonbonding Electrons. Saturated organic compounds containing such heteroatoms as oxygen, nitrogen, sulfur, or halogen have non-bonding electrons: can be excited by 170 -250 nm light. 歐亞書局 Ch 14 Applications of Ultraviolet-Visible Molecular Absorption Spectrometry P. 334
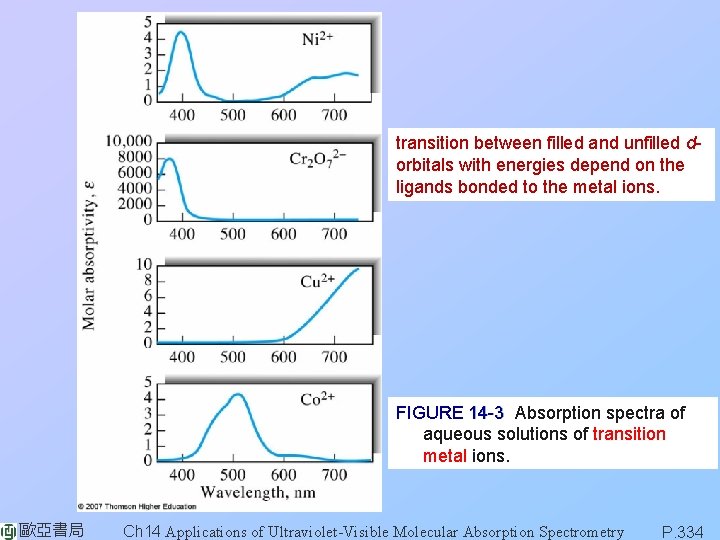
transition between filled and unfilled dorbitals with energies depend on the ligands bonded to the metal ions. FIGURE 14 -3 Absorption spectra of aqueous solutions of transition metal ions. 歐亞書局 Ch 14 Applications of Ultraviolet-Visible Molecular Absorption Spectrometry P. 334
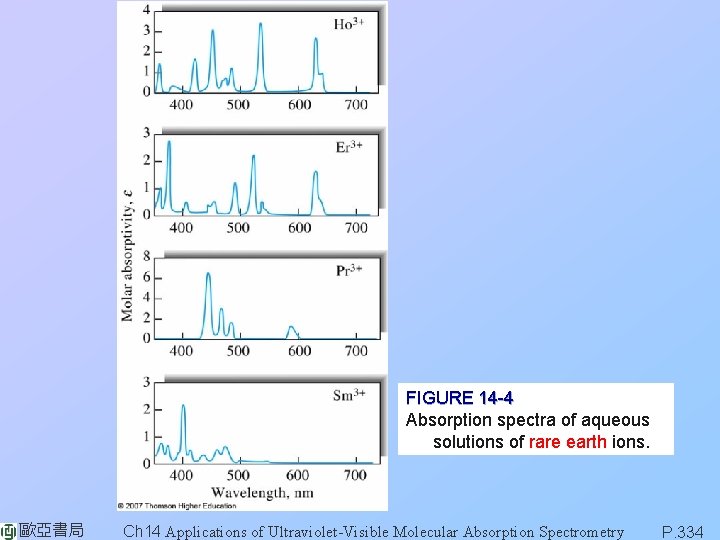
FIGURE 14 -4 Absorption spectra of aqueous solutions of rare earth ions. 歐亞書局 Ch 14 Applications of Ultraviolet-Visible Molecular Absorption Spectrometry P. 334
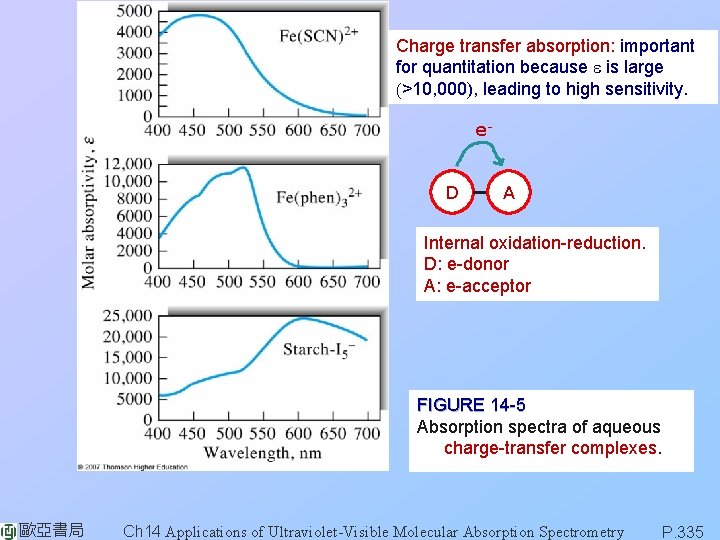
