Reversible Reactions Equilibrium Reversible Reactions n In a

Reversible Reactions & Equilibrium
Reversible Reactions: n In a closed system, reactions are reversible! A n + B AB The conversion of reactants to products (forward reaction) & the conversion of products to reactants (reverse reaction) occur simultaneously.

Equilibrium: A + B AB n When RATE of forward reaction is the same as the RATE of reverse reaction EQUILIBRIUM n Only REVERSIBLE reactions can achieve equilibrium n Must be closed system & constant temp

Analogy: Hockey players on a team & equi: q q q Players on bench rotate with players on ice Player leave bench at same rate as players come back on bench Same players on the team (never any new or different players on the team)
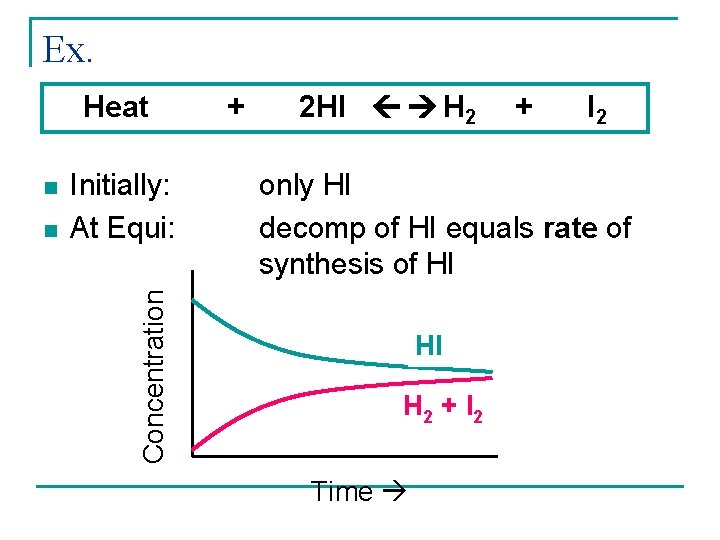
Ex. Heat n Initially: At Equi: Concentration n + 2 HI H 2 + I 2 only HI decomp of HI equals rate of synthesis of HI HI H 2 + I 2 Time
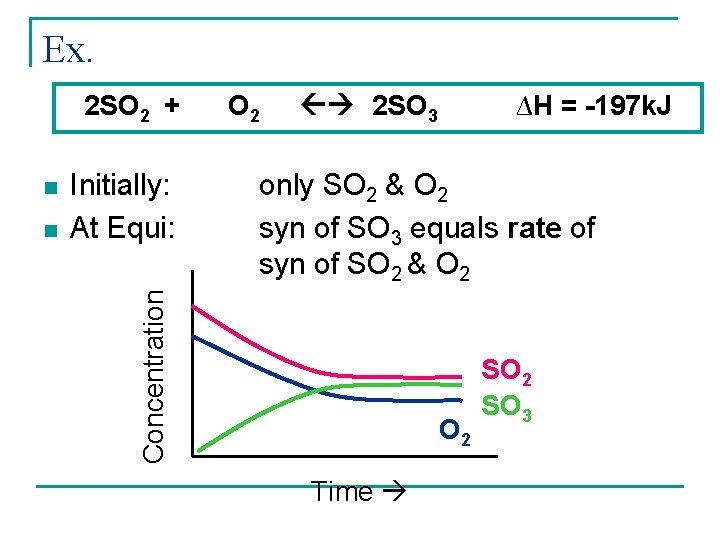
Ex. 2 SO 2 + n Initially: At Equi: 2 SO 3 ∆H = -197 k. J only SO 2 & O 2 syn of SO 3 equals rate of syn of SO 2 & O 2 Concentration n O 2 Time SO 2 SO 3

Equilibrium Constant – Keq

Keq: n expresses the concentrations of reactants & products at equilibrium n constant for every reversible reactions at equilibrium at a given temp & pressure a. A + b. B c. C + Keq = [C]c[D]d [A]a[B]b d. D
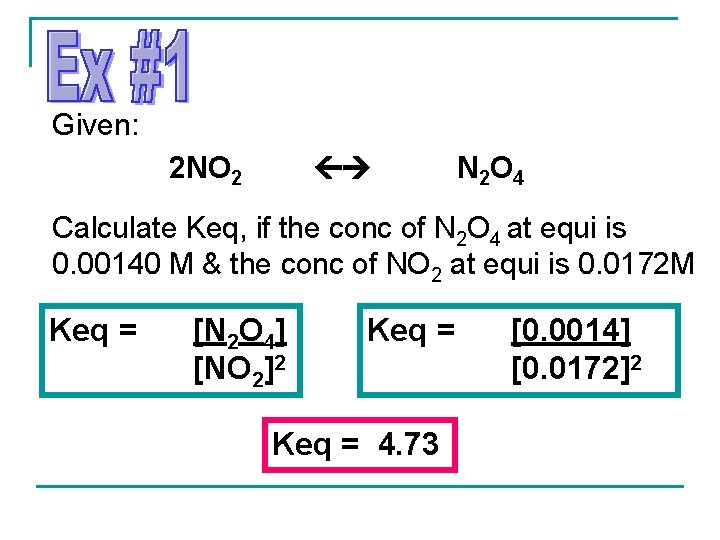
Given: 2 NO 2 N 2 O 4 Calculate Keq, if the conc of N 2 O 4 at equi is 0. 00140 M & the conc of NO 2 at equi is 0. 0172 M Keq = [N 2 O 4] [NO 2]2 Keq = 4. 73 [0. 0014] [0. 0172]2
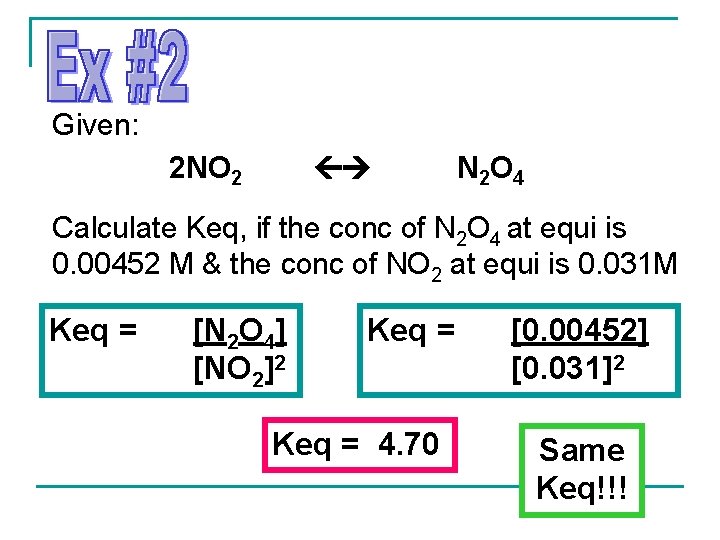
Given: 2 NO 2 N 2 O 4 Calculate Keq, if the conc of N 2 O 4 at equi is 0. 00452 M & the conc of NO 2 at equi is 0. 031 M Keq = [N 2 O 4] [NO 2]2 Keq = 4. 70 [0. 00452] [0. 031]2 Same Keq!!!

n n The value of Keq tells you exactly what is happening with a reaction If: q Keq = 1 n q Keq > 1 n q At equi, conc of products & reactants are the same At equi, greater conc of products, than reactants Keq < 1 n At equi, greater conc of reactants, than products

q q Keq is the same for a given reaction at equilibrium at the same temp no matter what the initial conc were. Keq does change with temp Do not include pure solids and pure liquids in the Keq expression because their concentrations vary little. Everything else must be included

- Slides: 13