Retrospective Chart Review of Treatment Patterns Healthcare Resource




- Slides: 10

Retrospective Chart Review of Treatment Patterns, Healthcare Resource Utilization in Patients With Severe Aplastic Anemia and Poor Response to Immunosuppression Integrating New Hematology Findings Into Practice: Independent Conference Coverage of ASH 2017, * December 9 -12, Atlanta, Georgia *CCO is an independent medical education company that provides state-of-the-art medical information to healthcare professionals through conference coverage and other educational programs. This activity is supported by educational grants from Abb. Vie; Astra. Zeneca; Celgene Corporation; Genentech; Janssen Biotech, Inc administered by Janssen Scientific Affairs, LLC; Jazz Pharmaceuticals; Novartis Pharmaceuticals Corporation; Pharmacyclics Inc; Seattle Genetics; and Takeda Oncology.

Treatment Patterns in Aplastic Anemia: Background § Aplastic anemia extremely rare, characterized by pancytopenia and hypocellular BM[1] § When matched related donors unavailable for SCT, frontline immunosuppressive therapy (eg, ATG ± cyclosporine/tacrolimus) used[2] – Pts responding poorly to initial immunosuppression can repeat treatment, add or switch to a TSA (eltrombopag, romiplostim) or androgens, or reconsider allo. HSCT § Health economic burden poorly understood, particularly in refractory pts § Current retrospective medical records review assessed real-world healthcare utilization in pts with severe aplastic anemia and insufficient response to immunosuppressive treatment[3] 1. Brodsky RA, Jones RJ. Lancet. 2005; 365: 1647 -1656. 2. Peslak SA, et al. Curr Treat Options Oncol. 2017; 18: 70. 3. Peffault De Latour R, et al. ASH 2017. Abstract 678. Slide credit: clinicaloptions. com

Treatment Patterns in Aplastic Anemia: Study Design § Retrospective noninterventional chart review at Hôpital Saint-Louis in Paris, France, and Dana-Farber Cancer Institute in Boston, Massachusetts (N = 40) § Inclusion criteria – Adult pts diagnosed with severe aplastic anemia between January 2006 and July 2016 having ≥ 12 mos of subsequent medical data available – BM cellularity < 25% or 25% to 50% with < 30% residual hematopoietic cells and ≥ 2 of the following: neutrophils < 500 cells/µL, PLTs < 20, 000 cells/µL, reticulocytes < 20, 000 cells/µL – Insufficient response to immunosuppressive therapy with ATG – ≥ 100 days of continuous medical data following HSCT or treatment with eltrombopag § Exclusion criteria: Fanconi anemia or SCT from umbilical cord blood § Study endpoints: treatment patterns, frequency of transfusions, healthcare resource utilization Peffault De Latour R, et al. ASH 2017. Abstract 678. Slide credit: clinicaloptions. com

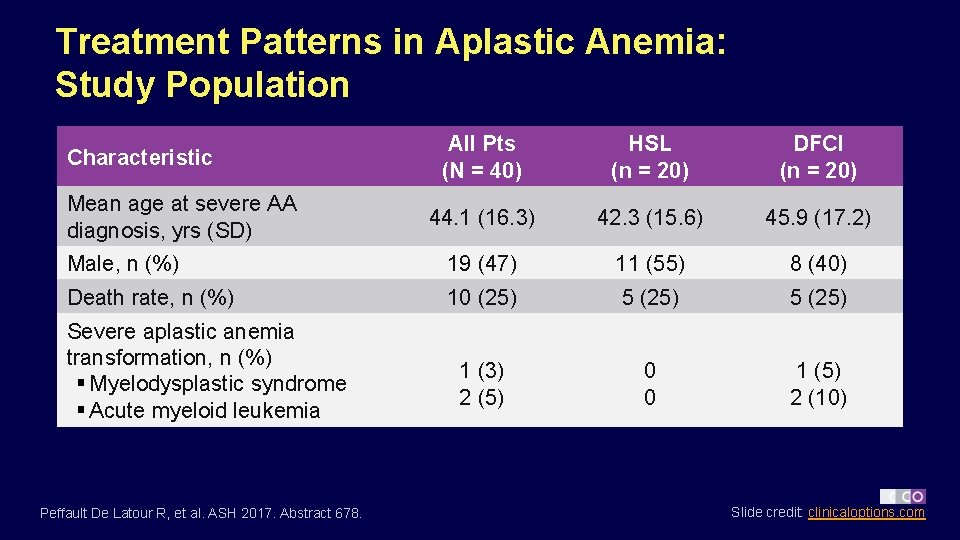
Treatment Patterns in Aplastic Anemia: Study Population All Pts (N = 40) HSL (n = 20) DFCI (n = 20) 44. 1 (16. 3) 42. 3 (15. 6) 45. 9 (17. 2) Male, n (%) 19 (47) 11 (55) 8 (40) Death rate, n (%) 10 (25) 5 (25) 1 (3) 2 (5) 0 0 1 (5) 2 (10) Characteristic Mean age at severe AA diagnosis, yrs (SD) Severe aplastic anemia transformation, n (%) § Myelodysplastic syndrome § Acute myeloid leukemia Peffault De Latour R, et al. ASH 2017. Abstract 678. Slide credit: clinicaloptions. com