Charge transfer absorption: important for quantitation because e is large (>10, 000), leading to high sensitivity. e. D A Internal oxidation-reduction. D: e-donor A: e-acceptor FIGURE 14 -5 Absorption spectra of aqueous charge-transfer complexes. 歐亞書局 Ch 14 Applications of Ultraviolet-Visible Molecular Absorption Spectrometry P. 335
![The blood-red coloured complex pentaaqua(thiocyanato-N)iron(III), [Fe(NCS)(H 2 O)5]2+, indicates the presence of Fe 3+ The blood-red coloured complex pentaaqua(thiocyanato-N)iron(III), [Fe(NCS)(H 2 O)5]2+, indicates the presence of Fe 3+](http://slidetodoc.com/presentation_image_h/2d227731a13db1c282ec926d7ead54d5/image-15.jpg)
The blood-red coloured complex pentaaqua(thiocyanato-N)iron(III), [Fe(NCS)(H 2 O)5]2+, indicates the presence of Fe 3+ in solution 歐亞書局 Iron(II) + tris(1, 10 -phenanthroline Fe(phen)32+
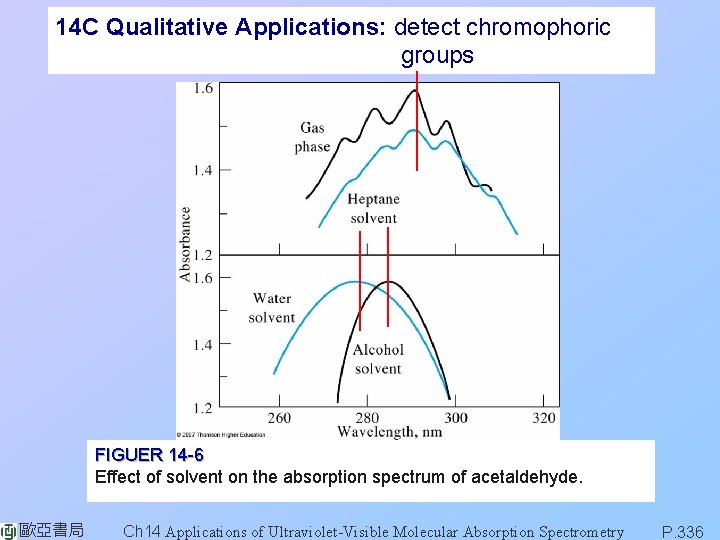
14 C Qualitative Applications: detect chromophoric groups FIGUER 14 -6 Effect of solvent on the absorption spectrum of acetaldehyde. 歐亞書局 Ch 14 Applications of Ultraviolet-Visible Molecular Absorption Spectrometry P. 336
