Results from a Phase 2 Randomized Placebo Controlled

Results from a Phase 2 Randomized, Placebo. Controlled, Double Blind Study of the Hedgehog Pathway Antagonist IPI-926 in Patients with Advanced Chondrosarcoma Andrew J. Wagner 1, Peter Hohenberger 2, Scott Okuno 3, Mikael Eriksson 4, Shreyaskumar Patel 5, Stefano Ferrari 6, Paolo G. Casali 7, Sant P. Chawla 8, Molly Woehr 9, Robert Ross 9, Jessica O’Keefe 9, Amy Hillock 9, George Demetri 1, Peter Reichardt 10 1 Dana-Farber Cancer Institute; 2 Universitatsmedizin Mannheim; 3 Mayo Clinic; 4 Skanes Universitetssjukhus i Lund; 5 MD Anderson Cancer Center; 6 IRCCS Istituto Ortopedico Rizzoli; 7 Fondazione IRCCS Istituto Nazionale dei Tumori; 8 Sarcoma Oncology Center; 9 Infinity Pharmaceuticals; 10 Helios Klinikum Bad Saarow CTOS 2013 New York
Hedgehog Signaling Pathway • Plays a critical role in development • Inactive in most adult cells • Regulates normal chondrocyte proliferation, terminal differentiation, and endochondral bone development Inactive Activated
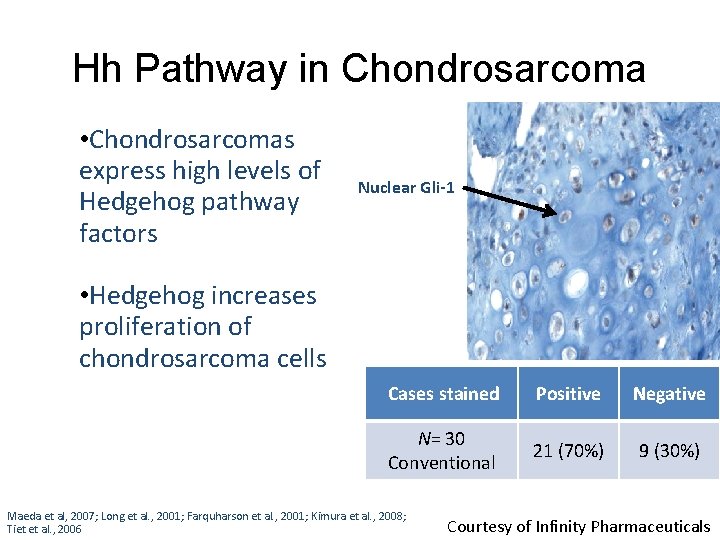