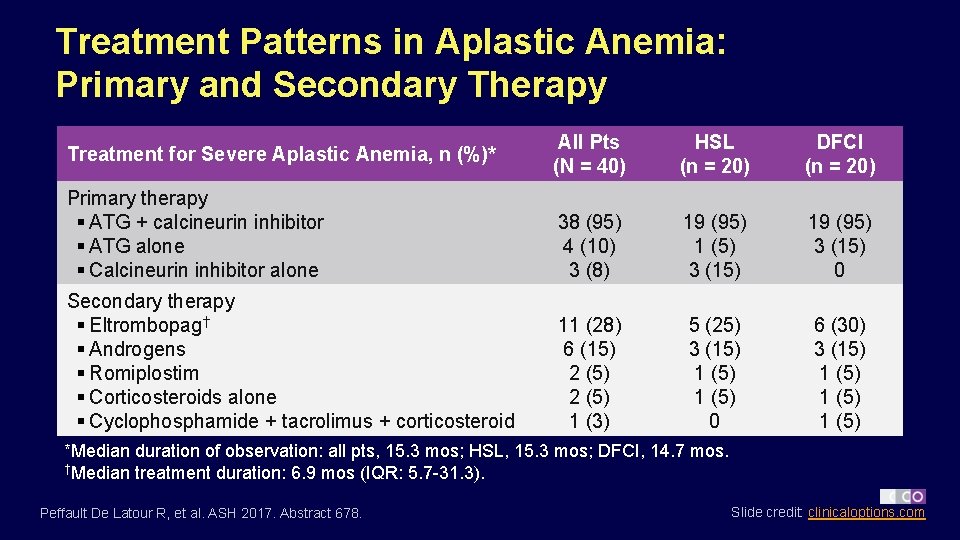
Treatment Patterns in Aplastic Anemia: Primary and Secondary Therapy All Pts (N = 40) HSL (n = 20) DFCI (n = 20) Primary therapy § ATG + calcineurin inhibitor § ATG alone § Calcineurin inhibitor alone 38 (95) 4 (10) 3 (8) 19 (95) 1 (5) 3 (15) 19 (95) 3 (15) 0 Secondary therapy § Eltrombopag† § Androgens § Romiplostim § Corticosteroids alone § Cyclophosphamide + tacrolimus + corticosteroid 11 (28) 6 (15) 2 (5) 1 (3) 5 (25) 3 (15) 1 (5) 0 6 (30) 3 (15) 1 (5) Treatment for Severe Aplastic Anemia, n (%)* *Median duration of observation: all pts, 15. 3 mos; HSL, 15. 3 mos; DFCI, 14. 7 mos. †Median treatment duration: 6. 9 mos (IQR: 5. 7 -31. 3). Peffault De Latour R, et al. ASH 2017. Abstract 678. Slide credit: clinicaloptions. com