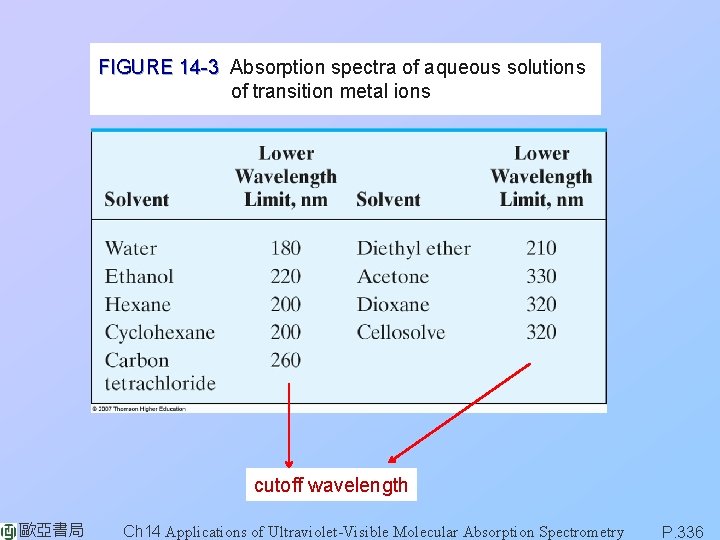
FIGURE 14 -3 Absorption spectra of aqueous solutions of transition metal ions cutoff wavelength 歐亞書局 Ch 14 Applications of Ultraviolet-Visible Molecular Absorption Spectrometry P. 336
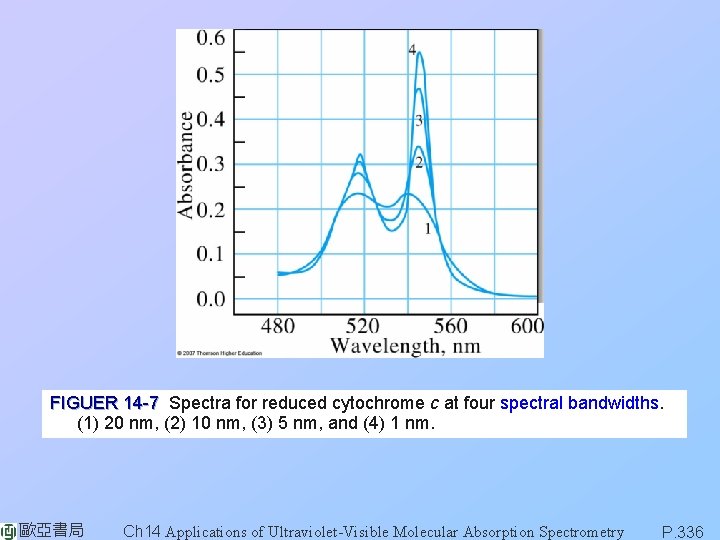
FIGUER 14 -7 Spectra for reduced cytochrome c at four spectral bandwidths. (1) 20 nm, (2) 10 nm, (3) 5 nm, and (4) 1 nm. 歐亞書局 Ch 14 Applications of Ultraviolet-Visible Molecular Absorption Spectrometry P. 336
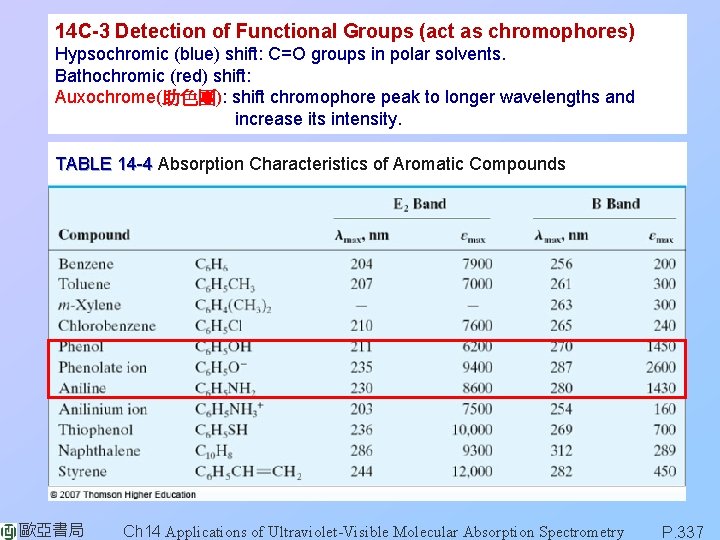
14 C-3 Detection of Functional Groups (act as chromophores) Hypsochromic (blue) shift: C=O groups in polar solvents. Bathochromic (red) shift: Auxochrome(助色團): shift chromophore peak to longer wavelengths and increase its intensity. TABLE 14 -4 Absorption Characteristics of Aromatic Compounds 歐亞書局 Ch 14 Applications of Ultraviolet-Visible Molecular Absorption Spectrometry P. 337