Hh Pathway in Chondrosarcoma • Chondrosarcomas express high levels of Hedgehog pathway factors Nuclear Gli-1 • Hedgehog increases proliferation of chondrosarcoma cells Cases stained Positive Negative N= 30 Conventional 21 (70%) 9 (30%) Maeda et al, 2007; Long et al. , 2001; Farquharson et al. , 2001; Kimura et al. , 2008; Tiet et al. , 2006 Courtesy of Infinity Pharmaceuticals
HH Pathway Inhibitors Block SMO No change in Gli-1, Cyclin D 1/D 2, Myc, or Bcl 2
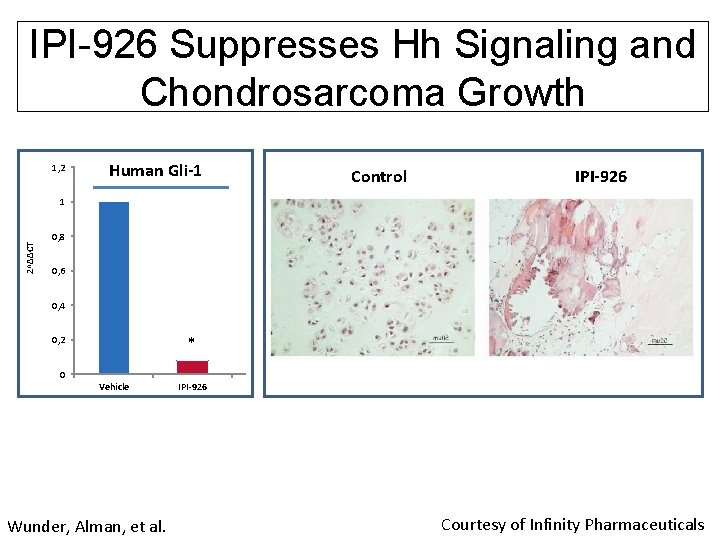
IPI-926 Suppresses Hh Signaling and Chondrosarcoma Growth 1, 2 Human Gli-1 Control IPI-926 2^ΔΔCT 1 0, 8 0, 6 0, 4 * 0, 2 0 Vehicle Wunder, Alman, et al. IPI-926 Vehicle IPI-926 Courtesy of Infinity Pharmaceuticals
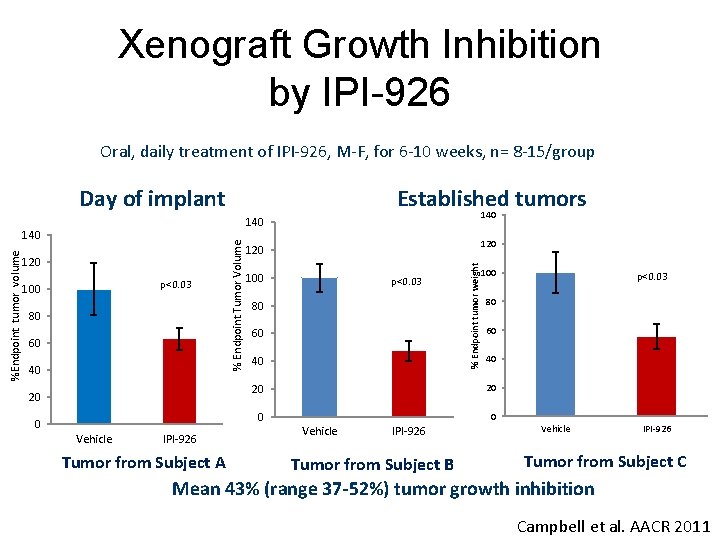
Xenograft Growth Inhibition by IPI-926 Oral, daily treatment of IPI-926, M-F, for 6 -10 weeks, n= 8 -15/group Day of implant 140 p<0. 03 100 80 60 40 20 0 Vehicle IPI-926 Tumor from Subject A 120 100 p<0. 03 80 60 40 % Endpoint tumor weight 120 % Endpoint Tumor Volume 140 %Endpoint tumor volume Established tumors 100 80 60 40 20 20 0 0 Vehicle IPI-926 Tumor from Subject B p<0. 03 Vehicle IPI-926 Tumor from Subject C Mean 43% (range 37 -52%) tumor growth inhibition Campbell et al. AACR 2011
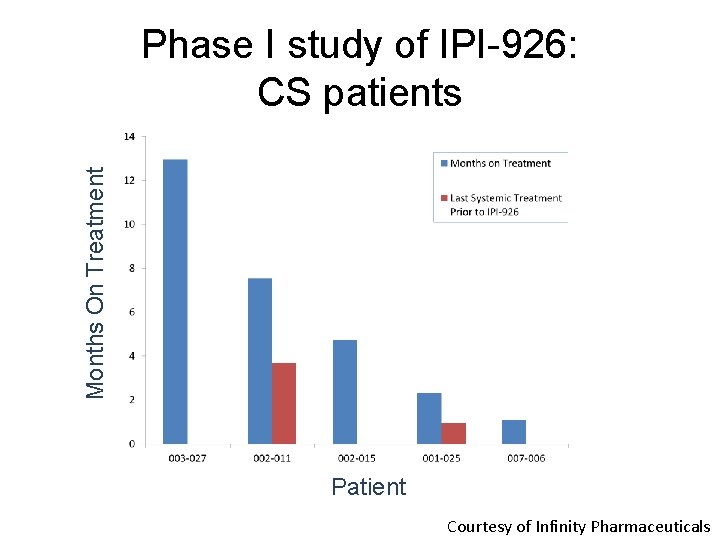
Months On Treatment Phase I study of IPI-926: CS patients Patient Courtesy of Infinity Pharmaceuticals