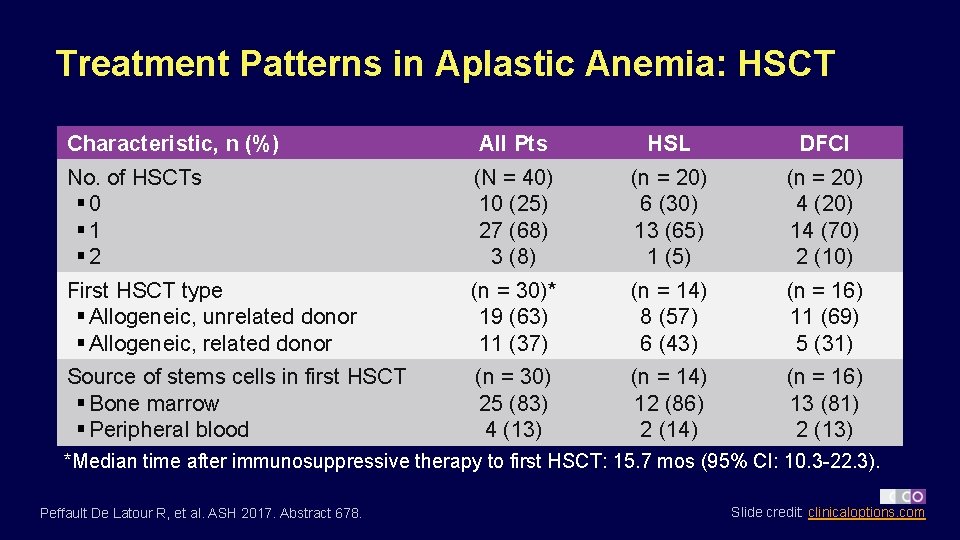
Treatment Patterns in Aplastic Anemia: HSCT Characteristic, n (%) All Pts HSL DFCI No. of HSCTs § 0 § 1 § 2 (N = 40) 10 (25) 27 (68) 3 (8) (n = 20) 6 (30) 13 (65) 1 (5) (n = 20) 4 (20) 14 (70) 2 (10) First HSCT type § Allogeneic, unrelated donor § Allogeneic, related donor (n = 30)* 19 (63) 11 (37) (n = 14) 8 (57) 6 (43) (n = 16) 11 (69) 5 (31) Source of stems cells in first HSCT § Bone marrow § Peripheral blood (n = 30) 25 (83) 4 (13) (n = 14) 12 (86) 2 (14) (n = 16) 13 (81) 2 (13) *Median time after immunosuppressive therapy to first HSCT: 15. 7 mos (95% CI: 10. 3 -22. 3). Peffault De Latour R, et al. ASH 2017. Abstract 678. Slide credit: clinicaloptions. com

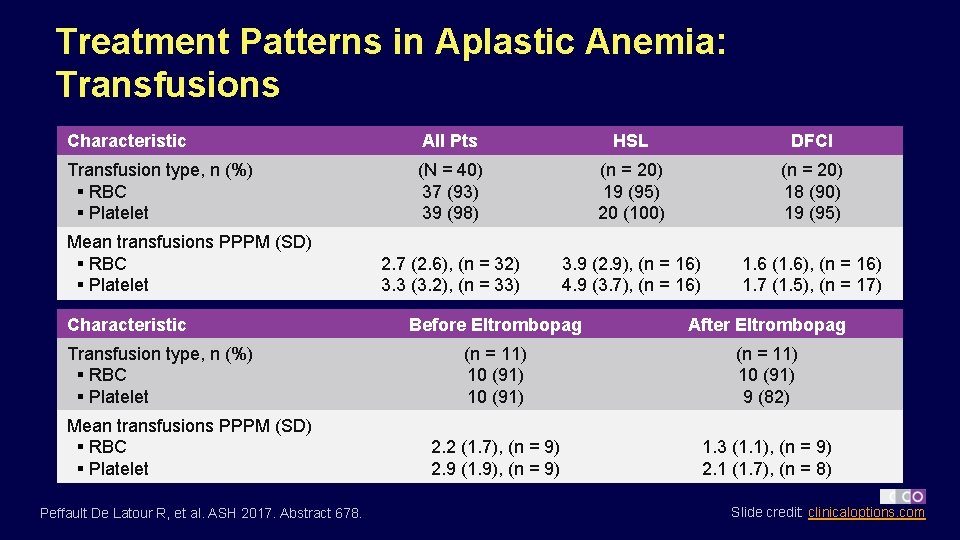
Treatment Patterns in Aplastic Anemia: Transfusions Characteristic All Pts HSL DFCI Transfusion type, n (%) § RBC § Platelet (N = 40) 37 (93) 39 (98) (n = 20) 19 (95) 20 (100) (n = 20) 18 (90) 19 (95) 2. 7 (2. 6), (n = 32) 3. 3 (3. 2), (n = 33) 3. 9 (2. 9), (n = 16) 4. 9 (3. 7), (n = 16) 1. 6 (1. 6), (n = 16) 1. 7 (1. 5), (n = 17) Mean transfusions PPPM (SD) § RBC § Platelet Characteristic Transfusion type, n (%) § RBC § Platelet Mean transfusions PPPM (SD) § RBC § Platelet Peffault De Latour R, et al. ASH 2017. Abstract 678. Before Eltrombopag After Eltrombopag (n = 11) 10 (91) 9 (82) 2. 2 (1. 7), (n = 9) 2. 9 (1. 9), (n = 9) 1. 3 (1. 1), (n = 9) 2. 1 (1. 7), (n = 8) Slide credit: clinicaloptions. com