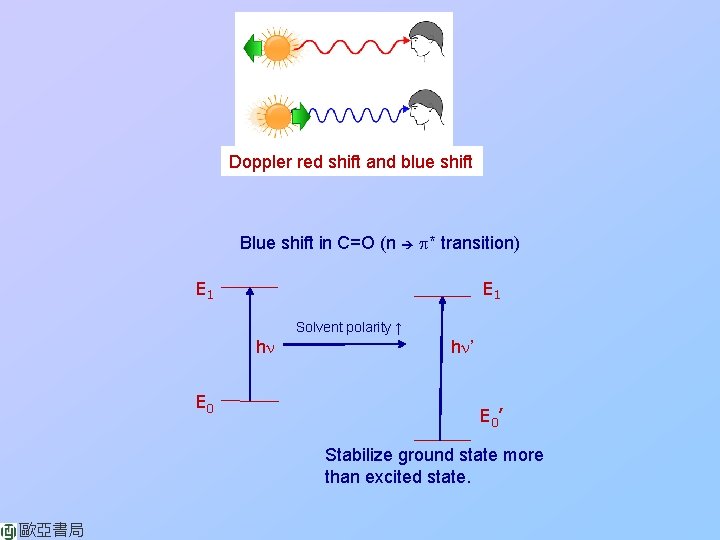
Doppler red shift and blue shift Blue shift in C=O (n p* transition) E 1 Solvent polarity ↑ hn E 0 hn’ E 0’ Stabilize ground state more than excited state. 歐亞書局
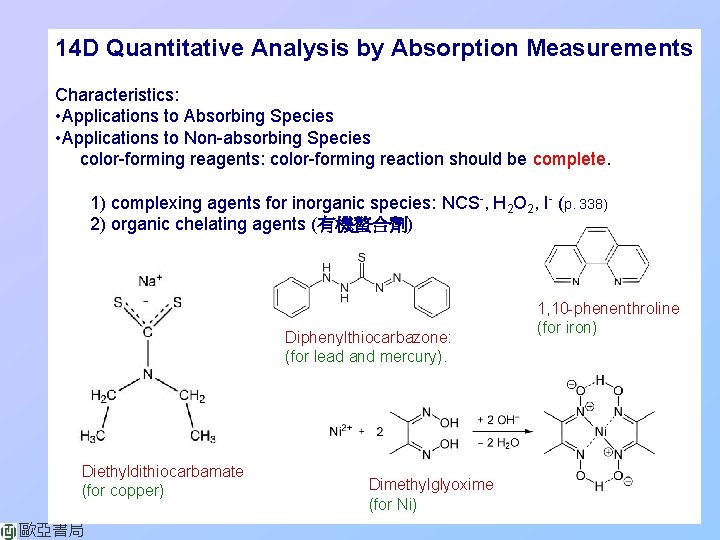
14 D Quantitative Analysis by Absorption Measurements Characteristics: • Applications to Absorbing Species • Applications to Non-absorbing Species color-forming reagents: color-forming reaction should be complete. 1) complexing agents for inorganic species: NCS-, H 2 O 2, I- (p. 338) 2) organic chelating agents (有機螯合劑) Diphenylthiocarbazone: (for lead and mercury). Diethyldithiocarbamate (for copper) 歐亞書局 Dimethylglyoxime (for Ni) 1, 10 -phenenthroline (for iron)
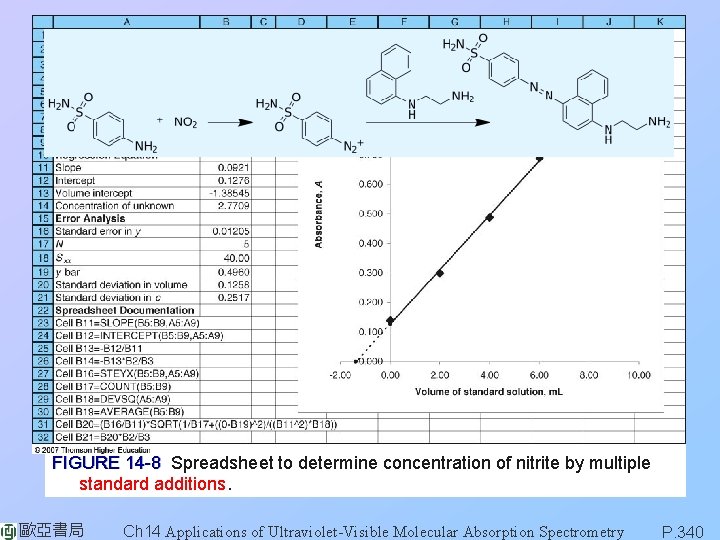
FIGURE 14 -8 Spreadsheet to determine concentration of nitrite by multiple standard additions. 歐亞書局 Ch 14 Applications of Ultraviolet-Visible Molecular Absorption Spectrometry P. 340