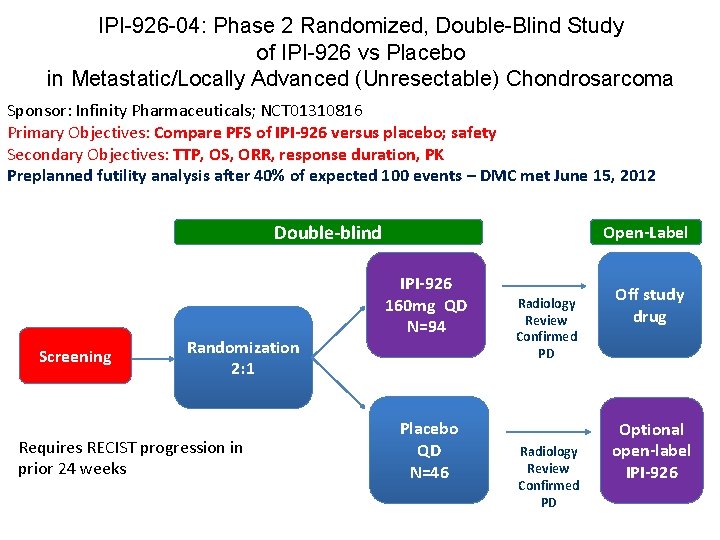
IPI-926 -04: Phase 2 Randomized, Double-Blind Study of IPI-926 vs Placebo in Metastatic/Locally Advanced (Unresectable) Chondrosarcoma Sponsor: Infinity Pharmaceuticals; NCT 01310816 Primary Objectives: Compare PFS of IPI-926 versus placebo; safety Secondary Objectives: TTP, OS, ORR, response duration, PK Preplanned futility analysis after 40% of expected 100 events – DMC met June 15, 2012 Double-blind Screening Randomization 2: 1 Requires RECIST progression in prior 24 weeks Open-Label IPI-926 160 mg QD N=94 Placebo QD N=46 Radiology Review Confirmed PD Off study drug Optional open-label IPI-926

Key Eligibility Criteria • Pathologically-diagnosed conventional chondrosarcoma • Metastasis to at least 1 location or locally advanced disease that is deemed unresectable by a surgeon • At least 1 measurable target lesion per RECIST 1. 1 • Radiographic progression of disease within 24 weeks prior to the start of screening (date ICF signed), through the screening period • Progression must be based on at least two sets of scans (CT or MRI), and as defined by RECIST 1. 1 • At least 18 years of age • ECOG performance status 0 or 1 • Life expectancy of at least 3 months