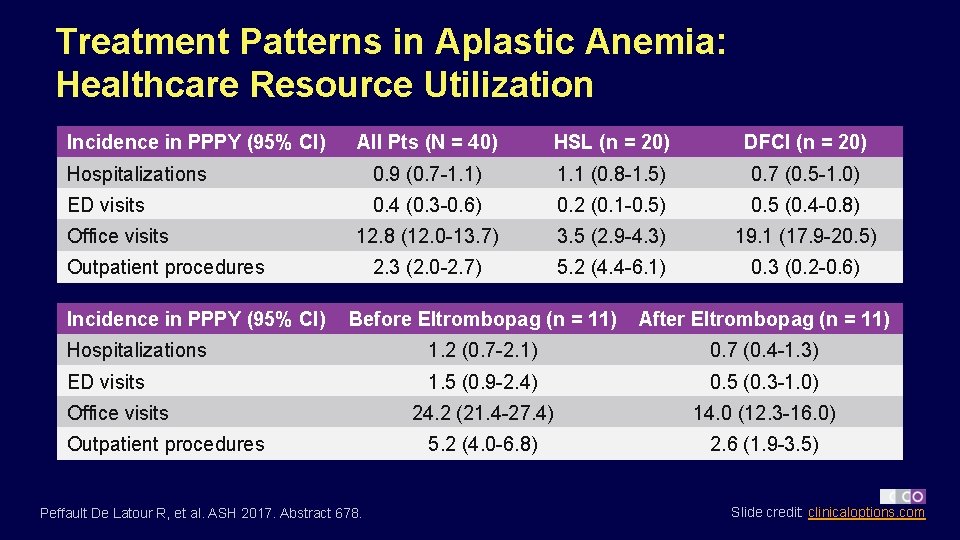
Treatment Patterns in Aplastic Anemia: Healthcare Resource Utilization Incidence in PPPY (95% CI) All Pts (N = 40) HSL (n = 20) DFCI (n = 20) Hospitalizations 0. 9 (0. 7 -1. 1) 1. 1 (0. 8 -1. 5) 0. 7 (0. 5 -1. 0) ED visits 0. 4 (0. 3 -0. 6) 0. 2 (0. 1 -0. 5) 0. 5 (0. 4 -0. 8) 12. 8 (12. 0 -13. 7) 3. 5 (2. 9 -4. 3) 19. 1 (17. 9 -20. 5) 2. 3 (2. 0 -2. 7) 5. 2 (4. 4 -6. 1) 0. 3 (0. 2 -0. 6) Office visits Outpatient procedures Incidence in PPPY (95% CI) Before Eltrombopag (n = 11) After Eltrombopag (n = 11) Hospitalizations 1. 2 (0. 7 -2. 1) 0. 7 (0. 4 -1. 3) ED visits 1. 5 (0. 9 -2. 4) 0. 5 (0. 3 -1. 0) 24. 2 (21. 4 -27. 4) 14. 0 (12. 3 -16. 0) 5. 2 (4. 0 -6. 8) 2. 6 (1. 9 -3. 5) Office visits Outpatient procedures Peffault De Latour R, et al. ASH 2017. Abstract 678. Slide credit: clinicaloptions. com

Treatment Patterns in Aplastic Anemia: Conclusions § In adults with severe AA and insufficient response to immunosuppression – Nearly all (98%) pts received RBC or PLT transfusions, 75% received ≥ 1 HSCT – Most pts received ATG with a calcineurin inhibitor (95%) for primary therapy and either eltrombopag (28%) or androgens (15%) as secondary therapy before HSCT – Mean healthcare resource utilization before HSCT: 0. 9 hospitalizations PPPY, 0. 4 ED visits PPPY, 12. 8 office visits PPPY – In subset receiving eltrombopag, mean number of transfusions and healthcare resource utilization rates both decreased after eltrombopag initiation vs before eltrombopag treatment § Investigators consider chart review a hypothesis-generating study limited by small pt number (due to rare incidence of disease) and data explicitly available in given medical records Peffault De Latour R, et al. ASH 2017. Abstract 678. Slide credit: clinicaloptions. com

Go Online for More CCO Coverage of ASH 2017! Short slideset summaries of all the key data Additional CME-certified analyses with expert faculty commentary on all the key studies in: § § § Leukemias Lymphomas/CLL Myeloma Nonmalignant hematology Hot topics in hematology clinicaloptions. com/oncology