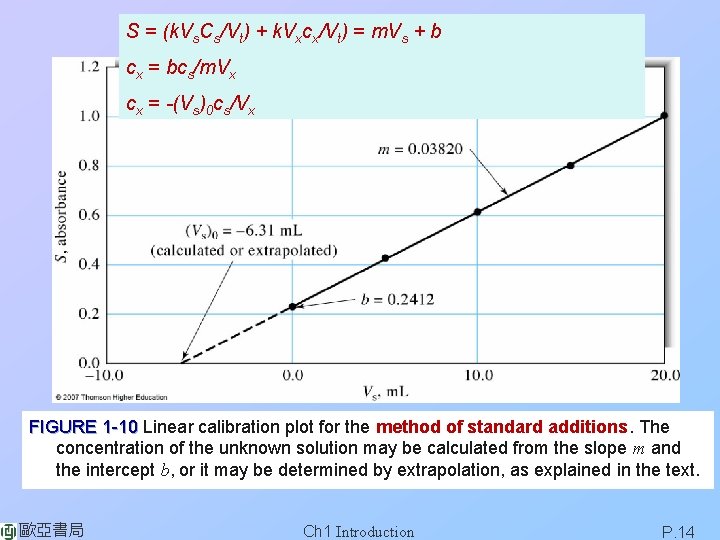
S = (k. Vs. Cs/Vt) + k. Vxcx/Vt) = m. Vs + b cx = bcs/m. Vx cx = -(Vs)0 cs/Vx FIGURE 1 -10 Linear calibration plot for the method of standard additions. The concentration of the unknown solution may be calculated from the slope m and the intercept b, or it may be determined by extrapolation, as explained in the text. 歐亞書局 Ch 1 Introduction P. 14
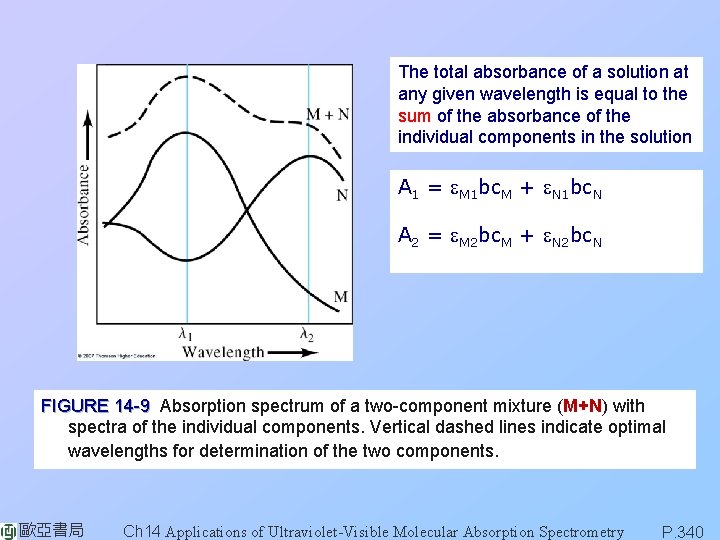
The total absorbance of a solution at any given wavelength is equal to the sum of the absorbance of the individual components in the solution A 1 = e. M 1 bc. M + e. N 1 bc. N A 2 = e. M 2 bc. M + e. N 2 bc. N FIGURE 14 -9 Absorption spectrum of a two-component mixture (M+N) with spectra of the individual components. Vertical dashed lines indicate optimal wavelengths for determination of the two components. 歐亞書局 Ch 14 Applications of Ultraviolet-Visible Molecular Absorption Spectrometry P. 340