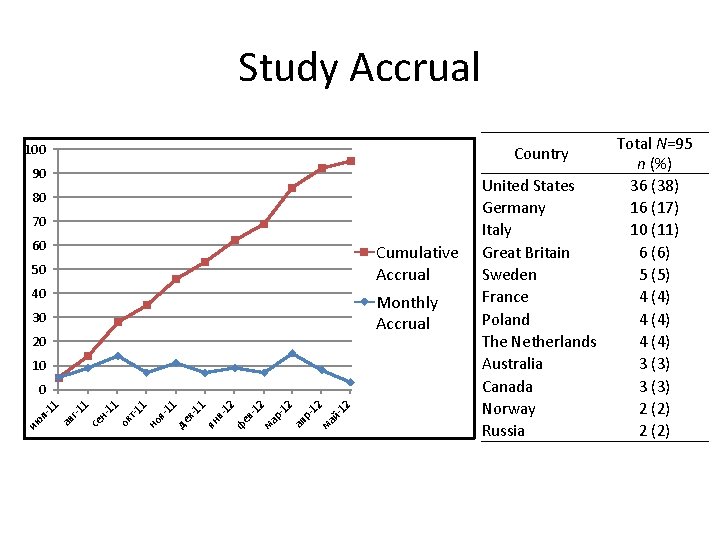
Study Accrual 100 Country 90 80 70 60 Cumulative Accrual 50 40 Monthly Accrual 30 20 10 11 де к 11 ян в 12 фе в 12 ма р12 ап р12 ма й 12 я- 1 но т-1 ок 11 1 се н- г-1 ав ию л 11 0 United States Germany Italy Great Britain Sweden France Poland The Netherlands Australia Canada Norway Russia Total N=95 n (%) 36 (38) 16 (17) 10 (11) 6 (6) 5 (5) 4 (4) 3 (3) 2 (2)
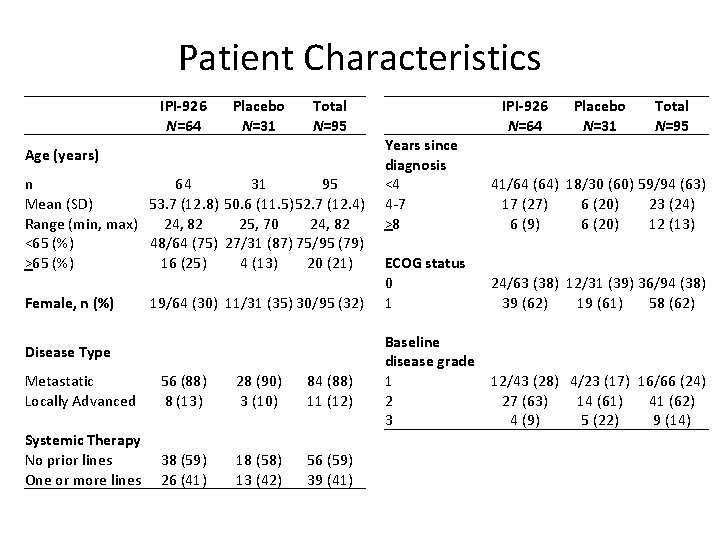
Patient Characteristics IPI-926 N=64 Placebo N=31 Total N=95 Age (years) n 64 31 95 Mean (SD) 53. 7 (12. 8) 50. 6 (11. 5)52. 7 (12. 4) Range (min, max) 24, 82 25, 70 24, 82 <65 (%) 48/64 (75) 27/31 (87) 75/95 (79) >65 (%) 16 (25) 4 (13) 20 (21) Female, n (%) 19/64 (30) 11/31 (35) 30/95 (32) Disease Type Metastatic Locally Advanced 56 (88) 8 (13) 28 (90) 3 (10) 84 (88) 11 (12) Systemic Therapy No prior lines One or more lines 38 (59) 26 (41) 18 (58) 13 (42) 56 (59) 39 (41) IPI-926 N=64 Placebo N=31 Total N=95 Years since diagnosis <4 4 -7 >8 41/64 (64) 18/30 (60) 59/94 (63) 17 (27) 6 (20) 23 (24) 6 (9) 6 (20) 12 (13) ECOG status 0 1 24/63 (38) 12/31 (39) 36/94 (38) 39 (62) 19 (61) 58 (62) Baseline disease grade 1 12/43 (28) 4/23 (17) 16/66 (24) 2 27 (63) 14 (61) 41 (62) 3 4 (9) 5 (22) 9 (14)