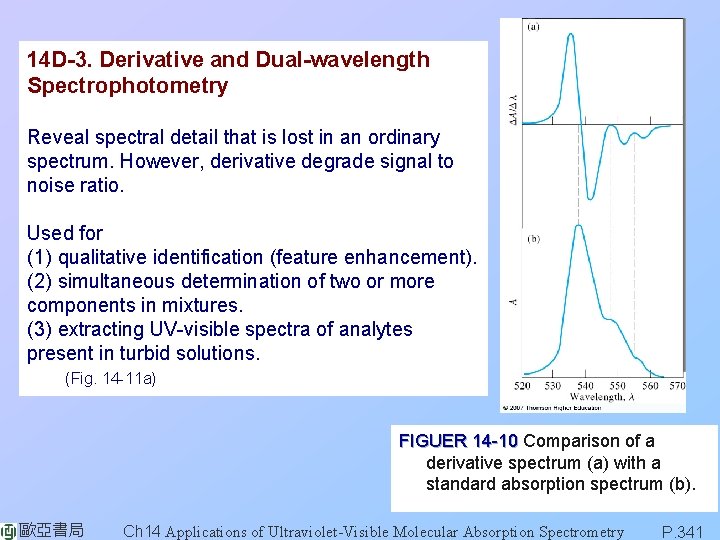
14 D-3. Derivative and Dual-wavelength Spectrophotometry Reveal spectral detail that is lost in an ordinary spectrum. However, derivative degrade signal to noise ratio. Used for (1) qualitative identification (feature enhancement). (2) simultaneous determination of two or more components in mixtures. (3) extracting UV-visible spectra of analytes present in turbid solutions. (Fig. 14 -11 a) FIGUER 14 -10 Comparison of a derivative spectrum (a) with a standard absorption spectrum (b). 歐亞書局 Ch 14 Applications of Ultraviolet-Visible Molecular Absorption Spectrometry P. 341
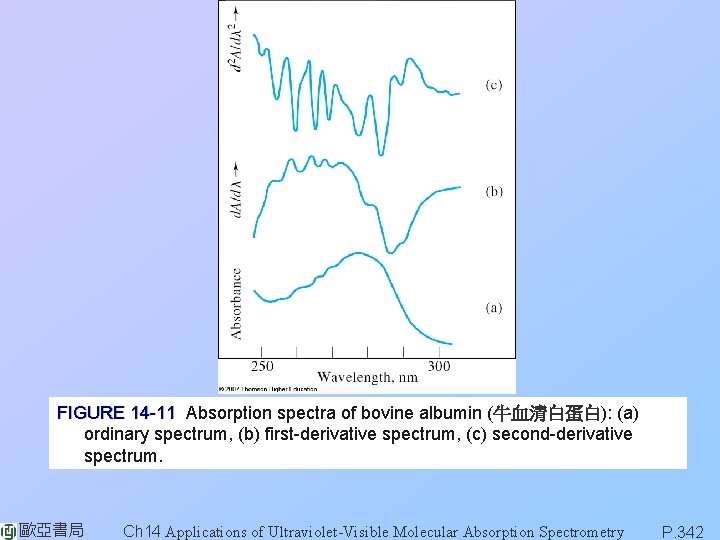
FIGURE 14 -11 Absorption spectra of bovine albumin (牛血清白蛋白): (a) ordinary spectrum, (b) first-derivative spectrum, (c) second-derivative spectrum. 歐亞書局 Ch 14 Applications of Ultraviolet-Visible Molecular Absorption Spectrometry P. 342
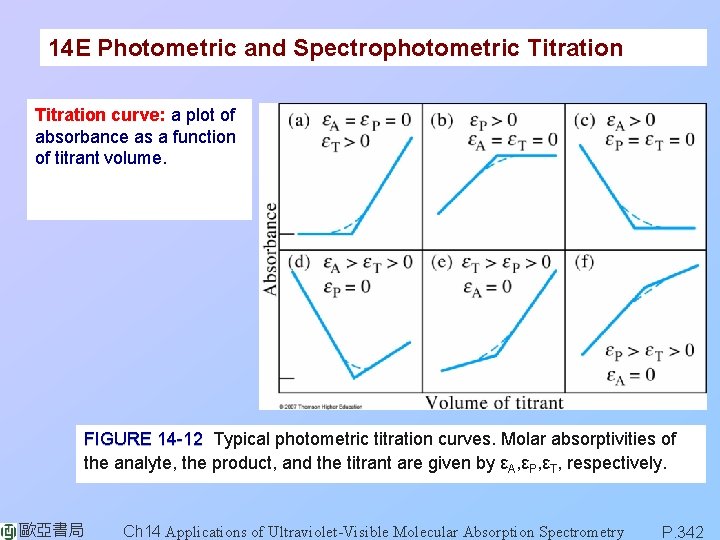
14 E Photometric and Spectrophotometric Titration curve: a plot of absorbance as a function of titrant volume. FIGURE 14 -12 Typical photometric titration curves. Molar absorptivities of the analyte, the product, and the titrant are given by εA, εP, εT, respectively. 歐亞書局 Ch 14 Applications of Ultraviolet-Visible Molecular Absorption Spectrometry P. 342
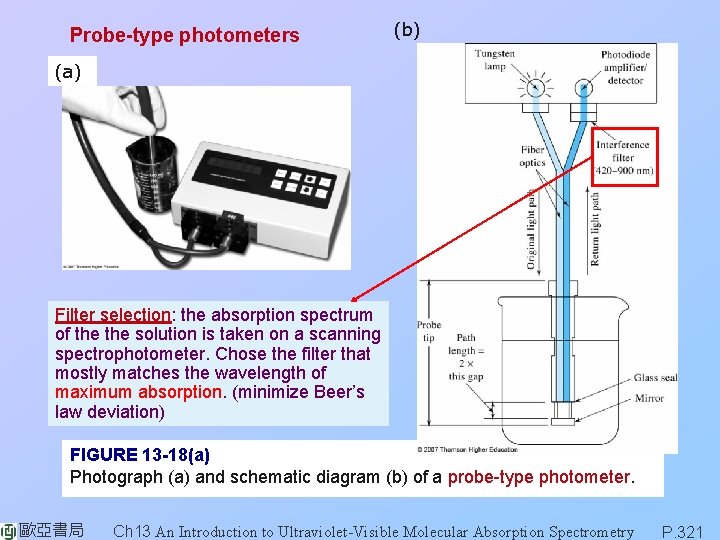
Probe-type photometers (b) (a) Filter selection: the absorption spectrum of the solution is taken on a scanning spectrophotometer. Chose the filter that mostly matches the wavelength of maximum absorption. (minimize Beer’s law deviation) FIGURE 13 -18(a) Photograph (a) and schematic diagram (b) of a probe-type photometer. 歐亞書局 Ch 13 An Introduction to Ultraviolet-Visible Molecular Absorption Spectrometry P. 321

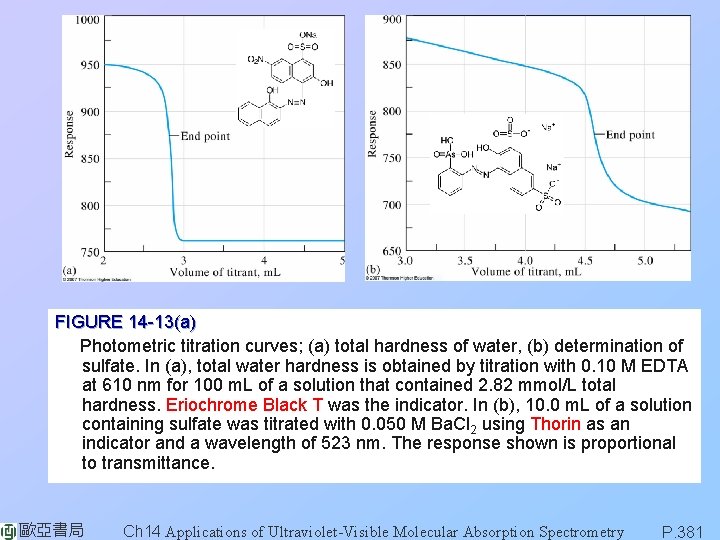