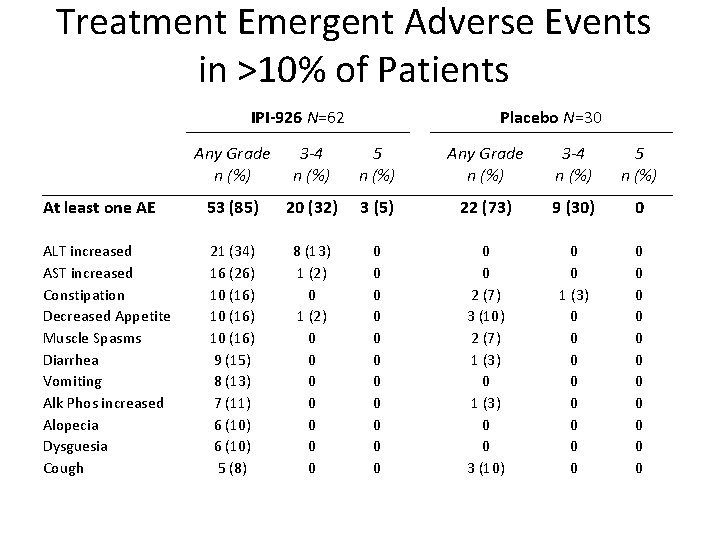
Treatment Emergent Adverse Events in >10% of Patients IPI-926 N=62 Placebo N=30 Any Grade n (%) 3 -4 n (%) 5 n (%) At least one AE 53 (85) 20 (32) 3 (5) 22 (73) 9 (30) 0 ALT increased AST increased Constipation Decreased Appetite Muscle Spasms Diarrhea Vomiting Alk Phos increased Alopecia Dysguesia Cough 21 (34) 16 (26) 10 (16) 9 (15) 8 (13) 7 (11) 6 (10) 5 (8) 8 (13) 1 (2) 0 0 0 0 0 0 2 (7) 3 (10) 2 (7) 1 (3) 0 0 3 (10) 0 0 1 (3) 0 0 0 0 0
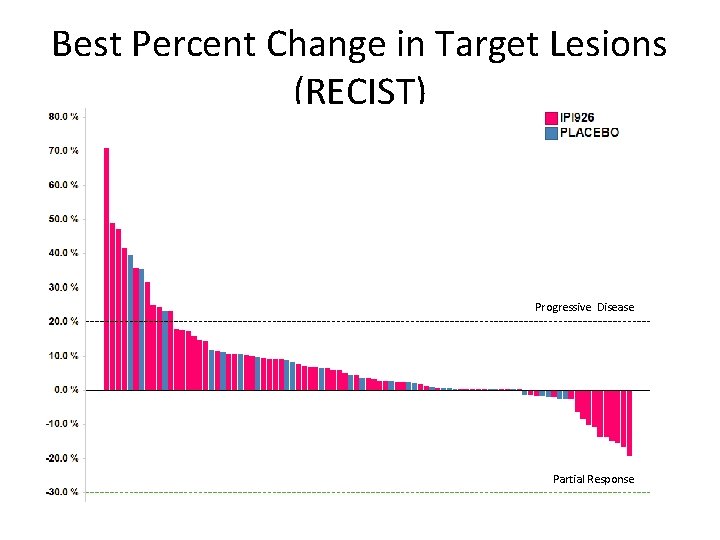
Best Percent Change in Target Lesions (RECIST) Progressive Disease Partial Response
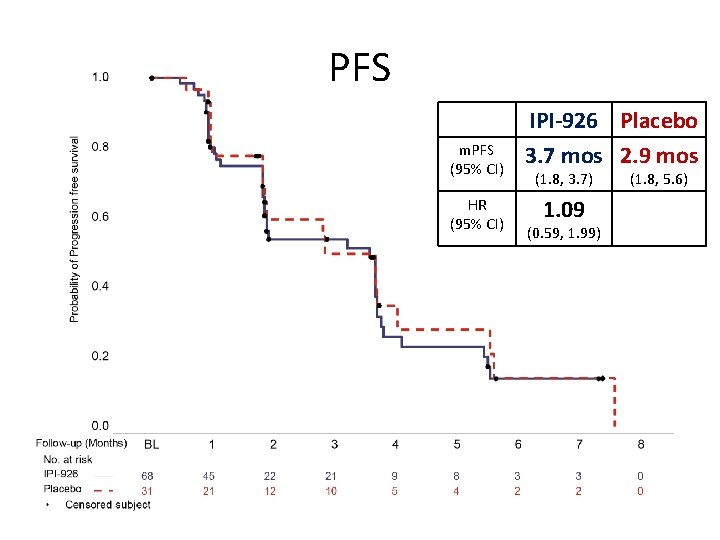
PFS m. PFS (95% CI) HR (95% CI) IPI-926 Placebo 3. 7 mos 2. 9 mos (1. 8, 3. 7) 1. 09 (0. 59, 1. 99) (1. 8, 5. 6)
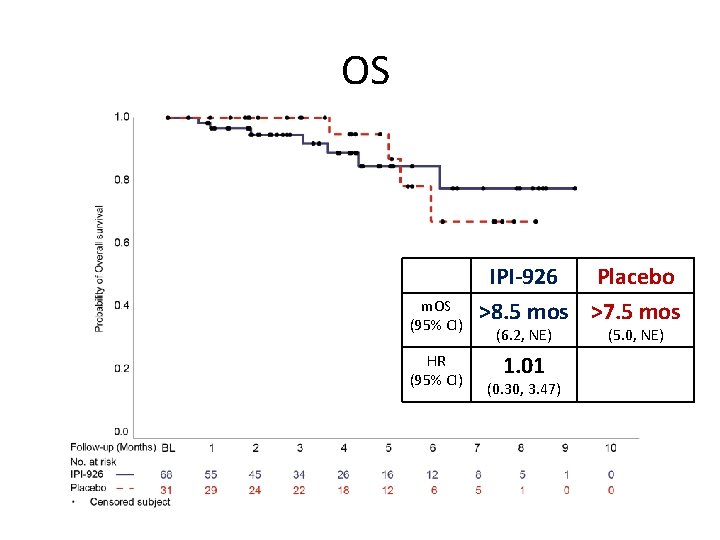
OS m. OS (95% CI) HR (95% CI) IPI-926 Placebo >8. 5 mos >7. 5 mos (6. 2, NE) 1. 01 (0. 30, 3. 47) (5. 0, NE)

Summary • Rapid accrual to randomized studies of rare diseases is feasible with world-wide collaboration • IPI-926 was generally well-tolerated when administered to patients with chondrosarcoma • There was no apparent improvement in PFS in patients with advanced, progressing chondrosarcoma • A small subset of patients treated with IPI-926 had minor reductions in tumor size
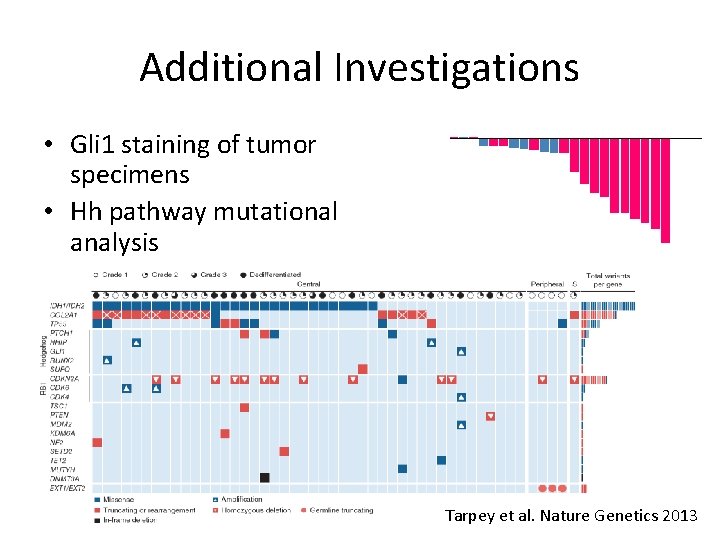
Additional Investigations • Gli 1 staining of tumor specimens • Hh pathway mutational analysis • IDH 1/IDH 2 mutational analysis, 2 HG plasma concentration, and correlation with rate of progression Tarpey et al. Nature Genetics 2013
Markman Australia Tattersall Australia Brodowicz Austria Samonigg Austria Blackstein Canada Blay France Duffaud France Le Cesne France Bompas France Hohenberger Germany Reichardt Germany Bauer Germany Ferrari Italy Casali Italy Gelderblom Netherlands Sundby-Hall Norway Mazurkiewicz Poland Rutkowski Poland Teplyakov Russia Lichinitser Russia Kudryavtseva Russia Valverde Morales. Spain Eriksson Sweden Biswas United Kingdom Whelan United Kingdom Grimer United Kingdom Cowie United Kingdom Wagner Chugh Okuno Patel Riedel Ryan von Mehren Chmielowski Jones Matushansky Meyer Seetharam Benedetto Priebat Elias Kraft Chawla Staddon Van Tine Gouw Attia USA USA USA USA USA USA Thank You Patients and their Families Study Teams Team at Infinity
- Slides: 18