FIGURE 14 -13(a) Photometric titration curves; (a) total hardness of water, (b) determination of sulfate. In (a), total water hardness is obtained by titration with 0. 10 M EDTA at 610 nm for 100 m. L of a solution that contained 2. 82 mmol/L total hardness. Eriochrome Black T was the indicator. In (b), 10. 0 m. L of a solution containing sulfate was titrated with 0. 050 M Ba. Cl 2 using Thorin as an indicator and a wavelength of 523 nm. The response shown is proportional to transmittance. 歐亞書局 Ch 14 Applications of Ultraviolet-Visible Molecular Absorption Spectrometry P. 381
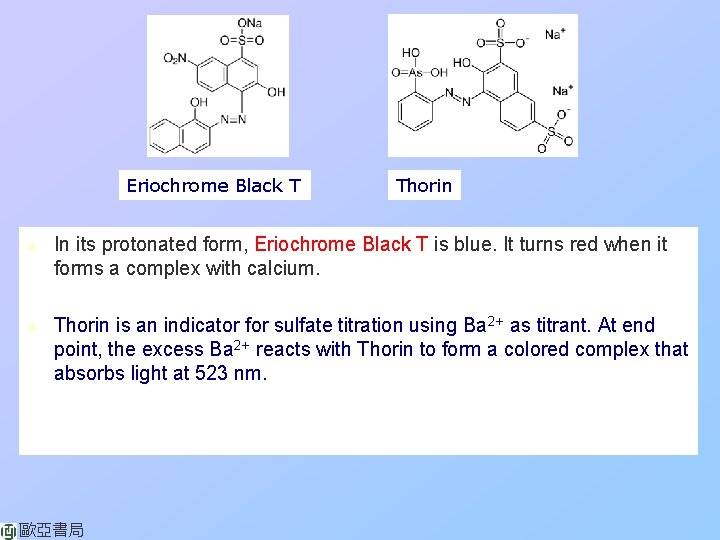
Eriochrome Black T n n Thorin In its protonated form, Eriochrome Black T is blue. It turns red when it forms a complex with calcium. Thorin is an indicator for sulfate titration using Ba 2+ as titrant. At end point, the excess Ba 2+ reacts with Thorin to form a colored complex that absorbs light at 523 nm. 歐亞書局
- Slides: 31